research use only
Voxelotor
Cat.No.S8540
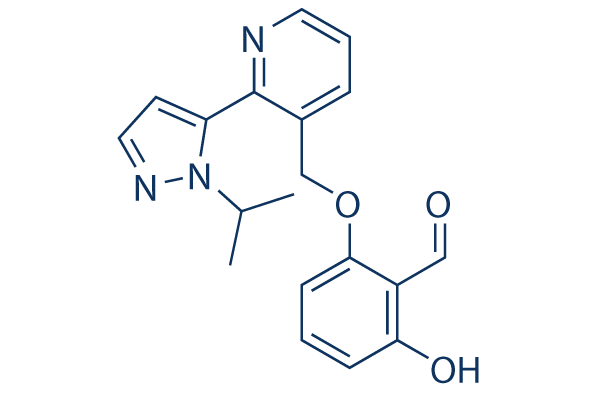
Chemical Structure
Molecular Weight: 337.37
Quality Control
Solubility
|
In vitro |
DMSO
: 67 mg/mL
(198.59 mM)
Ethanol : 5 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 337.37 | Formula | C19H19N3O3 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1446321-46-5 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | GBT440, GTx011 | Smiles | CC(C)N1C(=CC=N1)C2=C(C=CC=N2)COC3=CC=CC(=C3C=O)O | ||
Mechanism of Action
| In vitro |
GBT440 is a new potent allosteric effector of sickle cell hemoglobin that increases the affinity of hemoglobin for oxygen and consequently inhibits its polymerization when subjected to hypoxic conditions. GBT440 inhibits these isozymes(CYP 1A2, 2C8, 2C9, 2C19, 2D6, and 3A4) with IC50 ranging from 7.9 to 148 μM. It is not a substrate for either P-gp or BCRP transporters. It binds to the N-terminal a chain of Hb.
|
|---|---|
| In vivo |
GBT440 has favorable oral bioavailability of 60, 37, and 36% in rats, dogs, and monkeys, respectively, with similar blood and plasma half-lives of approximately 20 h each. T1/2 value of GBT440 in all animal species is significantly shorter than the T1/2 of red blood cells (∼20 days), which supports that binding of GBT440 to hemoglobin is a reversible process. GBT440 is currently in Phase 3 clinical trials (NCT03036813) in SCD patients. GBT440 increases haemoglobin oxygen affinity, reduces sickling and prolongs RBC half-life in a murine model of sickle cell disease. In a murine model of SCD, GBT440 extends the half-life of RBCs, reduces reticulocyte counts and prevents ex vivo RBC sickling. Importantly, oral dosing of GBT440 in animals demonstrates suitability for once daily dosing in humans and a highly selective partitioning into RBCs, which is a key therapeutic safety attribute. GBT440 shows dose proportional PK, a terminal half-life of 1.5-3 d.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06023199 | Recruiting | Sickle Cell Disease |
Inova Health Care Services|Pfizer |
October 23 2023 | Phase 2 |
| NCT05289570 | Terminated | Acute Lung Injury|End Stage Lung Disease |
Duke University |
May 3 2022 | Phase 2 |
| NCT05561140 | Active not recruiting | Sickle Cell Disease|Leg Ulcers |
Pfizer |
May 30 2022 | Phase 3 |
| NCT05018728 | Recruiting | Sickle Cell Anemia in Children |
Amy Tang|Pfizer|Emory University |
March 28 2022 | Phase 2 |
| NCT05494541 | Completed | Sickle Cell Disease |
Novartis Pharmaceuticals|Novartis |
August 30 2021 | -- |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































