research use only
Guanidine HCl
Cat.No.S4070
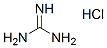
Chemical Structure
Molecular Weight: 95.53
Quality Control
Solubility
|
In vitro |
DMSO
: 19 mg/mL
(198.89 mM)
Water : 19 mg/mL Ethanol : 6 mg/mL |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 95.53 | Formula | CH5N3.HCl |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 50-01-1 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | Aminoformamidine HCl | Smiles | C(=N)(N)N.Cl | ||
Mechanism of Action
| In vitro |
Guanidine HCl is the most popular protein denaturant. Analysis of unfolding transitions by this compound provides several important parameters regarding the mechanism of conformational stability of proteins. It at low concentrations refolds acid-unfolds apomyoglobin and cytochrome c, stabilizing the molten globule state. This chemical (> 1 M) causes co-operative unfolding of the molten globule state. It is 2.8 times more effective than urea in unfolding ribonuclease but only 1.7 times more effective for lysozyme. △GH2Oapp. values of this compound are 9.7 Cal/mole for ribonuclease at pH 6.6, 6.1 for lysozyme at pH 2.9, 8.3 for α-chymotrypsin at pH 4.3, and 11.7 for β-lactoglobulin at pH 3.2.
It at millimolar concentrations, is able to causes efficient loss of the normally stable [PSI+] element from yeast cells. 5 mM of this chemical in growth media cures [PSI+] and other prions of yeast. 5 mM of this compound significantly reduces Hsp104-mediated basal and acquired thermotolerance by 30-fold and 50 fold, respectively. It also reduces the ability of Hsp104 to restore activity of thermally denatured luciferase.
|
References |
|
|---|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT03658902 | Completed | Botulism; Poisoning |
Centre Hospitalier le Mans |
January 1 2000 | -- |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































