research use only
Elimusertib (BAY-1895344) hydrochloride ATM/ATR inhibitor
Cat.No.S8666
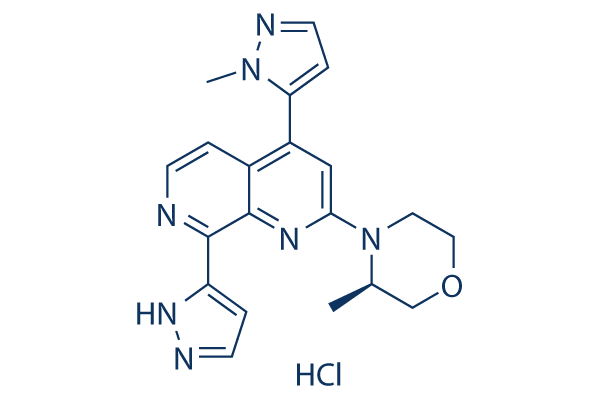
Chemical Structure
Molecular Weight: 411.89
Quality Control
| Related Targets | HDAC PARP DNA-PK WRN DNA/RNA Synthesis Topoisomerase PPAR Sirtuin Casein Kinase eIF |
|---|---|
| Other ATM/ATR Inhibitors | Ceralasertib (AZD6738) AZD1390 Berzosertib (VE-822) Lartesertib (M4076) Camonsertib (RP-3500) KU-60019 KU-55933 VE-821 AZ20 AZD0156 |
Solubility
|
In vitro |
DMSO
: 82 mg/mL
(199.08 mM)
Water : 82 mg/mL Ethanol : 82 mg/mL |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 411.89 | Formula | C20H22ClN7O |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | Download SDF | Storage of Stock Solutions |
|
||
| Synonyms | BAY-1895344 HCl | Smiles | CC1COCCN1C2=NC3=C(C=CN=C3C4=CC=NN4)C(=C2)C5=CC=NN5C | ||
Mechanism of Action
| Targets/IC50/Ki |
ATR
(Cell-free assay) 7 nM
|
|---|---|
| In vitro |
In vitro, BAY 1895344 is shown to be a very potent and highly selective ATR inhibitor (IC50 = 7 nM), which potently inhibits proliferation of a broad spectrum of human tumor cell lines (median IC50 = 78 nM). In cellular mechanistic assays BAY 1895344 potently inhibits induced H2AX phosphorylation (IC50 = 36 nM). |
| In vivo |
BAY 1895344 reveals significantly improved aqueous solubility, bioavailability across species and no activity in the hERG patch-clamp assay. It also demonstrates very promising efficacy in monotherapy in DNA damage deficient tumor models as well as combination treatment with DNA damage inducing therapies. BAY 1895344 exhibits strong in vivo anti-tumor efficacy in monotherapy in a variety of xenograft models of different indications that are characterized by DDR deficiencies, inducing stable disease in ovarian and colorectal cancer or even complete tumor remission in mantle cell lymphoma models. |
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05071209 | Active not recruiting | Recurrent Alveolar Rhabdomyosarcoma|Recurrent Ewing Sarcoma|Recurrent Lymphoma|Recurrent Malignant Solid Neoplasm|Refractory Alveolar Rhabdomyosarcoma|Refractory Ewing Sarcoma|Refractory Lymphoma|Refractory Malignant Solid Neoplasm |
National Cancer Institute (NCI) |
December 22 2021 | Phase 1|Phase 2 |
| NCT03188965 | Completed | Advanced Solid Tumor|Non-Hodgkin''s Lymphoma|Mantle Cell Lymphoma |
Bayer |
July 6 2017 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































