research use only
Samotolisib (LY3023414) PI3K inhibitor
Cat.No.S8322
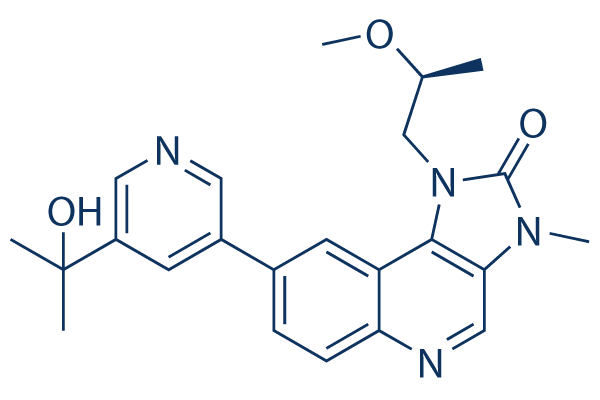
Chemical Structure
Molecular Weight: 406.48
Quality Control
| Related Targets | Akt mTOR GSK-3 ATM/ATR DNA-PK AMPK PDPK1 PTEN PP2A PDK |
|---|---|
| Other PI3K Inhibitors | GDC-0077 (Inavolisib) SAR405 Quercetin (Sophoretin) LY294002 XL147 analogue Tersolisib (STX-478) Buparlisib (BKM120) 740 Y-P (PDGFR 740Y-P) GO-203 TFA Eganelisib (IPI-549) |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| KB-8-5-11 | qHTS assay | P-glycoprotein substrates identified in KB-8-5-11 adenocarcinoma cell line, qHTS therapeutic library screen, Potency=0.6513μM. | 31515284 | |||
| KB-3-1 | qHTS assay | P-glycoprotein substrates identified in KB-3-1 adenocarcinoma cell line, qHTS therapeutic library screen. | 31515284 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 81 mg/mL
(199.27 mM)
Ethanol : 6 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 406.48 | Formula | C23H26N4O3 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1386874-06-1 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | GTPL8918 | Smiles | CC(CN1C2=C3C=C(C=CC3=NC=C2N(C1=O)C)C4=CC(=CN=C4)C(C)(C)O)OC | ||
Mechanism of Action
| Targets/IC50/Ki |
class I PI3K isoforms
mTOR kinase
DNA-PK
|
|---|---|
| In vitro |
Samotolisib (LY3023414) shows high solubility across a wide pH range. In vitro, inhibition of PI3K/AKT/mTOR signaling by this compound causes G1 cell-cycle arrest and resulted in broad antiproliferative activity in cancer cell panel screens. In cell-based assays, its inhibition of PI3K and mTOR is assessed in the PTEN-deficient U87 MG glioblastoma cell line. It inhibits the phosphorylation of AKT at position T308 downstream of PI3K at an IC50 of 106 nM. Similarly, it inhibits phosphorylation of AKT at position S473 (IC50 = 94.2 nM) by mTORC2 as well as phosphorylation of mTORC1 kinase targets p70S6K (position T389; IC50 =10.6 nM) and 4E-BP1 (positions T37/46; IC50 = 187 nM). The downstream phosphorylation of S6RP at positions pS240/244 (IC50 = 19.1 nM) by p70S6K was inhibited as well, indicating target inhibition along the entire PI3K/AKT/mTOR pathway by this compound.
|
| In vivo |
Samotolisib (LY3023414) demonstrates high bioavailability and dose-dependent dephosphorylation of PI3K/AKT/mTOR pathway downstream substrates such as AKT, S6K, S6RP, and 4E-BP1 for 4 to 6 hours in vivo, reflecting the drug's half-life of 2 hours. Intermittent target inhibition is sufficient for its antitumor activity. This compound shows time- and dose-dependent target inhibition in vivo. It is currently being evaluated in phase 1 and 2 trials for the treatment of human malignancies.
|
References |
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | pS6K1 / S6K1 / pAKT-S473 / AKT1 / pERK / ERK p-mTOR / mTOR / ATG5 / Beclin-1 / p62 |
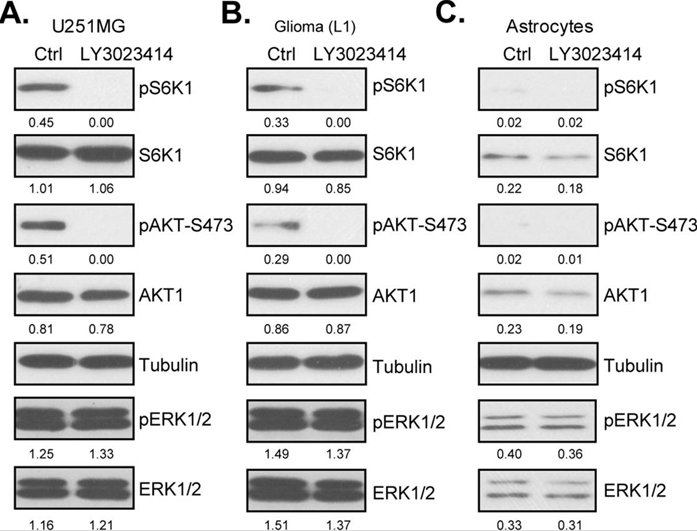
|
29228741 |
| Growth inhibition assay | Cell viability |
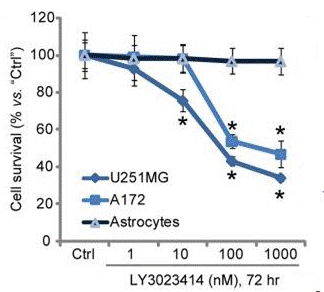
|
29228741 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT02575703 | Completed | Healthy |
Eli Lilly and Company |
October 2015 | Phase 1 |
| NCT02536586 | Completed | Neoplasm |
Eli Lilly and Company |
September 2015 | Phase 1 |
| NCT02443337 | Terminated | Non-small Cell Lung Cancer Metastatic |
Eli Lilly and Company|SCRI Development Innovations LLC |
July 2015 | Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































