research use only
Wortmannin (SL-2052) PI3K inhibitor
Cat.No.S2758
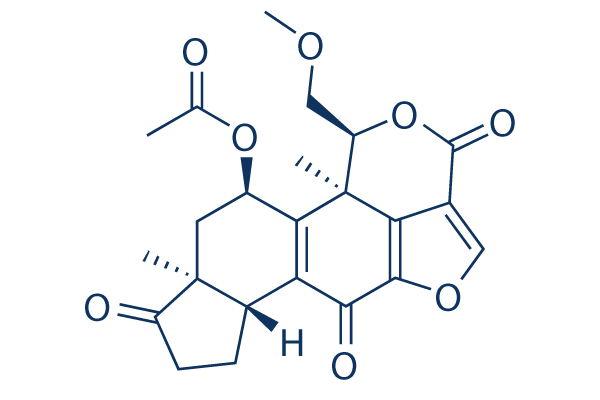
Chemical Structure
Molecular Weight: 428.43
Quality Control
| Related Targets | Akt mTOR GSK-3 ATM/ATR DNA-PK AMPK PDPK1 PTEN PP2A PDK |
|---|---|
| Other PI3K Inhibitors | GDC-0077 (Inavolisib) SAR405 Quercetin (Sophoretin) LY294002 XL147 analogue Tersolisib (STX-478) Buparlisib (BKM120) 740 Y-P (PDGFR 740Y-P) GO-203 TFA Eganelisib (IPI-549) |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| Mel-HO | Apoptosis Assay | 4/8 μM | 24 h | enhances TRAIL-induced apoptosis | 24113173 | |
| A-375-TS | Apoptosis Assay | 4/8 μM | 24 h | enhances TRAIL-induced apoptosis | 24113173 | |
| A-375 | Apoptosis Assay | 4/8 μM | 24 h | enhances TRAIL-induced apoptosis | 24113173 | |
| Huh7 | Function Assay | 3 μM | 1 h | reduces the virus entry into the cells | 24184196 | |
| BEL/FU | Function Assay | 1 mM | 24 h | decreases protein levels of the PI3K/Akt pathway | 24232099 | |
| HCT 116 | Function Assay | 100 nM | 24 h | attenuates the colonies of the tumor cells with upregulation of Akt1 | 24297510 | |
| HepG2 | Function Assay | 100 nM | 24 h | attenuates the colonies of the tumor cells with upregulation of Akt1 | 24297510 | |
| SW480 | Function Assay | 150nM | 20 h | DMSO | reduces cellular accumulation of β-catenin | 24324366 |
| HEK-293 | Function Assay | 150nM | 16 h | DMSO | decreases CRT activity | 24324366 |
| 5637 | Apoptosis Assay | 10 μM | 40 min | reverses p21WAF1 expression, CDK expression, and cell inhibition induced by fucoidan | 24333868 | |
| MG-63 | Apoptosis Assay | 10 µM | 12 h | enhances DP-induced apoptosis | 24358301 | |
| H1975 | Function Assay | 10 μM | 1 h | DMSO | decreases cellular phospho-AKT protein levels | 24447935 |
| H520 | Function Assay | 10 μM | 1 h | DMSO | decreases cellular phospho-AKT protein levels | 24447935 |
| HepG2 | Function Assay | 200 nM | 0.5 h | attenuates FoxO phosphorylation | 24535192 | |
| HL-60 | Function Assay | 0.1 μM | 72 h | blocks cell differentiation | 24607273 | |
| SK-N-LO | Function Assay | 100 nM | 0.5 h | decreases the stimulant effects of on Akt phosphorylation | 24654606 | |
| A549 | Function Assay | 10 μm | 16 h | DMSO | modulates the IAV replication and causes retention of NP in the nucleus. | 24802111 |
| A549 | Growth Inhibition Assay | 3 µM | 2 h | suppresses Akt and GSK3β activation, S-phase arrest, cell apoptosis and caspase-3 activation | 24847863 | |
| HepG2 | Function Assay | 100 nM | 0.5 h | DMSO | blocks MA-induced Akt phosphorylation | 24863350 |
| MO59J | Apoptosis Assay | 10 μM | 24 h | DMSO | increases the DSB level induced by or | 24953561 |
| MO59K | Apoptosis Assay | 10 μM | 24 h | DMSO | increases the DSB level induced by or | 24953561 |
| MO59J | Cytotoxicity Assay | 5 μM | 7 d | DMSO | enhances the cytotoxicity of or | 24953561 |
| MO59K | Cytotoxicity Assay | 5 μM | 7 d | DMSO | enhances the cytotoxicity of or | 24953561 |
| HT-29 | Growth Inhibition Assay | 1.5 µM | 96 h | decreases cell growth which can be inhibited by KYNA | 25012123 | |
| MCF7 | Function Assay | 100 nM | 24 h | eliminates E2-induced ARE-Luc activity | 25172557 | |
| MDA-MB-231 | Apoptosis Assay | 1 μM | 48 h | DMSO | decreases the cell survival treated with 25 μM of F1 or F2 | 25300932 |
| APRE-19 | Apoptosis Assay | 5 μM | 24 h | abolishes FLZ-mediated pro-survival/anti-apoptosis activity | 25329617 | |
| HUVECs | Cytotoxicity Assay | 100 nM | 24 h | attenuates the abrogative effects of calycosin on VRI-induced cytotoxicity | 25450186 | |
| H1703 | Growth Inhibition Assay | 2.5 μM | 1-4 d | DMSO | enhances cell growth inhibition treatment with | 25490383 |
| A459 | Growth Inhibition Assay | 2.5 μM | 1-4 d | DMSO | enhances cell growth inhibition treatment with | 25490383 |
| Mel-HO-TS | Apoptosis Assay | 4/8 μM | 24 h | enhances TRAIL-induced apoptosis | 24113173 | |
| MeWo | Apoptosis Assay | 4/8 μM | 24 h | enhances TRAIL-induced apoptosis | 24113173 | |
| Mel-2a | Apoptosis Assay | 4/8 μM | 24 h | enhances TRAIL-induced apoptosis | 24113173 | |
| MDA-MB-231 | Function Assay | 0–400 nM | 4 h | suppresses Akt phosphorylation in a dose-dependent manner | 22906259 | |
| MDA-MB-231 | Function Assay | 400 nM | 4 h | decreases MMP-9 and IL-8 protein in a dose-dependent manner | 22906259 | |
| Jurkat | Growth Inhibition Assay | 0.25-1.25 μM | 24/48 h | DMSO | inhibits cell proliferation in both time- and dose- dependent manner | 19757185 |
| Namalwa | Growth Inhibition Assay | 0.25-1.25 μM | 24/48 h | DMSO | inhibits cell proliferation in both time- and dose- dependent manner | 19757185 |
| Jurkat | Apoptosis Assay | 0.25-1.25 μM | 24/48 h | DMSO | induces cell apoptosis in both time- and dose- dependent manner | 19757185 |
| Namalwa | Apoptosis Assay | 0.25-1.25 μM | 24/48 h | DMSO | induces cell apoptosis in both time- and dose- dependent manner | 19757185 |
| K562 | Growth Inhibition Assay | 24 h | IC50=25±0.14 nM | 19662361 | ||
| SW1990 | Function Assay | 0.01-1 μM | 1 h | inhibits HA-induced Akt phosphorylation | 19469020 | |
| RT112 | Growth Inhibition Assay | 10 μM | 24 h | DMSO | decreases the proportion of G2/M cells | 18787832 |
| MHG-U1 | Growth Inhibition Assay | 10 μM | 24 h | DMSO | decreases the proportion of G2/M cells | 18787832 |
| SMMC-7721 | Apoptosis Assay | 200 nM | 24 h | increases CHX-induced apoptosis | 17557191 | |
| SMMC-7721 | Function Assay | 200 nM | 24 h | up-regulates β1,4GT1 expression | 17557191 | |
| HeLa | Function Assay | 100 nM | 1 h | alters the morphology of the transferrin recycling compartment | 16890915 | |
| MRC5VI | Function Assay | 12.5 mM | 0.5 h | DMSO | abolishes the Ser473/Thr308 phosphorylation of AktPKB | 16227394 |
| AT5BIVA | Function Assay | 12.5 mM | 0.5 h | DMSO | abolishes the Ser473/Thr308 phosphorylation of AktPKB | 16227394 |
| M059J | Function Assay | 12.5 mM | 0.5 h | DMSO | abolishes the Ser473/Thr308 phosphorylation of AktPKB | 16227394 |
| HeLa | Function Assay | 12.5 mM | 0.5 h | DMSO | abolishes the Ser473/Thr308 phosphorylation of AktPKB | 16227394 |
| N2a | Apoptosis Assay | 0.1-10 μM | 2 h | induces decreased cell viability in a concentration-dependent manner | 15842767 | |
| Jurkat | Kinase Assay | IC50 of 24 nM | 15664519 | |||
| Sf9 | Function assay | Inhibition of human PI3Kalpha expressed in Sf9 cells by fluorescent polarization assay, IC50 = 0.012 μM. | 21121631 | |||
| HeLa | Function assay | Inhibition of AX-7503 binding to recombinant Plk3 expressed in HeLa cells by Western blot, IC50 = 0.049 μM. | 17135248 | |||
| HeLa | Function assay | Binding affinity for DNA dependent protein kinase isolated from HeLa cells; Range is 20-120, Ki = 0.12 μM. | 15658870 | |||
| HeLa | Function assay | Binding affinity for Phosphatidylinositol 3-kinase isolated from HeLa cells; Range is 20-120, Ki = 0.12 μM. | 15658870 | |||
| A549 | Function assay | Inhibition of Plk3 in human A549 cells assessed as casein substrate phosphorylation by Western blot, IC50 = 0.22 μM. | 17135248 | |||
| A549 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human A549 cells after 48 hrs by SRB method, IC50 = 11.4 μM. | 18630894 | ||
| GM00637 | Function assay | 1 uM | Inhibition of recombinant Plk3 expressed in human GM00637 cells at 1 uM assessed as decrease in p53 serine-20 phosphorylation | 17135248 | ||
| MDA-MB-231 | Function assay | 1 to 10 uM | 24 hrs | Inhibition of PI3K in human MDA-MB-231 cells assessed as inhibition of AKT phosphorylation at 1 to 10 uM after 24 hrs by Western blot analysis | 24828286 | |
| HeLa | Function assay | 100 nM | 10 mins | Inhibition of PI3K in human HeLa cells assessed as reduction in EGF-stimulated AKT phosphorylation at S473 at 100 nM preincubated for 10 mins followed by EGF stimulation measured after 5 mins by Western blot analysis | 30380865 | |
| HeLa | Function assay | 100 nM | 10 mins | Inhibition of PI3K in human HeLa cells assessed as reduction in EGF-stimulated AKT phosphorylation at T308 at 100 nM preincubated for 10 mins followed by EGF stimulation measured after 5 mins by Western blot analysis | 30380865 | |
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 85 mg/mL
(198.39 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 428.43 | Formula | C23H24O8 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 19545-26-7 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | KY 12420, SL-2052, BRN 0067676, NSC 627609 | Smiles | CC(=O)OC1CC2(C(CCC2=O)C3=C1C4(C(OC(=O)C5=COC(=C54)C3=O)COC)C)C | ||
Mechanism of Action
| Targets/IC50/Ki |
PI3K
(Cell-free assay) 3 nM
DNA-PK
(Cell-free assay) 16 nM
ATM
(Cell-free assay) 150 nM
MLCK
(Cell-free assay) 170 nM
|
|---|---|
| In vitro |
The inhibition of MLCK by Wortmannin is not affected by calmodulin or peptide substrat, while reduced by high concentration of ATP. This compound directly interacts with the catalytic domain of MLCK and leads to an irreversible loss of the enzyme activity. It has no inhibitory to cAMP-dependent protein kinase, cGMP-dependent protein kinase, and calmodulin-dependent protein kinase II, and has little effect on protein kinase C activity. This inhibitor inhibits N-formylmethionyl-leucylphenylalanine (fMLP)-stimulated PtdInsP3 (phosphatidylinositol 3,4,5-trisphosphate) formation with IC50 of 5 nM and this inhibition is completely abolished when pretreated with 100 nM of this compound in human neutrophils, with increased PtdInsP2 levels and no effects on cellular PtdInsP and PtdIns contents. It could develop oscillatory changes in F-actin content and does not inhibit fMLP-stimulated actin polymerization in neutrophils. This chemical irreversibly inhibits phosphatidylinositol 3-kinase (PI3-kinase) activity with binding to the 110-kDa protein (IC50 of 3 nM) and has no effect PI4-kinase in RBL-2H3 cells. It also inhibits leukotriene release, with no effect on the activation of the tyrosine kinase Lyn. This compound completely abolishes the induced hexose uptake in isolated rat adipocytes at 0.1 μM, without impairing stimulated lipolytic activity. It suppresses induced production of nitric oxide by 50% at 500 nM in human umbilical vein endothelial cells, which is in response to IGF-1. This chemical suppresses DNA double strand break (DSB) repair and has no effect on DSB levels or the kinetics of single strand break (SSB) repair in Chinese hamster ovary cells at 50 μM. It could potentiate ionizing radiation (IR)-induced cytotoxicity with no toxicity by itself. This inhibitor inhibits polo-like kinase (PLK1) activity IC50 of 24 nM in intact G2/M-arrested cells. It increases Toll-like receptor (TLR)-mediated accumulation of IL-6 in human macrophages with EC50 of 50 nM. Meanwhile this compound significantly enhances TLR-mediated inducible nitric-oxide synthase (iNOS) expression and nitrite accumulation in mouse macrphages. It activates the nuclear factor-κB and up-regulates the cytokine mRNA production. This chemical also inhibits Polo-like kinase (PlK) 1 and PlK3, which play important roles in mitosis. Its treatment could lead to a reduction in phosphorylation of p53 on serine 20 induced by DNA damage. It suppresses hyaluronan-induced Akt phosphorylation and cell motility/migration in SW1990 cells. |
| Kinase Assay |
MLCK assay
|
|
MLCK activity is assayed with peptide substrate (KKRPQRATSNVFS-NH2) or myosin light chain. The peptide substrate (24 μM) is phosphorylated in a reaction mixture containing 25 mM Tris-HC1 (pH 7.5), 0.5 mg/mL bovine serum albumin, 4 mM MgCl2, 0.5 mM CaCl2, 2.6 nM calmodulin, 1.5 nM MLCK, and 400 μM ATP in a final volume of 0.25 mL. After a 10-min preincubation at 28 ºC without ATP, the reaction is started by addition of ATP at 28 ºC and terminated by the addition of 0.1 mL of 10% (v/v) acetic acid after 30 min. The mixture is analyzed by high performance liquid chromatography: column, Unisil Pack 5C18 4.6 X 150 mm; solvent, 18% (v/v) acetonitrile, 0.1% (v/v) trifluoroacetic acid in water; flow rate, 1.0 mL/min; temperature, 40 ºC; detection, absorbance at 220 nm. Percent of reaction is calculated from the ratio of peak areas of phosphorylated form to those of unphosphorylated form. Specific activity measured under the conditions described above is 0.81 μmol/min/mg. Myosin light chain (108 μg/mL) is phosphorylated in a reaction mixture containing 25 mM Tris-HC1 (pH 7.5), 0.5 mg/mL bovine serum albumin, 4 mM MgCl2, 0.5 mM CaCl2, 4.2 nM calmodulin, 0.92 nM enzyme, and 10 μM[γ-32P]ATP (100-900 cpm/pmol) in a final volume of 0.25 mL. After a 3-min preincubation at 30 ºC without ATP, the reaction is started by the addition of [γ-32P]ATP at 30 ºC and stopped by the addition of 0.125 mL of trichloroacetic acid after 5 min. The acid-precipitable materials are collected on a nitrocellulose membrane filter and washed with four 1-mL aliquots of 5% (v/v) trichloroacetic acid. The radioactivity on the filter is measured in a toluene scintillation fluid, using a Packard Tri-Carb liquid scintillation spectrometer Model 4530. The specific activity measured under the conditions is 1.23 μmol/min/mg.
|
|
| In vivo |
Wortmannin inhibits peritoneal metastasis of SW1990 in mice at 1 mg/kg, without any weight loss. This compound inhibits phosphatidylinositide 3-kinase-protein kinase B (PKB)/Akt phosphorylation in both normal tissues (lung, heart and brain homogenates) and tumor tissue in mice, without mortality or acute toxicity at 0.7 mg/kg. Combination with LY188011, this chemical significantly increases apoptosis and inhibit tumor growth in orthotopic tumor, while both monotherapy could not. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
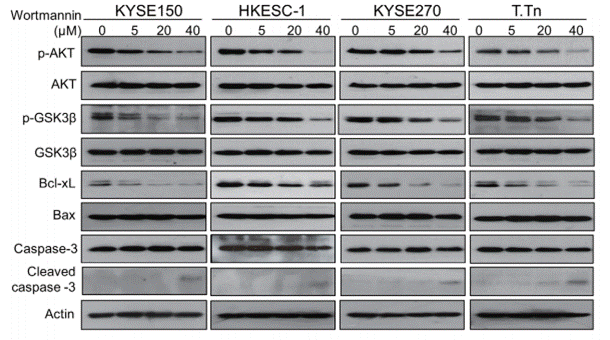
| Western blot | p-AKT / AKT / p-GSK3β / GSK3β / Bcl-xl / Bax / Caspase-3 / Cleaved caspase-3 |
|
25344912 |
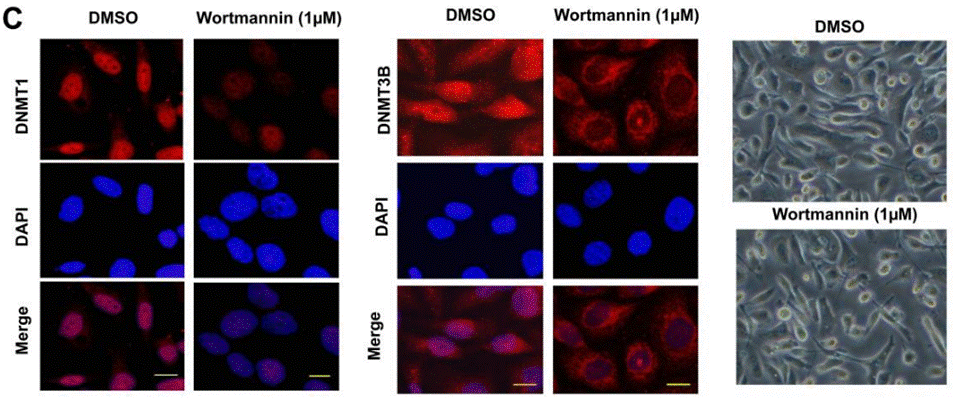
| Immunofluorescence | DNMT1 |
|
24001151 |
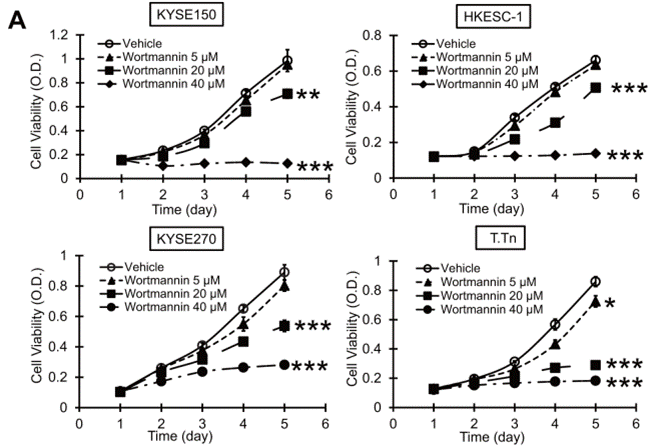
| Growth inhibition assay | Cell viability |
|
25344912 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































