research use only
ITF-2357 (Givinostat) Hydrochloride Monohydrate HDAC inhibitor
Cat.No.S2170
Chemical Structure
Molecular Weight: 475.97
Quality Control
Solubility
|
In vitro |
DMSO
: 95 mg/mL
(199.59 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 475.97 | Formula | C24H27N3O4.HCl.H2O |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 732302-99-7 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | Givinostat (ITF2357) | Smiles | CCN(CC)CC1=CC2=C(C=C1)C=C(C=C2)COC(=O)NC3=CC=C(C=C3)C(=O)NO.O.Cl | ||
Mechanism of Action
| Features |
An orally active, potent inhibitor of histone deacetylases (HDACs).
|
|---|---|
| Targets/IC50/Ki |
HD1-B
(Cell-free assay) 7.5 nM
HD2
(Cell-free assay) 10 nM
HD1-A
(Cell-free assay) 16 nM
|
| In vitro |
In LPS-stimulated cultured human peripheral blood mononuclear cells (PBMCs), ITF2357 reduces the release of TNFα, IL-1α, IL-1β, and IFNγ, with IC50 of 10-25 nM, respectively. Using the combination of IL-12 plus IL-18, ITF2357 reduces IFNγ and IL-6 production with IC50 of 12.5-25 nM, independent of decreased IL-1 or TNFα. ITF2357 is cytotoxic in multiple myeloma (MM) cell lines (RPMI8226, NCI-H929, JJN3, KMS 11, KMS 12, KMS 18, and KMS 20) and acute myelogenous leukemia (AML) cell lines (HL-60, THP-1, U937, KASUMI, KG-1, and TF-1), with IC50 of 200 nM. ITF2357 activates the intrinsic apoptotic pathway, upregulates p21 and downmodulates Bcl-2 and Mcl-1. ITF2357 inhibits the production of IL-6, VEGF, and IFNγ in mesenchymal stromal cells (MSCs) by 80-95%.
ITF2357 favors β-cell survival during inflammatory conditions. ITF2357 at concentrations of 25 and 250 nM increases islet cell viability, enhances insulin secretion, inhibits release of MIP-1α and MIP-2, reduces NO production and decreases apoptosis rates.
|
| Kinase Assay |
Enzymatic Assay for HDAC Inhibitory Activity of Synthetic Compounds
|
|
The assay is performed by adding 100 μL substrate (2×105 cpm), 40 μL buffer (50 mM Tris-HCl, pH 8.0, 750 mM NaCl, 5 mM PMSF, 50% glycerol) and 95 μL distilled water to the crude cellular extract (5 μL). ITF2357 (50 μL) is added to test for HDAC inhibition. The mixture is incubated overnight at room temperature and the reaction quenched by adding 50 μL of a solution containing 259 μL 37% HCl and 28 μL acetic acid in 1 mL distilled water. The [3H]acetyl residues released from the substrate are separated by organic extraction with 600 μL of ethyl acetate, 200 μL of the organic phase is added to standard scintillation fluid, and radioactivity is measured by a beta-counter. Inhibition of HDACs is expressed as the concentration inhibiting 50% of the control activity (by comparing the radioactivity of the samples containing inhibitors to that of the control containing cellular crude extract alone).
|
|
| In vivo |
ITF2357 (1-10 mg/kg) reduces LPS-induced serum TNFα and IFNγ by more than 50% in mice. Anti-CD3-induced cytokines are not suppressed by ITF2357 in PBMCs in the circulation in mice. In concanavalin-A-induced hepatitis, ITF2357 (1 or 5 mg/kg) significantly reduces liver damage.
ITF2357 (10 mg/kg) significantly prolongs survival of severe combined immunodeficient mice inoculated with the AML-PS in vivo passaged cell line.
In a mouse model of closed head injury (CHI), ITF2357 (10 mg/kg) improves neurobehavioral recovery, decreases neuronal degeneration, reduces lesion volume, and induces glial apoptosis.
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
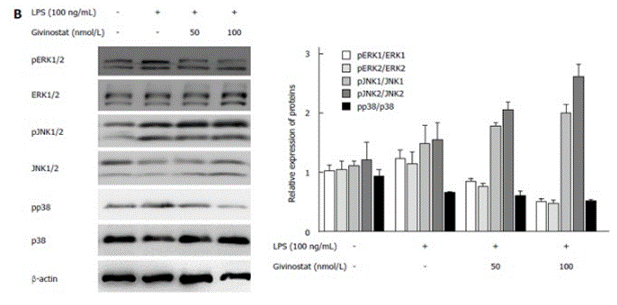
| Western blot | pERK / ERK / pJNK / JNK / pp38 / p38 SOD2(K68) / SOD2 / p53(K120) / p53(K382) / p53 / p65(K310) / p65 α-SMA / TGF-β1 / VEGF CHK1 / p53 / p21 PARP / Cleaved PARP / Pro-Caspase 7 / Cleaved caspase 7 / Pro-caspase 3 / Cleaved Caspase 3 |
|
26217084 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05933057 | Recruiting | Duchenne Muscular Dystrophy |
Italfarmaco |
February 19 2024 | Phase 3 |
| NCT05860114 | Completed | Drug Drug Interaction |
Italfarmaco |
March 21 2022 | Phase 1 |
| NCT05845567 | Completed | Drug Drug Interaction |
Italfarmaco |
March 21 2022 | Phase 1 |
| NCT01901432 | Completed | Polycythemia Vera |
Italfarmaco |
October 2013 | Phase 1|Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































