research use only
Abemaciclib (LY2835219) CDK4/6 inhibitor
Cat.No.S5716
Chemical Structure
Molecular Weight: 506.59
Quality Control
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| COLO205 cells | Function assay | 24 h | Inhibition of CDK4/6 in human COLO205 cells assessed as inhibition of Rb phosphorylation after 24 hrs by propidium iodide staining-based laser-scanning fluorescence microplate cytometric analysis, IC50=0.12 μM | 26115571 | ||
| SK-ES-1 | Proliferation assay | 96 hours | antiproliferative effects with an absolute EC50 value below 1 μmol/L | 30131386 | ||
| Cado-ES-1 | Proliferation assay | 96 hours | antiproliferative effects with an absolute EC50 value below 1 μmol/L | 30131386 | ||
| ES-7 | Proliferation assay | 96 hours | antiproliferative effects with an absolute EC50 value below 1 μmol/L | 30131386 | ||
| TC-71 | Proliferation assay | 96 hours | antiproliferative effects with an absolute EC50 value below 1 μmol/L | 30131386 | ||
| A673 | Proliferation assay | 96 hours | antiproliferative effects with an absolute EC50 value below 1 μmol/L | 30131386 | ||
| RD-ES | Proliferation assay | 96 hours | antiproliferative effects with an absolute EC50 value below 1 μmol/L | 30131386 | ||
| ES-1 | Proliferation assay | 96 hours | antiproliferative effects with an absolute EC50 value below 1 μmol/L | 30131386 | ||
| ES-3 | Proliferation assay | 96 hours | antiproliferative effects with an absolute EC50 value below 1 μmol/L | 30131386 | ||
| CHLA-258 | Proliferation assay | 96 hours | antiproliferative effects with an absolute EC50 value below 1 μmol/L | 30131386 | ||
| MHH-ES-1 | Proliferation assay | 96 hours | antiproliferative effects with an absolute EC50 value below 1 μmol/L | 30131386 | ||
| ES-8 | Proliferation assay | 96 hours | antiproliferative effects with an absolute EC50 value below 1 μmol/L | 30131386 | ||
| EW8 | Proliferation assay | 96 hours | antiproliferative effects with an absolute EC50 value below 1 μmol/L | 30131386 | ||
| ES-6 | Proliferation assay | 96 hours | antiproliferative effects with an absolute EC50 value below 1 μmol/L | 30131386 | ||
| ES-2 | Proliferation assay | 96 hours | antiproliferative effects with an absolute EC50 value below 1 μmol/L | 30131386 | ||
| SMS-CTR | Proliferation assay | 96 hours | antiproliferative effects with an absolute EC50 value below 1 μmol/L | 30131386 | ||
| ES-4 | Proliferation assay | 96 hours | antiproliferative effects with an absolute EC50 value below 1 μmol/L | 30131386 | ||
| CT26 | Cell viability assay | 96 hours | IC50=2.7 μM | 29539425 | ||
| insect cells | Function assay | 50 mins | Competitive inhibition of human CDK4/cyclin D1 expressed in insect cells assessed as phosphorylation of CTRF after 50 mins by Michaelis-Menten plot analysis in presence of ATP, Ki = 0.0006 μM. | 26115571 | ||
| insect cells | Function assay | 50 mins | Inhibition of human CDK4/cyclin D1 expressed in insect cells assessed as phosphorylation of CTRF after 50 mins by microplate scintillation counter, IC50 = 0.002 μM. | 26115571 | ||
| COLO205 | Function assay | 24 hrs | Inhibition of CDK4/6 in human COLO205 cells assessed as maximum cell cycle arrest at G1 phase after 24 hrs by propidium iodide staining-based flow cytometric analysis, INH = 6.7 μM. | 26115571 | ||
| Sf9 | Function assay | Inhibition of CDK1/Cyclin B (unknown origin) expressed in baculoviral infected insect Sf9 cells using histone H1 as substrate in presence of [gamma-33P]ATP, IC50 = 0.371 μM. | 26741853 | |||
| Sf9 | Function assay | 90 mins | Inhibition of recombinant human N-terminal GST-tagged CDK4 (4 to 303 residues)/cyclin D1 (4 to 295 residues) expressed in sf9 cells using C-terminal retinoblastoma fragment as substrate after 90 mins by [gamma-33P]ATP based microbeta scintillation countin, Ki = 0.002 μM. | 27171036 | ||
| Sf9 | Function assay | 90 mins | Inhibition of recombinant full length human N-terminal GST-tagged CDK6 (1 to 326 residues)/cyclin D1 (4 to 295 residues) expressed in sf9 cells using C-terminal retinoblastoma fragment as substrate after 90 mins by [gamma-33P]ATP based microbeta scintilla, Ki = 0.01 μM. | 27171036 | ||
| Sf21 | Function assay | Inhibition of recombinant human full length C-terminal His6-tagged CDK7/cyclin H/N-terminal GST-tagged MAT1 expressed in baculovirus infected Sf21 insect cells using cdk7 substrate peptide, Ki = 3.91 μM. | 27171036 | |||
| COLO205 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human COLO205 cells after 96 hrs by CCK-8 assay, IC50 = 0.46 μM. | 29074254 | ||
| U87MG | Antiproliferative assay | 72 hrs | Antiproliferative activity against human U87MG cells after 72 hrs by DAPI staining based assay, IC50 = 0.0481 μM. | 29247857 | ||
| Sf9 | Function assay | Inhibition of recombinant human N-terminal GST-tagged CDK4 (S4 to E303 residues)/Cyclin D1 (Q4 to I295 residues) expressed in sf9 cells using Rb protein (773 to 928 residues) as substrate in presence of [33P]-ATP by scintillation counting method, IC50 = 0.002 μM. | 29429832 | |||
| Sf9 | Function assay | 90 mins | Inhibition of recombinant human full length CDK6 expressed in baculovirus infected Sf9 insect cells using histone H1 substrate after 90 mins by ADP-Glo assay, IC50 = 0.0078 μM. | 29429832 | ||
| Sf9 | Function assay | Inhibition of recombinant human N-terminal GST-tagged CDK4 (M1 to A326 residues)/Cyclin D1 (Q4 to I295 residues) expressed in sf9 cells using Rb protein (773 to 928 residues) as substrate in presence of [33P]-ATP by scintillation counting method, IC50 = 0.01 μM. | 29429832 | |||
| Sf9 | Function assay | 90 mins | Inhibition of recombinant human full length CDK1/Cyclin D3 expressed in baculovirus infected Sf9 insect cells using histone H1 substrate after 90 mins by ADP-Glo assay, IC50 = 0.056 μM. | 29429832 | ||
| MDA-MB-231 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MDA-MB-231 cells after 72 hrs by DAPI staining based assay, IC50 = 0.191 μM. | 29429832 | ||
| COLO205 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human COLO205 cells after 96 hrs by CCK8 assay, IC50 = 0.217 μM. | 29459274 | ||
| MDA-MB-468 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human MDA-MB-468 cells after 96 hrs by CCK8 assay, IC50 = 4.808 μM. | 29459274 | ||
| COLO205 | Function assay | 0.1 to 10 uM | 24 hrs | Inhibition of CDK4 in human COLO205 cells assessed as reduction in total Rb protein level at 0.1 to 10 uM after 24 hrs by immunoblotting method | 29459274 | |
| COLO205 | Cell cycle arrest assay | up to 10 uM | 24 hrs | Cell cycle arrest in human COLO205 cells assessed as accumulation at G2/M phase up to 10 uM after 24 hrs by propidium iodide staining based FACS analysis | 29459274 | |
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
Ethanol : 8 mg/mL
DMSO
: 6 mg/mL
(11.84 mM)
Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 506.59 | Formula | C27H32F2N8 |
Storage (From the date of receipt) | 3 years -20°C powder |
|---|---|---|---|---|---|
| CAS No. | 1231929-97-7 | -- | Storage of Stock Solutions |
|
|
| Synonyms | LY2835219 | Smiles | CCN1CCN(CC1)CC2=CN=C(C=C2)NC3=NC=C(C(=N3)C4=CC5=C(C(=C4)F)N=C(N5C(C)C)C)F | ||
Mechanism of Action
| Targets/IC50/Ki |
CDK4
(Cell-free assay) 2 nM
CDK6
(Cell-free assay) 10 nM
|
|---|---|
| In vitro |
Abemaciclib highly selective inhibits the complexes CDK4/ cyclin D1 (IC50 =2 nmol/L) and CDK6/cyclin D1 (IC50 =10 nmol/L), with no activity against other CDK/cyclin complexes or cell-cycle-related kinases within the nanomolar ranges, except for inhibition of CDK9 at IC50 at least five times higher. Besides the cell-cycle dependent activity, abemaciclib is able to boost antitumor immunity by potentiating tumor antigen presentation and selectively suppressing proliferation of regulatory T (Treg) cells at the same time. Consistent with its activity against CDK4 and CDK6, abemaciclib inhibits RB phosphorylation and leads to G1 arrest in RB-proficient cell lines. In vitro, treatment with abemaciclib resulted in increased activation of human T cells and upregulated expression of antigen presentation genes in MCF-7 breast cancer cells.
|
| In vivo |
In a colorectal cancer xenograft model used to develop an integrated pharmacokinetic/pharmacodynamic model, abemaciclib can be dosed orally on a continuous schedule to achieve sustained target inhibition and demonstrates not only durable cell-cycle inhibition but also single-agent antitumor activity. Tumor growth inhibition is observed in multiple other human cancer xenograft models, including those derived from non-small cell lung cancer (NSCLC), melanoma, glioblastoma, and mantle cell lymphoma. Abemaciclib distributes across the blood–brain barrier and prolongs survival in an intracranial glioblastoma xenograft model. In human, The pharmacokinetics of abemaciclib shows a slow absorption phase with a median time from oral dose to maximum plasma concentration (tmax) ranging from 4 to 6 hours. It is extensively cleared and distributed. The mean terminal elimination half-life (t1/2) ranged from 17.4 to 38.1 hours with no apparent dose-dependent change in clearance.
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | p-Rb / Rb / p-EGFR / EGFR / p-HER2 / HER2 / p-HER3 / HER3 / p-P70S6K / P70S6K / p-S6RP / S6RP / Cyclin D1 β-catenin GSK3β / CAMKII pan CDK6 / CDK4 / AXL |
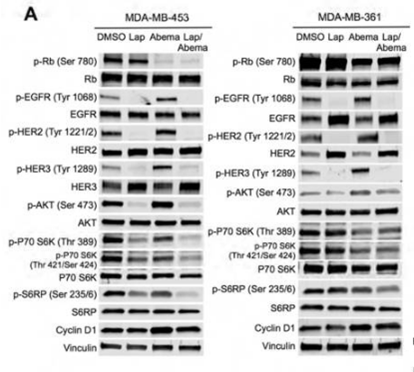
|
26977878 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06139107 | Recruiting | Breast Cancer |
Mridula George MD|Rutgers The State University of New Jersey |
June 21 2024 | Phase 1 |
| NCT06259929 | Not yet recruiting | Breast Cancer |
Fondazione Oncotech |
April 1 2024 | Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































