research use only
Cisplatin DNA Synthesis inhibitor
Cat.No.S1166
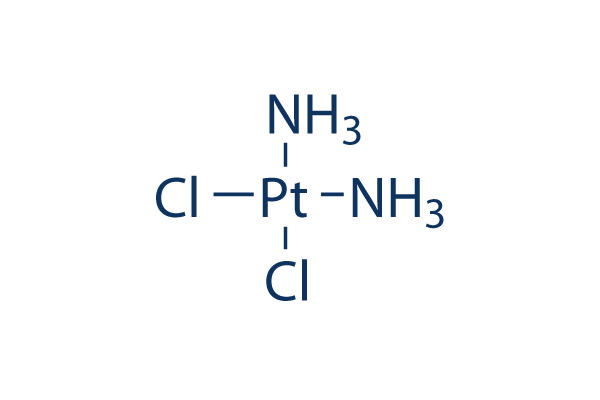
Chemical Structure
Molecular Weight: 300.05
Quality Control
Products Often Used Together with Cisplatin
| Related Targets | HDAC PARP ATM/ATR DNA-PK WRN Topoisomerase PPAR Sirtuin Casein Kinase eIF |
|---|---|
| Other DNA/RNA Synthesis Inhibitors | CX-5461 (Pidnarulex) B02 SCR7 Favipiravir (T-705) EED226 RK-33 BMH-21 Triapine (3-AP) Carmofur YK-4-279 |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| Human osteosarcoma cells (HOS, 143B, U2OS and MG‑63) | Cell cycle analysis | 2 μM | 48 h | Cisplatin treatment markedly increased the G2/M population in all cell lines. | 31059083 | |
| OVC cells (A2780, TOV-112D, and cis-A2780) | Cell Cytotoxicity Assay | 0.5, 1, 2.5, 5, 10, 20, and 50 μM | 48 h | Combination of cisplatin and MEK inhibitor cobimetinib (10 nM) enhances cell death in three ovarian cancer cell lines (A2780, TOV-112D, and cis-A2780). | 31057611 | |
| HCC cell lines HepG2 and Huh7 | Cell viability assay | 0-30 μM | 48 h | CD133+ HCC cells exhibit resistance to cisplatin. | 31056532 | |
| Saos-2 cells | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Saos-2 cells | 29435139 | ||||
| OHS-50 cells | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for OHS-50 cells | 29435139 | ||||
| SK-N-MC cells | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | ||||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMF : 15 mg/mL Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 300.05 | Formula | Cl2H6N2Pt |
Storage (From the date of receipt) | 2 years 4°C(in the dark) powder |
|---|---|---|---|---|---|
| CAS No. | 15663-27-1 | Download SDF | Storage of Stock Solutions | Solutions are unstable. Prepare fresh or purchase small, pre-packaged sizes. Repackage upon receipt. | |
| Synonyms | NSC 119875, Cisplatinum, cis-diamminedichloroplatinum II, CDDP, cis DDP, DDP | Smiles | [NH2-].[NH2-].Cl[Pt+2]Cl | ||
Mechanism of Action
| Features |
One of the most widely used and most potent chemotherapeutic agents. This product is not recommended to be dissolved in dimethylsulfoxide (DMSO).
|
|---|---|
| Targets/IC50/Ki |
DNA synthesis
(Tumor cells) |
| In vitro |
Cisplatin induces cytotoxic by interaction with DNA to form DNA adducts which activate several signal transduction pathways, including Erk, p53, p73, and MAPK, which culminates in the activation of apoptosis. This compound (30 μM) treated for 6 h induces an apparent activation of Erk in HeLa cells, which is sustained over the following 14 h period. It also shows an effective antineoplastic activity by inducing tumor cells death. It displays ability to cause renal proximal tubular cell (RPTC) apoptosis, causing cell shrinkage, a 50-fold increase in caspase 3 activity, a 4-fold increase in phosphatidylserine externalization, and 5- and 15-fold increases in chromatin condensation and DNA hypoploidy, respectively. This chemical (800 μM) causes typical features of necrosis of RPTC after treatment for 4 hr. |
| In vivo |
Cisplatin has been demonstrated to be efficient in regression tumor growth in a wide variety of animal tumors models, including head and neck cancer xenografts, cervical squamous carcinoma xenografts, testicular carcinoma xenografts, ovarian cancer xenografts, breast carcinoma xenografts, colonic carcinoma, heterotransplanted hepatoblastoma, and so on. This compound (5 mg/kg) given weekly i.v. at the day 1 and 7 induces a tumor growth inhibition (GI) of 77.5% and 85.1% of the serous xenografts Ov.Ri(C) and OVCAR-3, respectively. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | ATF3 FEN1 PD-L1 / p-MEK / MEK / p-STAT3 / STAT3 LC3B-I / LC3B-II / Beclin-1 p-AMPK / AMPK / p-mTOR / mTOR |
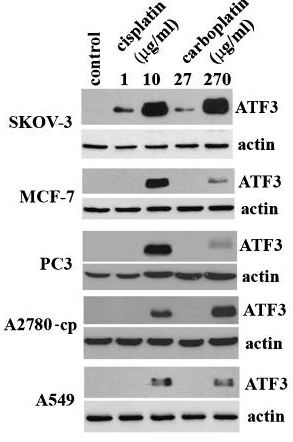
|
20651982 |
| Immunofluorescence | H2A.X / RPA γ-H2A.X / 53BP1 N-cadherin / E-cadherin / Vimentin LC3B |
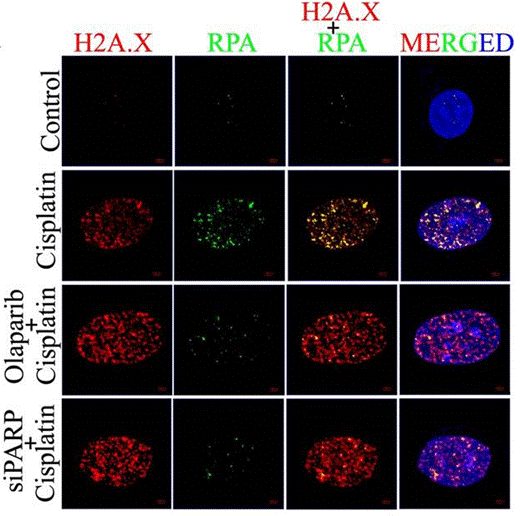
|
28993682 |
| Growth inhibition assay | Cell viability |
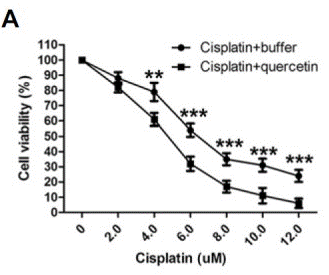
|
26062553 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06356155 | Not yet recruiting | Urothelial Carcinoma |
University of Michigan Rogel Cancer Center |
October 2024 | Phase 2 |
| NCT06393816 | Not yet recruiting | Large Cell Neuroendocrine Carcinoma of the Lung |
Centre Leon Berard|Groupe Français de Pneumo-Cancérologie |
May 2024 | Phase 2 |
| NCT06406465 | Not yet recruiting | Carcinoma Neuroendocrine|Tumor Neuroendocrine|Tumors Neuroendocrine|Neuroendocrine; Carcinoma|Small Cell; Receptors |
National Cancer Institute (NCI)|National Institutes of Health Clinical Center (CC) |
May 15 2024 | Phase 2 |
| NCT04915183 | Recruiting | Hearing Loss|Head and Neck Cancer |
National Institute on Deafness and Other Communication Disorders (NIDCD)|National Institutes of Health Clinical Center (CC) |
May 15 2024 | Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
Frequently Asked Questions
Question 1:
What is the appropriate concentration of DMF for cell culture and animal study?
Answer:
It depends on the cell type. The final concentration of DMF should be better limited to less than 0.1% if possible, or below 1%. Using saline as a vehicle for it at up to 3mg/ml is recommended. It's a suspension and can be administrated via oral gavage.






































