research use only
Amenamevir DNA/RNA Synthesis inhibitor
Cat.No.S5552
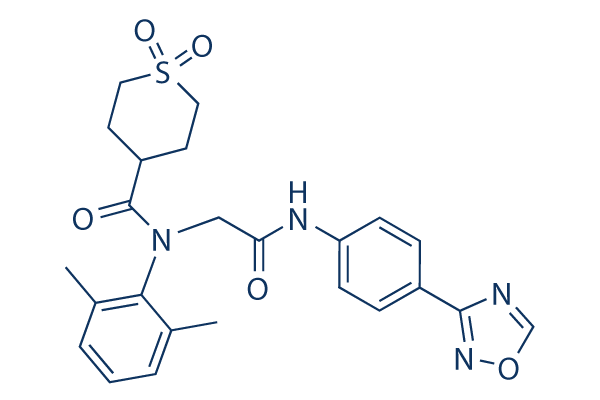
Chemical Structure
Molecular Weight: 482.55
Quality Control
| Related Targets | HDAC PARP ATM/ATR DNA-PK WRN Topoisomerase PPAR Sirtuin Casein Kinase eIF |
|---|---|
| Other DNA/RNA Synthesis Inhibitors | CX-5461 (Pidnarulex) SCR7 Favipiravir (T-705) EED226 RK-33 BMH-21 Carmofur Triapine (3-AP) YK-4-279 Halofuginone |
Solubility
|
In vitro |
|
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 482.55 | Formula | C24H26N4O5S |
Storage (From the date of receipt) | 3 years -20°C powder |
|---|---|---|---|---|---|
| CAS No. | 841301-32-4 | -- | Storage of Stock Solutions |
|
|
| Synonyms | ASP2151 | Smiles | CC1=C(C(=CC=C1)C)N(CC(=O)NC2=CC=C(C=C2)C3=NOC=N3)C(=O)C4CCS(=O)(=O)CC4 | ||
Mechanism of Action
| Targets/IC50/Ki |
helicase-primase
|
|---|---|
| In vitro |
ASP2151 inhibited the in vitro replication of HSV-1. The mean EC50s of ASP2151 against HSV-1 and HSV-2 were 14 (range, 7.7 to 20) and 30 ng/ml (range, 15 to 58), respectively.
|
| In vivo |
In the cutaneously HSV-1-infected mouse model, ASP2151 dose dependently suppressed intradermal HSV-1 growth, with the effect reaching a plateau at a dose of 30 mg/kg of body weight/day.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT02223351 | Completed | Healthy |
Maruho Europe Limited |
September 2014 | Phase 1 |
| NCT00487682 | Completed | Herpes Zoster |
Astellas Pharma Inc |
November 13 2007 | Phase 2 |
| NCT00486200 | Completed | Herpes Genitalis |
Astellas Pharma Inc |
June 21 2007 | Phase 2 |
| NCT02796118 | Completed | Healthy Subjects |
Astellas Pharma Inc |
July 2006 | Phase 1 |
| NCT02852876 | Completed | Herpes Genitalis|Herpes Zoster |
Astellas Pharma Europe Ltd.|Astellas Pharma Inc |
September 2005 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































