research use only
Favipiravir (T-705) RNA Polymerase Inhibitor
Cat.No.S7975
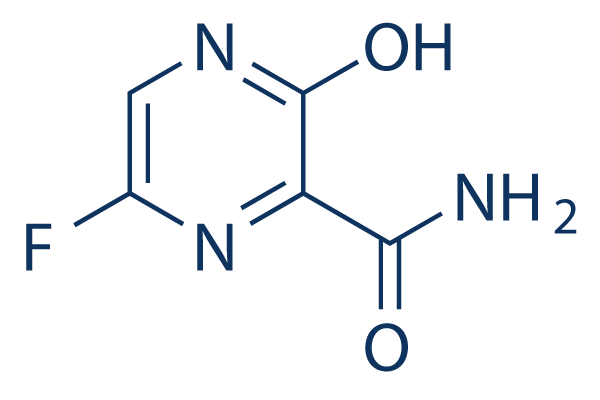
Chemical Structure
Molecular Weight: 157.1
Quality Control
| Related Targets | HDAC PARP ATM/ATR DNA-PK WRN Topoisomerase PPAR Sirtuin Casein Kinase eIF |
|---|---|
| Other DNA/RNA Synthesis Inhibitors | CX-5461 (Pidnarulex) SCR7 EED226 RK-33 BMH-21 Carmofur Triapine (3-AP) YK-4-279 Halofuginone Tegafur (FT-207) |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-, Influenza A virus (A/Ann Arbor/6/1960(H2N2)) infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic analysis using crystal violet stain, EC50 = 0.38 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-resistant , Influenza A virus (A/New York/34/2008(H1N1)) harboring M2 L26I mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic analysi, EC50 = 0.19 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-resistant,Influenza A virus (A/Florida/01/2009(H3N2)) harboring M2 S31N mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic analysis, EC50 = 0.45 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against, adamantane-resistant Influenza B virus B/Memphis/20/1996 harboring neuraminidase R152K mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic ana, EC50 = 0.57 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-, Influenza A virus (A/Swine/South Dakota/03/2008(H1N1)) infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic analysis using crystal violet, EC50 = 0.83 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-susceptible ,Influenza A virus (A/Brazil/1633/2008(H1N1)) harboring neuraminidase Q136K mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by micros, EC50 = 0.83 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-resistant,Influenza A virus (A/pdm/California/05/2009 (H1N1)) harboring M2 V28I, S31N mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by micros, EC50 = 0.83 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-resistant,Influenza A virus (A/pdm/New York/18/2009 (H1N1)) harboring M2 V28I, S31N mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by microsco, EC50 = 0.89 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-, Influenza A virus (A/turkey/Minnesota/833/1980(H4N2)) harboring neuraminidase E119G mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days , EC50 = 0.89 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-, Influenza A virus (A/turkey/Minnesota/833/1980(H4N2)) infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic analysis using crystal violet , EC50 = 0.96 μM. | 20350949 | ||
| MDCK | Antiviral assay | Antiviral activity against, adamantane-resistant Influenza A virus (A/Bethesda/956/2006(H3N2)) harboring neuraminidase R292K mutant and M2 S31N mutant infected in MDCK cells assessed as inhibition of viral replication af, EC50 = 1.21 μM. | 20350949 | |||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-resistant ,Influenza A virus (A/pdm/Mexico/4604/2009 (H1N1)) harboring M2 V28I, S31N mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by microsco, EC50 = 1.21 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-resistant,Influenza B virus B/Memphis/20/1996 infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic analysis using crystal violet stain, EC50 = 1.21 μM. | 20350949 | ||
| MDCK | Antiviral assay | Antiviral activity against adamantane,Influenza A virus (A/chicken/Vietnam/NCVD103/2007 clade 2.3.4 (H5N1)) harboring neuraminidase I222T mutant infected in MDCK cells assessed as inhibition of viral replication after, EC50 = 1.27 μM. | 20350949 | |||
| MDCK | Antiviral assay | Inhibition of viral replication of influenza A virus (A/Hong Kong/213/03(H5N1)) and influenza A virus (A/Ann Arbor/6/60(H2N2)) hybrid virus in MDCK cells by virus yield reduction assay, EC90 = 1.3 μM. | 17194832 | |||
| MDCK | Antiviral assay | 1 hr | Antiviral activity against Influenza B virus (B/Brisbane/60/2008) infected in MDCK cells assessed as inhibition of virus-induced cytopathogenicity at 1 hr post-infection measured after 3 days by fluorescein-diacetate based fluorimetric analysis, EC50 = 1.32 μM. | 23419738 | ||
| MDCK | Antiviral assay | Antiviral activity against adamantane-, Influenza A virus (A/Vietnam/HN30408/2005 N294S clade 1 (H5N1)) harboring M2 L26I, S31N mutant and neuraminidase N294S mutant infected in MDCK cells assessed as inhibition of viral , EC50 = 1.341 μM. | 20350949 | |||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-resistant,Influenza A virus (A/pdm/California/07/2009 (H1N1)) harboring M2 V28I, S31N mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by micros, EC50 = 1.4 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-resistant,Influenza B virus (B/Rochester/01/2001) infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic analysis using crystal violet stain, EC50 = 1.4 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-, Influenza A virus (A/Georgia/17/2006(H1N1)) infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic analysis using crystal violet stain, EC50 = 1.46 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-,Influenza A virus (A/turkey/VA/4529/2002 (H7N2)) infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic analysis using crystal violet stain, EC50 = 1.53 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-,Influenza A virus (A/Florida/21/2008(H1N1)) harboring neuraminidase H274Y mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by microsc, EC50 = 1.59 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-,Influenza A virus (A/duck/Vietnam/NCVD93/2007 clade 2.3.4 (H5N1)) infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic analysis using cry, EC50 = 1.59 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against, adamantane-resistant Influenza B virus (B/Rochester/01/2001) harboring neuraminidase D198N mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic, EC50 = 1.72 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-, Influenza A virus (A/Brazil/1067/2008(H1N1)) infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic analysis using crystal violet stain, EC50 = 1.85 μM. | 20350949 | ||
| MDCK | Antiviral assay | 1 hr | Antiviral activity against Influenza B virus (B/Lee/40) infected in MDCK cells assessed as inhibition of virus-induced cytopathogenicity at 1 hr post-infection measured after 3 days by fluorescein-diacetate based fluorimetric analysis, EC50 = 1.93 μM. | 23419738 | ||
| MDCK | Antiviral assay | 1 hr | Antiviral activity against Influenza B virus (B/Panama/45/1990) infected in MDCK cells assessed as inhibition of virus-induced cytopathogenicity at 1 hr post-infection measured after 3 days by fluorescein-diacetate based fluorimetric analysis, EC50 = 1.94 μM. | 23419738 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-resistant,Influenza A virus (A/pdm/California/04/2009 (H1N1)) harboring M2 V28I, S31N mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by micros, EC50 = 1.97 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-,Influenza A virus (A/swine/Michigan/09/2007 (H1N2)) harboring M2 V27I, V28D mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic a, EC50 = 2.23 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-,Influenza A virus (A/Ecuador/5179/2008(H1N1)) infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic analysis using crystal violet stain, EC50 = 2.48 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-,Influenza A virus (A/Georgia/20/2006(H1N1)) harboring neuraminidase H274Y mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by microsc, EC50 = 2.55 μM. | 20350949 | ||
| MDCK | Antiviral assay | Inhibition of viral replication of influenza A virus (A/Hong Kong/213/03(H5N1)) and influenza A virus (A/Ann Arbor/6/60(H2N2)) hybrid virus in MDCK cells by neutral red uptake assay, EC50 = 2.6 μM. | 17194832 | |||
| MDCK | Antiviral assay | Inhibition of influenza A virus (A/duck/Minnesota/1525/1981 (H5N1)) replication in MDCK cells by virus yield reduction assay, EC90 = 2.6 μM. | 17194832 | |||
| MDCK | Antiviral assay | 24 hrs | Antiviral activity against Influenza A virus A/WSN/33(H1N1) infected in MDCK cells assessed as inhibition of neuraminidase activity using 2'-(4-methylumbelliferyl)-alpha-D-N-acetylneuraminic acid substrate pretreated with cells for 24 hrs followed by vira, EC50 = 2.7 μM. | 27120583 | ||
| MDCK | Antiviral assay | 1 hr | Antiviral activity against Influenza B virus (B/Taiwan/2/62) infected in MDCK cells assessed as inhibition of virus-induced cytopathogenicity at 1 hr post-infection measured after 3 days by fluorescein-diacetate based fluorimetric analysis, EC50 = 2.84 μM. | 23419738 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-resistant,Influenza A virus (A/Wisconsin/16/2008(H1N1)) harboring neuraminidase H274Y mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by micros, EC50 = 2.87 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-, Influenza A virus (A/Idaho/01/2009(H1N1)) harboring neuraminidase H274Y mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic analysi, EC50 = 2.93 μM. | 20350949 | ||
| MDCK | Antiviral assay | Antiviral activity against, adamantane-resistant Influenza A virus (A/Luhansk/18/2008(H1N1)) harboring neuraminidase H274Y mutant and M2 G34E mutant infected in MDCK cells assessed as inhibition of viral replication afte, EC50 = 2.93 μM. | 20350949 | |||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-,Influenza B virus (B/Illinois/03/2008) harboring neuraminidase E119A mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic analysis u, EC50 = 3 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-,Influenza A virus (A/Washington/10/2008(H1N1)) harboring M2 S31N mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic anal, EC50 = 3.25 μM. | 20350949 | ||
| MDCK | Antiviral assay | Antiviral activity against adamantane-susceptible , Influenza A virus (A/duck/Vietnam/NCVD94/2007 clade 2.3.4 (H5N1)) harboring neuraminidase I117V mutant infected in MDCK cells assessed as inhibition of viral replication a, EC50 = 3.38 μM. | 20350949 | |||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-,Influenza A virus (A/California/27/2007(H1N1)) infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic analysis using crystal violet stain, EC50 = 3.63 μM. | 20350949 | ||
| MDCK | Antiviral assay | Inhibition of viral replication of influenza A virus (A/Vietnam/1203/2004 (H5N1)) and influenza A virus (A/Ann Arbor/6/60(H2N2)) hybrid virus in MDCK cells by virus yield reduction assay, EC90 = 3.8 μM. | 17194832 | |||
| MDCK | Antiviral assay | Antiviral activity against, adamantane-resistant Influenza A virus (A/Texas/12/2007 (clone)(H3N2)) harboring neuraminidase E119I mutant and harboring M2 S31N mutant infected in MDCK cells assessed as inhibition of viral , EC50 = 3.95 μM. | 20350949 | |||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-resistant, Influenza B virus (B/Illinois/47/2005) infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic analysis using crystal violet stain, EC50 = 4.01 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-resistant,Influenza A virus (A/New Hampshire/01/2009(H3N2)) harboring M2 S31N mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic an, EC50 = 4.08 μM. | 20350949 | ||
| MDCK | Antiviral assay | Antiviral activity against, adamantane-resistant Influenza A virus (A/Vietnam/HN30408/2005 H274Y clade 1 (H5N1)) harboring M2 L26I, S31N mutant and neuraminidase H274Y mutant infected in MDCK cells assessed as inhibition, EC50 = 4.14 μM. | 20350949 | |||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-resistant,Influenza A virus (A/Massachusetts/03/2009(H3N2)) harboring M2 S31N mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic an, EC50 = 4.2 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-resistant,Influenza A virus (A/Washington/01/2007(H3N2)) harboring M2 S31N mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic analy, EC50 = 4.46 μM. | 20350949 | ||
| MDCK | Antiviral assay | Inhibition of influenza A virus (A/duck/Minnesota/1525/1981 (H5N1)) replication in MDCK cells by neutral red uptake assay, EC50 = 4.5 μM. | 17194832 | |||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-,Influenza A virus (A/Swine/Texas/14/2008 (H1N1)) harboring M2 V27T, V28D mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic anal, EC50 = 4.52 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-susceptible,Influenza A virus (A/Santiago/5248/2008(H1N1)) harboring neuraminidase D198E mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by micr, EC50 = 4.7 μM. | 20350949 | ||
| MDCK | Antiviral assay | 1 hr | Antiviral activity against Influenza A virus (A/Hong kong/8/1968(H2N3)) infected in MDCK cells assessed as inhibition of virus-induced cytopathogenicity at 1 hr post-infection measured after 3 days by fluorescein-diacetate based fluorimetric analysis, EC50 = 4.71 μM. | 23419738 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-,Influenza A virus (A/New Jersey/15/2007(H1N1)) harboring neuraminidase H274Y mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by micr, EC50 = 4.9 μM. | 20350949 | ||
| Vero | Antiviral assay | 7 to 8 days | Antiviral activity against Junin virus Candid-1 in Vero cells assessed as inhibition of virus-induced visual cytopathic effect after 7 to 8 days, EC50 = 5 μM. | 17606691 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-,Influenza A virus (A/North Carolina/02/ 2009(H1N1)) harboring M2 S31N mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic, EC50 = 5.03 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against, adamantane-resistant Influenza B virus (B/Michigan/20/2005) harboring neuraminidase H274Y mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic , EC50 = 5.03 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-resistant,Influenza A virus (A/Vietnam/1203/2004 clade 1 (H5N1)) harboring M2 L26I, S31N mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by mic, EC50 = 5.22 μM. | 20350949 | ||
| MDCK | Antiviral assay | Antiviral activity agains, adamantane-resistant Influenza A virus (A/Texas/12/2007 (clone)(H3N2)) harboring neuraminidase E119V mutant and harboring M2 S31N mutant infected in MDCK cells assessed as inhibition of viral , EC50 = 5.22 μM. | 20350949 | |||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-resistant Influenza B virus (B/New York/22/2008) infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic analysis using crystal violet stain, EC50 = 5.3 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against, adamantane-resistant Influenza A virus (A/Wuhan/395/1995-like (H3N2)) harboring neuraminidase E119V mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by mi, EC50 = 5.41 μM. | 20350949 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-resistant, Influenza A virus (A/Wuhan/395/1995-like (H3N2)) infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic analysis using crystal viole, EC50 = 5.99 μM. | 20350949 | ||
| Vero | Antiviral assay | 7 to 8 days | Antiviral activity against Pichinde virus An 4763 in Vero cells assessed as inhibition of virus-induced visual cytopathic effect after 7 to 8 days, EC50 = 6 μM. | 17606691 | ||
| Vero | Antiviral assay | 7 to 8 days | Antiviral activity against Tacaribe virus TRVL11573 in Vero cells assessed as inhibition of virus-induced visual cytopathic effect after 7 to 8 days, EC50 = 6 μM. | 17606691 | ||
| MDCK | Antiviral assay | Antiviral activity against, adamantane-resistant Influenza A virus (A/pdm/Washington/29/2009 (H1N1)) harboring M2 V28I, S31N mutant and neuraminidase H274Y mutant infected in MDCK cells assessed as inhibition of viral re, EC50 = 6.62 μM. | 20350949 | |||
| MDCK | Antiviral assay | 1 hr | 3 days | Antiviral activity against Influenza A virus (A/Taiwan/1/1986(H1N1)) infected in MDCK cells assessed as inhibition of virus-induced cytopathogenicity at 1 hr post-infection measured after 3 days by fluorescein-diacetate based fluorimetric analysis, EC50 = 6.95 μM. | 23419738 | |
| MDCK | Antiviral assay | Inhibition of Influenza A virus (A/gull/Pennsylvania/4175/83 (H5N1)) replication in MDCK cells by virus yield reduction assay, EC90 = 7.7 μM. | 17194832 | |||
| MDCK | Antiviral assay | 3 days | Antiviral activity against Influenza virus B/Ned/537/05 infected in MDCK cells assessed as reduction in viral replication at 3 days post infection by formazan-based MTS assay, EC50 = 8.3 μM. | 29906392 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against Influenza virus B/Ned/537/05 infected in MDCK cells assessed as reduction in viral replication at 3 days post infection by microscopic analysis, EC50 = 9 μM. | 29906392 | ||
| MDCK | Antiviral assay | 3 days | Antiviral activity against adamantane-resistant,Influenza A virus (A/New York/107/2003(H7N2)) harboring M2 V28A, S31N mutant infected in MDCK cells assessed as inhibition of viral replication after 3 days by microscopic , EC50 = 10.2 μM. | 20350949 | ||
| MDCK | Antiviral assay | Inhibition of Influenza A virus (A/gull/Pennsylvania/4175/83 (H5N1)) replication in MDCK cells by neutral red uptake assay, EC50 = 11.5 μM. | 17194832 | |||
| MDCK | Antiviral assay | Inhibition of viral replication of influenza A virus (A/Vietnam/1203/2004 (H5N1)) and influenza A virus (A/Ann Arbor/6/60(H2N2)) hybrid virus in MDCK cells by neutral red uptake assay, EC50 = 12.1 μM. | 17194832 | |||
| Vero-A | Antiviral assay | Antiviral activity against Chikungunya virus venturini isolate (Italy 2008) infected in African green monkey Vero-A cells assessed as inhibition of viral-induced cytopathic effect measured on day 7 post infection by MTS/PMS assay, EC50 = 16 μM. | 28689975 | |||
| MDCK | Antiviral assay | Antiviral activity against oseltamivir-susceptible, adamantane-resistant Influenza A virus (A/pdm/Illinois/10/2009(H1N1)) harboring M2 V28I, S31N mutant and neuraminidase H274Y mutant infected in MDCK cells assessed as inhibition of viral repli, EC50 = 22.48 μM. | 20350949 | |||
| MDCK | Antiviral assay | 3 days | Antiviral activity against Influenza virus A/X-31 infected in MDCK cells assessed as reduction in viral replication at 3 days post infection by microscopic analysis, EC50 = 24 μM. | 29906392 | ||
| Vero-A | Antiviral assay | Antiviral activity against Chikungunya virus 899 infected in African green monkey Vero-A cells assessed as inhibition of viral-induced cytopathic effect measured on day 7 post infection by MTS/PMS assay, EC50 = 25 μM. | 28689975 | |||
| Vero-A | Antiviral assay | Antiviral activity against Chikungunya virus LR2006-OPY1 infected in African green monkey Vero-A cells assessed as inhibition of viral-induced cytopathic effect measured on day 7 post infection by MTS/PMS assay, EC50 = 25 μM. | 28689975 | |||
| MDCK | Antiviral assay | 3 days | Antiviral activity against Influenza virus A/X-31 infected in MDCK cells assessed as reduction in viral replication at 3 days post infection by formazan-based MTS assay, EC50 = 26 μM. | 29906392 | ||
| MDCK | Antiviral assay | Antiviral activity against 0.001 MOI adamantane-resistant,oseltamivir-susceptible Influenza A virus (A/pdm/Mexico/4604/2009(H1N1)) harboring M2 V28I, S31N mutant infected in MDCK cells assessed as determination of viral titer in cell culture at, NULL = NULL μM. | 20350949 | |||
| MDCK | Antiviral assay | Antiviral activity against 0.001 MOI adamantane-resistant,oseltamivir-susceptible Influenza A virus (A/pdm/California/04/2009(H1N1)) harboring M2 V28I, S31N mutant infected in MDCK cells assessed as determination of viral titer in cell culture , NULL = NULL μM. | 20350949 | |||
| MDCK | Antiviral assay | 128 uM | Antiviral activity against adamantane-resistant,oseltamivir-susceptible Influenza A virus (A/pdm/Mexico/4604/2009(H1N1)) harboring M2 V28I, S31N mutant infected in MDCK cells assessed as determination of viral titer in cell culture at 128 uM af, NULL = NULL μM. | 20350949 | ||
| MDCK | Antiviral assay | 128 uM | Antiviral activity against adamantane-resistant,oseltamivir-susceptible Influenza A virus (A/pdm/California/04/2009(H1N1)) harboring M2 V28I, S31N mutant infected in MDCK cells assessed as determination of viral titer in cell culture at 128 uM , NULL = NULL μM. | 20350949 | ||
| Vero | Antiviral assay | 6400 uM | up to 12 hrs | Antiviral activity against Yellow fever virus 17D infected in African green monkey Vero cells assessed as time required for inhibition of viral replication at 6400 uM up to 12 hrs post-viral infection, NULL = NULL μM. | 18955536 | |
| MDCK | Antiviral assay | 0 to 2 hrs | Inhibition of influenza A virus infected in MDCK cells assessed as inhibition of viral load treated with compound at 0 to 2 hrs post-infection by RT-qPCR method based time of addition assay, NULL = NULL μM. | 30192544 | ||
| MDCK | Antiviral assay | 5 to 8 hrs | Inhibition of influenza A virus infected in MDCK cells assessed as inhibition of viral load treated with compound at 5 to 8 hrs post-infection by RT-qPCR method based time of addition assay, NULL = NULL μM. | 30192544 | ||
| MDCK | Antiviral assay | 0 to 10 hrs | Inhibition of influenza A virus infected in MDCK cells assessed as inhibition of viral load treated with compound at 0 to 10 hrs post-infection by RT-qPCR method based time of addition assay, NULL = NULL μM. | 30192544 | ||
| MDCK | Antiviral assay | 2 to 5 hrs | Inhibition of influenza A virus infected in MDCK cells assessed as inhibition of viral load treated with compound at 2 to 5 hrs post-infection by RT-qPCR method based time of addition assay, NULL = NULL μM. | 30192544 | ||
| MDCK | Antiviral assay | 10 uM | up to 8 hrs | Antiviral activity against Influenza A virus (A/chicken/Hubei/327/2004(H5N1)) infected in MDCK cells assessed as inhibition of early stages of viral replication at 10 uM up to 8 hrs by crystal violet staining based plaque reduction assay dependent time-of, NULL = NULL μM. | 30292897 | |
| MDCK | Antiviral assay | 100 uM | 12 hrs | Antiviral activity against Influenza virus A/X-31 infected in MDCK cells assessed as inhibition of one cycle viral RNA synthesis at 100 uM preincubated with cells for 12 hrs followed by viral infection measured after 10 hrs post infection by RT-qPCR metho, NULL = NULL μM. | 29906392 | |
| MDCK | Antiviral assay | 250 uM | 10 hrs | Antiviral activity against Influenza virus A/X-31 infected in MDCK cells assessed as inhibition of one cycle viral RNA sytheis at 250 uM added together with virus and measured at 10 hrs post infection by RT-qPCR method, NULL = NULL μM. | 29906392 | |
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 31 mg/mL
(197.32 mM)
Water : 2 mg/mL Ethanol : 2 mg/mL |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 157.1 | Formula | C5H4FN3O2 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 259793-96-9 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | C1=C(N=C(C(=O)N1)C(=O)N)F | ||
Mechanism of Action
| Targets/IC50/Ki |
RNA-dependent RNA polymerase
|
|---|---|
| In vitro |
Favipiravir (T-705) shows anti-influenza virus activities with IC50 ranged from 0.013 to 0.48 μg/ml for the influenza A viruses, from 0.039 to 0.089 μg/ml for the influenza B viruses, and from 0.030 to 0.057 μg/ml for the influenza C viruses. In mammalian cell lines (MDCK cells, Vero cells, HEL cells, A549 cells, HeLa cells, and HEp-2 cells), it shows no cytotoxicity at concentrations up to 1,000 μg/ml. In MDCK cells inoculated with seasonal influenza A (H1N1) viruses, this compound induces lethal mutagenesis.
|
| In vivo |
Favipiravir (T-705) protects influenza virus-infected mice from death when administered at 200 mg/kg/day (p.o.). In mice experimentally infected with Ebola virus, this compound efficiently blocks viral production, reaching an antiviral effectiveness of 95% and 99.6% at 2 and 6 days after initiation of treatment, respectively.
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Growth inhibition assay | Survival Cell viability |
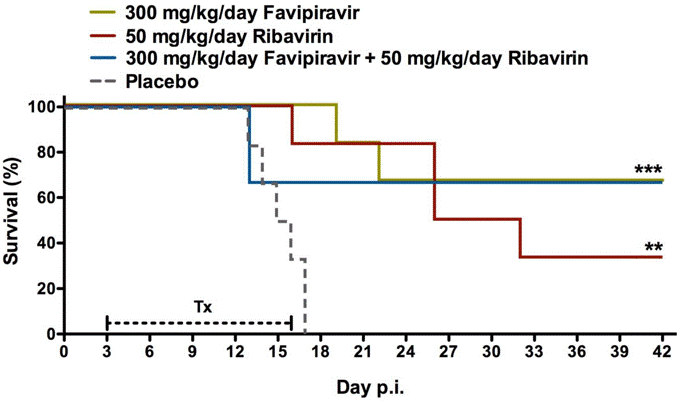
|
26711718 |
| IHC | H&E Staining NiV nucleoprotein LASV antigen H&E stain |
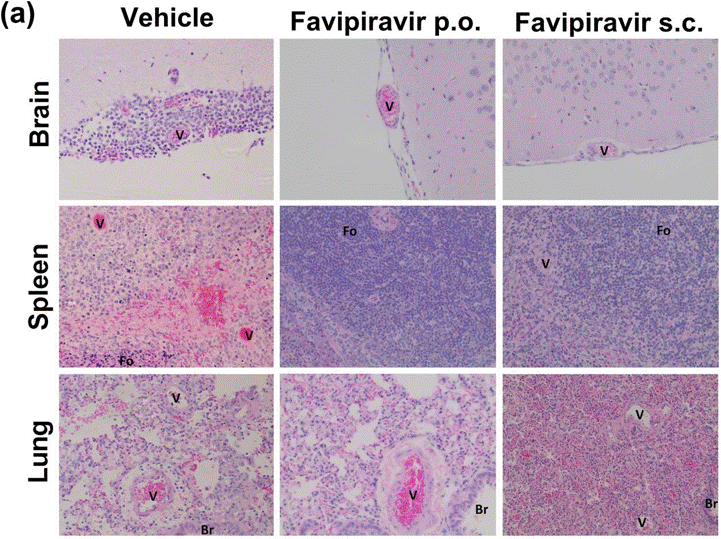
|
29765101 |
| Immunofluorescence | rNiV-Gluc |
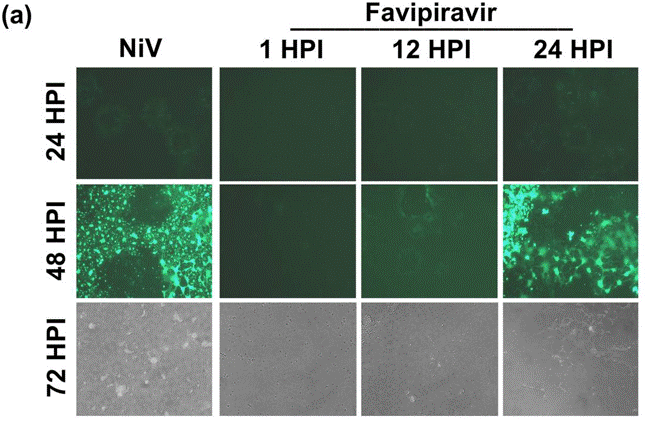
|
29765101 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06024421 | Recruiting | Infectious Disease|Pharmacology |
Institut National de la Santé Et de la Recherche Médicale France|FUJIFILM Toyama Chemical Co. Ltd. |
April 2024 | Phase 1 |
| NCT05940545 | Recruiting | CCHF |
Liverpool School of Tropical Medicine |
July 12 2023 | Phase 1|Phase 2 |
| NCT04907682 | Completed | Lassa Fever |
Bernhard Nocht Institute for Tropical Medicine|University of Hamburg-Eppendorf|Alliance for International Medical Action|Institut National de la Santé Et de la Recherche Médicale France|University of Bordeaux|Federal Medical Centre Owo|Irrua Specialist Teaching Hospital |
July 30 2021 | Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































