research use only
Coumarin Immunology & Inflammation related chemical
Cat.No.S4170
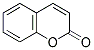
Chemical Structure
Molecular Weight: 146.14
Quality Control
| Related Targets | PD-1/PD-L1 CXCR STING AhR CD markers Interleukins Anti-infection Antioxidant COX Histamine Receptor |
|---|---|
| Other Immunology & Inflammation related Inhibitors | Cl-amidine Bestatin (Ubenimex) Bindarit (AF 2838) Tempol Tranilast Sinomenine GI254023X (GI4023) Geniposidic acid CORM-3 Germacrone |
Solubility
|
In vitro |
DMSO
: 29 mg/mL
(198.43 mM)
Ethanol : 29 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 146.14 | Formula | C9H6O2 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 91-64-5 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | C1=CC=C2C(=C1)C=CC(=O)O2 | ||
Mechanism of Action
| In vitro |
Coumarin is a fragrant organic chemical compound in the benzopyrone chemical class, which is a colorless crystalline substance in its standard state. It is a naturally occurring constituent of many plants and essential oils, including tonka beans, sweet clover, woodruff, oil of cassia, and lavender. This compound is a member of a class of compounds called benzopyrones. These compounds have been used to treat such diverse ailments as cancer, bums, brucellosis, and rheumatic disease, and they have been used as antispasmodics. Although this chemical itself has no anticoagulant properties, it is transformed into the natural anticoagulant dicoumarol by a number of species of fungi. This occurs as the result of the production of 4-hydroxycoumarin, then further into the actual anticoagulant dicoumarol, a fermentation product and mycotoxin. This substance is responsible for the bleeding disease known historically as "sweet clover disease" in cattle eating moldy sweet clover silage.
|
References |
|---|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT04952792 | Completed | Atrial Fibrillation|Hemodialysis Complication |
Hospital Universitari de Bellvitge|Spanish Society of Nephrology|Baxter Healthcare Corporation |
May 20 2021 | Phase 2 |
| NCT01119300 | Completed | Venous Thromboembolism|Atrial Fibrillation |
Utrecht Institute for Pharmaceutical Sciences|Utrecht University|Leiden University Medical Center|Erasmus Medical Center|University of Ulm|Newcastle University|University of Liverpool|LGC Limited|Uppsala University|Democritus University of Thrace|Elisabethinen Hospital |
January 2011 | Phase 4 |
| NCT00157118 | Completed | Protein C Deficiency |
Baxalta now part of Shire|Takeda |
August 22 2003 | Phase 2|Phase 3 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































