research use only
Galunisertib (LY2157299) TGF-beta/Smad inhibitor
Cat.No.S2230
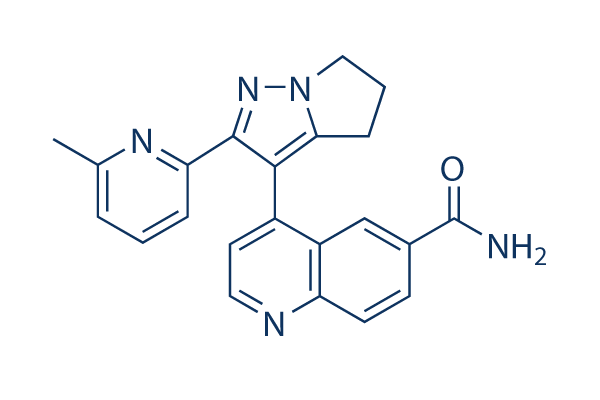
Chemical Structure
Molecular Weight: 369.42
Quality Control
| Related Targets | PKC ROCK Bcr-Abl |
|---|---|
| Other TGF-beta/Smad Inhibitors | SB431542 RepSox (E-616452) Vactosertib (TEW-7197) LDN-193189 A-83-01 LDN-193189 Dihydrochloride SRI-011381 (C381) LY2109761 SIS3 Hydrochloride SB-525334 |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| Panc-1 | Function Assay | 10 μM | 48 h | DMSO | stimulates the cell invasion into the collagen gel and the Matrigel-coated collagen gel | 24780821 |
| Sf9 | Function assay | Inhibition of human recombinant GST-fused ALK5 expressed in Sf9 insect cells using casein as substrate by proprietary radioisotopic protein kinase assay, IC50 = 0.0694 μM. | 26483198 | |||
| Sf9 | Function assay | Inhibition of human ALK5 expressed in Sf9 insect cells using casein as substrate in presence of [gamma-33P]ATP, IC50 = 0.051 μM. | 28135685 | |||
| Sf9 | Function assay | 1 hr | Inhibition of human His-tagged TGFbetaR1 T204D mutant expressed in Sf9 insect cells after 1 hr by HTRF assay, IC50 = 0.0041 μM. | 29422332 | ||
| B16 | Antitumor assay | 75 mg/kg | Antitumor activity against mouse B16 cells in mouse assessed as inhibition of tumor growth at 75 mg/kg, po bid relative to vehicle-treated control | 24786585 | ||
| B16 | Antitumor assay | 75 mg/kg | Antitumor activity against mouse B16 cells in mouse assessed as suppression of tumor volume at 75 mg/kg, po bid relative to vehicle-treated control | 24786585 | ||
| B16 | Antitumor assay | 75 mg/kg | Antitumor activity against mouse B16 cells in mouse assessed as inhibition of lymph node metastasis at 75 mg/kg, po bid relative to vehicle-treated control | 24786585 | ||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 74 mg/mL
(200.31 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 369.42 | Formula | C22H19N5O |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 700874-72-2 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | CC1=NC(=CC=C1)C2=NN3CCCC3=C2C4=C5C=C(C=CC5=NC=C4)C(=O)N | ||
Mechanism of Action
| Targets/IC50/Ki |
TβRI
(Cell-free assay) 56 nM
|
|---|---|
| In vitro |
Galunisertib (LY2157299) potently inhibits the TGFβ receptor signaling. It abolishes the TGFβ induced Smad2 phosphorylation in HUVEC cells, and also shows dose dependent potentiation of VEGF or bFGF induced cell proliferation in HUVEC. This compound promotes VEGF induced HUVEC cell migration and potentiates angiogenesis in the in vitro VEGF-stimulated cord formation assay.It inhibits TGF-β-mediated SMAD2 activation and hematopoietic suppression in primary hematopoietic stem cells in a dose-dependent manner. LY2157199 treatment stimulates hematopoiesis from primary MDS bone marrow specimens.In human glioblastoma (GBM) cells, it blocks signaling through the heteromeric TGFβ receptor complex to reduce levels of active, phosphorylated SMAD. |
| Kinase Assay |
TGF-β Type I (RIT204D) Receptors reaction
|
|
Reactions: 170-200 nM enzyme in 1 × KB (50 mM Tris pH 7.5, 150 mM NaCl, 4 mM MgCl2, 1 mM NaF, 2 mM β-mercaptoethanol), with a dilution series of Galunisertib (LY2157299) in 1 × KB /16% DMSO (20 μM to 1 nM final concentration with 4% DMSO final concentration). Reactions are started by adding ATP mix (4 μM ATP/ 1 μCi 33P-α-ATP final concentrations) in 1 × KB. Reactions are incubated at 30 °C for 1 hour. Reactions are stopped and quantitated using standard TCA/BSA precipitation onto Millipore FB glass fiber filter plates and by liquid scintillation counting on a MicroBeta JET.
|
|
| In vivo |
Although anti-tumor activity has been observed in several pre-clinical models, Galunisertib (LY2157299) fails to show significant in vivo angiogenic effects in the 4T1, Colo205, or A549 xenograft models. Administration of this compound ameliorates anemia in a TGF-β overexpressing transgenic mouse model of bone marrow failure. Oral administration of it at 75 mg/kg/day displays significant antitumor activity against both Calu6 and MX1 xenografts in mice. In vivo, it induces angiogenesis and enhances VEGF and basic-fibroblast-growth-factor-induced angiogenesis in a Matrigel-plug assay, whereas adding an alpha5-integrin-neutralizing antibody to the Matrigel selectively inhibits this enhanced response. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | Periostin / Fibronectin / Snail / Twist / pSmad2 / pERK / pAkt / pFAK TGF-βR1 / p-ERK |
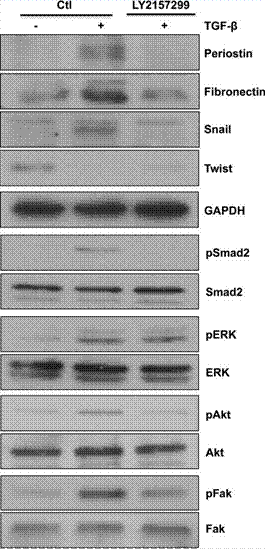
|
29774119 |
| Growth inhibition assay | Cell viability |
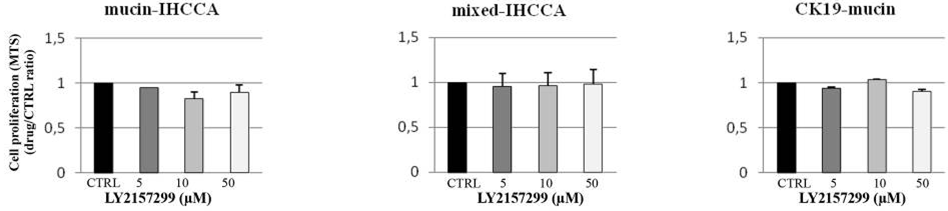
|
28873435 |
| Immunofluorescence | E-cadherin |
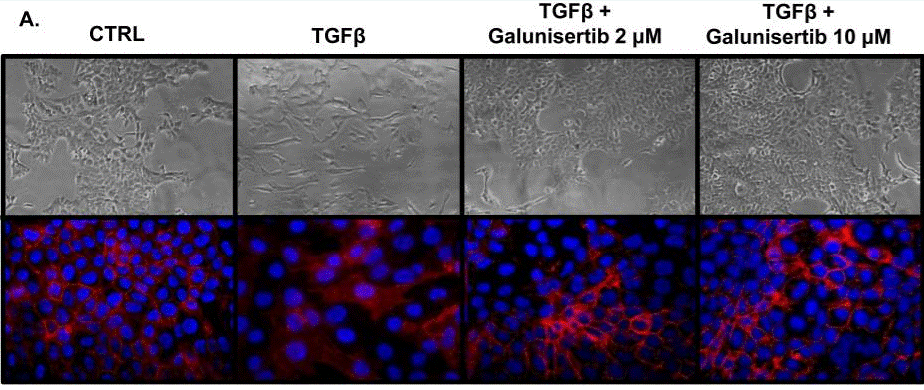
|
29467918 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT04031872 | Unknown status | Colorectal Cancer Metastatic |
The Netherlands Cancer Institute|Vall d''Hebron Institute of Oncology|Agendia|European Organisation for Research and Treatment of Cancer - EORTC|Azienda Ospedaliera Niguarda Cà Granda|Fundación para la Investigación del Hospital Clínico de Valencia|University of Campania Luigi Vanvitelli|University of Turin Italy|Eli Lilly and Company|Catalan Institute of Health|Universitaire Ziekenhuizen KU Leuven |
February 2020 | Phase 1|Phase 2 |
| NCT03470350 | Withdrawn | Colorectal Cancer Metastatic |
The Netherlands Cancer Institute|Vall d''Hebron Institute of Oncology|Agendia|European Organisation for Research and Treatment of Cancer - EORTC|Azienda Ospedaliera Niguarda Cà Granda|Fundación para la Investigación del Hospital Clínico de Valencia|University of Campania Luigi Vanvitelli|University of Turin Italy|Eli Lilly and Company|Catalan Institute of Health|Universitaire Ziekenhuizen KU Leuven |
August 24 2018 | Phase 1|Phase 2 |
| NCT02734160 | Completed | Metastatic Pancreatic Cancer |
Eli Lilly and Company|AstraZeneca |
June 15 2016 | Phase 1 |
| NCT02452008 | Recruiting | Prostate Cancer |
Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins|Eli Lilly and Company |
May 3 2016 | Phase 2 |
| NCT02752919 | Completed | Healthy |
Eli Lilly and Company |
April 2016 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































