research use only
SB-525334 ALK5 inhibitor
Cat.No.S1476
Chemical Structure
Molecular Weight: 343.42
Quality Control
| Related Targets | PKC ROCK Bcr-Abl |
|---|---|
| Other TGF-beta/Smad Inhibitors | SB431542 RepSox (E-616452) Vactosertib (TEW-7197) LDN-193189 A-83-01 LDN-193189 Dihydrochloride Galunisertib (LY2157299) SRI-011381 (C381) LY2109761 SIS3 Hydrochloride |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| MiaPaCa2 | Cell Viability Assay | 0-10 μM | 72 h | efficiently reduces the viability with | 22399597 | |
| HUVECs | Function Assay | 10 μM | 72 h | reduces the phosphorylation of smad2 | 23968981 | |
| HUVECs | Function Assay | 10 μM | 72 h | blocks signaling | 23968981 | |
| HUVECs | Function Assay | 10 μM | 24/48/72 h | leads to a disruption of the HUVEC monolayer after 72 hr | 23968981 | |
| rhPOSTN | Apoptosis Assay | 0.5 μM | 48 h | significantly decreases the number of apoptotic cells | 24129188 | |
| AsPC1 | Cell Viability Assay | 0-10 μM | 72 h | efficiently reduces the viability with | 22399597 | |
| PASMCs | Function Assay | 1 μM | 15 min | inhibits TGF-β1-mediated proliferation of familial iPAH PASMCs at an IC50 of 295 nmol/L | 19116361 | |
| ELT-3 | Function Assay | 0.5/1.0/2.0 μM | 1 h | DMSO | inhibits TGF-β signaling in leiomyomas | 17505012 |
| Sf9 | Function assay | Inhibition of human recombinant GST-fused ALK5 expressed in Sf9 cells, IC50 = 0.162 μM. | 20472445 | |||
| TC32 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for TC32 cells | 29435139 | |||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | |||
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Saos-2 cells | 29435139 | |||
| BT-37 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-37 cells | 29435139 | |||
| BT-12 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-12 cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | |||
| OHS-50 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for OHS-50 cells | 29435139 | |||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh41 cells | 29435139 | |||
| SJ-GBM2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SJ-GBM2 cells | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 69 mg/mL
(200.92 mM)
Ethanol : 69 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 343.42 | Formula | C21H21N5 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 356559-20-1 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | CC1=NC(=CC=C1)C2=C(N=C(N2)C(C)(C)C)C3=CC4=NC=CN=C4C=C3 | ||
Mechanism of Action
| Targets/IC50/Ki |
TGFβR1(ALK5)
(Cell-free assay) 14.3 nM
|
|---|---|
| In vitro |
SB 525334 shows no inhibition in the enzymes ALK2, 3, and 6, with IC50 values > 10 μM. SB 525334 blocks phosphorylation induced by TGF-β1 and nuclear translocation of Smad2/3 in renal proximal tubule cells. SB 525334 also inhibits the increased mRNA expression levels of plasminogen activator inhibitor-1 (PAI-1) and procollagen α1(I) induced by TGF-β1 in A498 renal epithelial carcinoma cells at 1 μM). SB 525334 (1 μM) attenuates the heightened sensitivity to TGF-β1 exhibited by pulmonary artery smooth muscle cells (PASMCs) from patients with familial forms of idiopathic pulmonary arterial hypertension (PAH). |
| Kinase Assay |
Kinase assay to determine the potency and selectivity of SB 525334
|
|
In order to determine the potency of SB 525334, purified GST-tagged kinase domain of ALK5 is incubated with purified GST-tagged full-length Smad3 in the presence of 33P-γATP and different concentrations of SB 525334. The readout is radioactively labeled Smad3.To determine the selectivity of SB 525334, purified GST-tagged kinase domain of ALK2 and ALK4 are incubated with GST-tagged full-length Smad1 and Smad3, respectively, in the presence of different concentrations of SB 525334. IC50 values are calculated.
|
|
| In vivo |
SB 525334 (10 mg/kg/day) decreases the renal mRNA levels of PAI-1, procollagen α1(I), and procollagen α1(III) in a nephritis-induced renal fibrosis rat model. Furthermore, PAN-induced proteinuria is significantly inhibited by SB 525334 (10 mg/kg/day). SB 525334 may also be efficacious in mesenchymal tumors. SB 525334 (10 mg/kg/day) significantly decreases uterine mesenchymal tumor incidence, multiplicity, and size in Eker rats. SB 525334 significantly reverses pulmonary arterial pressure and inhibits right ventricular hypertrophy in a rat model of PAH. This is revealed by a significant reduction in pulmonary arteriole muscularization induced by monocrotaline (used to induce PAH) after treatment with SB 525334 (3 or 30 mg/kg). In a induced pulmonary fibrosis mice model, SB 525334 (10 mg/kg or 30 mg/kg) attenuates the histopathological alterations in the lung, and significantly decreased mRNA expression of Type I and III procollagen and fibronectin. SB 525334 also attenuates Smad2/3 nuclear translocation, myofibroblast proliferation, deposition of Type I collagen, and decreases CTGF-expressing cells. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | Fibronectin / E-cadherin / Hif-1α |
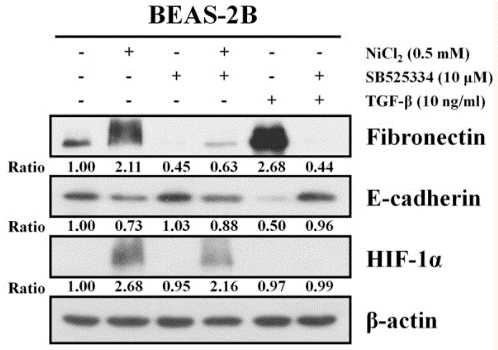
|
29127306 |
| Immunofluorescence | E-cadherin / Vimentin |
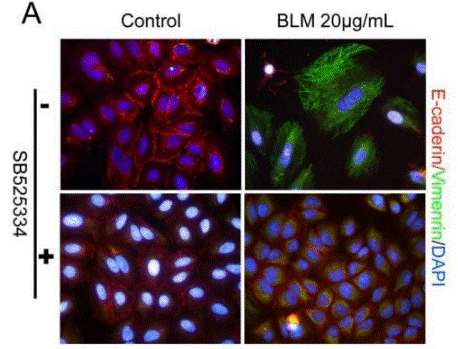
|
27471572 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
Frequently Asked Questions
Question 1:
I want to know the feasibility of using it via oral gavage in rodents (dosing, frequency, formulation…)?
Answer:
It can be used for oral gavage in rodents, and the vehicle we suggest is: 30% Propylene glycol, 5% Tween 80, 65% D5W (dextrose(5%)in water) at 20mg/ml maximum. Based on the following reference they administrated this compound at an estimated dose of 10 mg/kg/day in rat for 2 months: http://clincancerres.aacrjournals.org/content/13/10/3087.long






































