research use only
Veliparib (ABT-888) PARP inhibitor
Cat.No.S1004
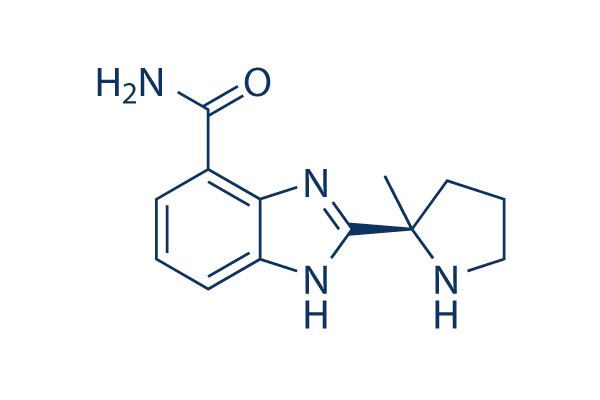
Chemical Structure
Molecular Weight: 244.29
Quality Control
| Related Targets | HDAC ATM/ATR DNA-PK WRN DNA/RNA Synthesis Topoisomerase PPAR Sirtuin Casein Kinase eIF |
|---|---|
| Other PARP Inhibitors | XAV-939 AZD5305 (Saruparib) PJ34 HCl AG-14361 Iniparib (BSI-201) G007-LK UPF 1069 A-966492 Pamiparib ME0328 |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| A4-Fuk | Growth Inhibition Assay | IC50=43.5691 μM | SANGER | |||
| JVM-2 | Growth Inhibition Assay | IC50=42.9207 μM | SANGER | |||
| MOLT-4 | Growth Inhibition Assay | IC50=42.2538 μM | SANGER | |||
| KARPAS-45 | Growth Inhibition Assay | IC50=41.4818 μM | SANGER | |||
| HCC1187 | Growth Inhibition Assay | IC50=41.2771 μM | SANGER | |||
| NB14 | Growth Inhibition Assay | IC50=40.7031 μM | SANGER | |||
| SW982 | Growth Inhibition Assay | IC50=38.0998 μM | SANGER | |||
| IGROV-1 | Growth Inhibition Assay | IC50=39.3304 μM | SANGER | |||
| CAL-54 | Growth Inhibition Assay | IC50=37.966 μM | SANGER | |||
| T47D | Growth Inhibition Assay | IC50=37.7018 μM | SANGER | |||
| H4 | Growth Inhibition Assay | IC50=37.567 μM | SANGER | |||
| LAMA-84 | Growth Inhibition Assay | IC50=36.7345 μM | SANGER | |||
| MV-4-11 | Growth Inhibition Assay | IC50=35.8499 μM | SANGER | |||
| KG-1 | Growth Inhibition Assay | IC50=33.6001 μM | SANGER | |||
| JVM-3 | Growth Inhibition Assay | IC50=35.5868 μM | SANGER | |||
| MFE-280 | Growth Inhibition Assay | IC50=33.3889 μM | SANGER | |||
| KU812 | Growth Inhibition Assay | IC50=32.3642 μM | SANGER | |||
| COLO-684 | Growth Inhibition Assay | IC50=33.3599 μM | SANGER | |||
| A2780 | Growth Inhibition Assay | IC50=30.7457 μM | SANGER | |||
| RS4-11 | Growth Inhibition Assay | IC50=30.4241 μM | SANGER | |||
| EM-2 | Growth Inhibition Assay | IC50=29.4901 μM | SANGER | |||
| L-363 | Growth Inhibition Assay | IC50=29.4798 μM | SANGER | |||
| MOLT-13 | Growth Inhibition Assay | IC50=29.3814 μM | SANGER | |||
| EW-13 | Growth Inhibition Assay | IC50=29.3814 μM | SANGER | |||
| LU-139 | Growth Inhibition Assay | IC50=29.3748 μM | SANGER | |||
| 697 | Growth Inhibition Assay | IC50=29.0235 μM | SANGER | |||
| LB771 | Growth Inhibition Assay | IC50=28.8373 μM | SANGER | |||
| SK-MEL-1 | Growth Inhibition Assay | IC50=12.4663 μM | SANGER | |||
| CAL-33 | Growth Inhibition Assay | IC50=10.434 μM | SANGER | |||
| HAL-01 | Growth Inhibition Assay | IC50=9.8862 μM | SANGER | |||
| KASUMI-1 | Growth Inhibition Assay | IC50=8.89266 μM | SANGER | |||
| NCI-H720 | Growth Inhibition Assay | IC50=8.43603 μM | SANGER | |||
| C41 | Kinase Assay | 30 min | Inhibition of PARP1 with EC50 of 0.002 μM | 19888760 | ||
| Jurkat | Kinase Assay | 96 h | DMSO | Inhibition of PARP1 assessed as reduction of cell viability with EC50 of 3 μM | 23850199 | |
| Capan1 | Growth Inhibition Assay | 72 h | DMSO | Antiproliferative activity against BRCA2 gene mutated human Capan1 cells with IC50 of 39.7 μM | 24398383 | |
| ML-1 | Apoptotic Assay | 2.5 μM | 24 h | DMSO | Synergistically enhances TRAIL-induced apoptosis in ML-1 cells | 24895135 |
| HCT-116 | Kinase Assay | 0.5 μM | 24 h | PARP activity decreases | 23054213 | |
| UM-SCC1 | Cytotoxic Assay | 10 μM | 24 h | Reduces the cell viability | 21912620 | |
| FaDu | Cytotoxic Assay | 10 μM | 24 h | Reduces the cell viability | 21912620 | |
| LoVo | Function assay | 30 mins | Inhibition of PARP in human LoVo cells assessed as inhibition of poly(ADP)-ribose polymerization for 30 mins by fluorescence assay, EC50 = 0.00594 μM. | 26652717 | ||
| LoVo | Function assay | 30 mins | Inhibition of PARP in human LoVo cells assessed as inhibition of poly(ADP)-ribose polymerization for 30 mins by fluorescence assay, EC50 = 0.00594 μM. | 26652717 | ||
| LoVo | Function assay | 30 mins | Inhibition of PARP in human LoVo cells assessed as inhibition of poly(ADP)-ribose polymerization for 30 mins by fluorescence assay, EC50 = 0.00594 μM. | 26652717 | ||
| VC8 | Cytotoxicity assay | 3 days | Cytotoxicity against Chinese hamster VC8 cells harboring BRCA2 deficient after 3 days by CCK8 assay, CC50 = 2.344 μM. | 29335205 | ||
| LoVo | Cytotoxicity assay | 0.4 uM | 5 days | Potentiation of -induced cytotoxicity in human LoVo cells assessed as GI50 at 0.4 uM after 5 days by Celltiter-Glo assay, GI50 = 6.203 μM. | 26652717 | |
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Saos-2 cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Orthogonal 3D viability screen for SK-N-SH cells | 29435139 | |||
| TC32 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for TC32 cells | 29435139 | |||
| T98G | Growth Inhibition Assay | IC50=44.8517 μM | SANGER | |||
| BALL-1 | Growth Inhibition Assay | IC50=43.9532 μM | SANGER | |||
| MDA-MB-361 | Growth Inhibition Assay | IC50=43.8414 μM | SANGER | |||
| SBC-1 | Growth Inhibition Assay | IC50=41.3063 μM | SANGER | |||
| MOLT-16 | Growth Inhibition Assay | IC50=36.952 μM | SANGER | |||
| BEN | Growth Inhibition Assay | IC50=27.9566 μM | SANGER | |||
| HCC70 | Growth Inhibition Assay | IC50=27.7246 μM | SANGER | |||
| NCI-H1693 | Growth Inhibition Assay | IC50=27.2898 μM | SANGER | |||
| HLE | Growth Inhibition Assay | IC50=27.054 μM | SANGER | |||
| NCI-H2029 | Growth Inhibition Assay | IC50=26.4238 μM | SANGER | |||
| CTV-1 | Growth Inhibition Assay | IC50=25.8969 μM | SANGER | |||
| TGBC11TKB | Growth Inhibition Assay | IC50=25.6863 μM | SANGER | |||
| HCC1937 | Growth Inhibition Assay | IC50=24.746 μM | SANGER | |||
| MHH-NB-11 | Growth Inhibition Assay | IC50=23.1363 μM | SANGER | |||
| A388 | Growth Inhibition Assay | IC50=21.9091 μM | SANGER | |||
| ECC10 | Growth Inhibition Assay | IC50=20.7455 μM | SANGER | |||
| Daoy | Growth Inhibition Assay | IC50=19.5649 μM | SANGER | |||
| KYSE-150 | Growth Inhibition Assay | IC50=18.9986 μM | SANGER | |||
| NKM-1 | Growth Inhibition Assay | IC50=18.5119 μM | SANGER | |||
| H9 | Growth Inhibition Assay | IC50=18.2833 μM | SANGER | |||
| COLO-668 | Growth Inhibition Assay | IC50=17.6294 μM | SANGER | |||
| GP5d | Growth Inhibition Assay | IC50=17.053 μM | SANGER | |||
| DEL | Growth Inhibition Assay | IC50=16.6717 μM | SANGER | |||
| ChaGo-K-1 | Growth Inhibition Assay | IC50=16.5325 μM | SANGER | |||
| RPMI-8226 | Growth Inhibition Assay | IC50=16.2042 μM | SANGER | |||
| OS-RC-2 | Growth Inhibition Assay | IC50=15.9589 μM | SANGER | |||
| EW-3 | Growth Inhibition Assay | IC50=14.5565 μM | SANGER | |||
| DU-145 | Growth Inhibition Assay | IC50=13.9053 μM | SANGER | |||
| MN-60 | Growth Inhibition Assay | IC50=13.5389 μM | SANGER | |||
| SK-NEP-1 | Growth Inhibition Assay | IC50=13.166 μM | SANGER | |||
| HEC-1 | Growth Inhibition Assay | IC50=12.9196 μM | SANGER | |||
| KY821 | Growth Inhibition Assay | IC50=12.485 μM | SANGER | |||
| Ramos-2G6-4C10 | Growth Inhibition Assay | IC50=12.4752 μM | SANGER | |||
| DT40 | Cytotoxic Assay | 72 h | DMSO | Cytotoxicity against chicken BRCA2-deficient DT40 cells | 24922587 | |
| PC-3 | Growth Inhibition Assay | 10 μM | Induces a significant inhibition in colony formation | 21571912 | ||
| C41 | Function assay | Inhibition of PARP1 in human C41 cells, EC50 = 0.002 μM. | 19143569 | |||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | |||
| SJ-GBM2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Orthogonal 3D viability screen for SJ-GBM2 cells | 29435139 | |||
| EoL-1-cell | Growth Inhibition Assay | IC50=1.0798 μM | SANGER | |||
| NCI-SNU-5 | Growth Inhibition Assay | IC50=3.12841 μM | SANGER | |||
| BV-173 | Growth Inhibition Assay | IC50=5.45409 μM | SANGER | |||
| HCC1806 | Growth Inhibition Assay | IC50=5.75173 μM | SANGER | |||
| COLO-680 | Growth Inhibition Assay | IC50=6.21406 μM | SANGER | |||
| HCC2218 | Growth Inhibition Assay | IC50=7.79704 μM | SANGER | |||
| SK-MEL-24 | Growth Inhibition Assay | IC50=7.81924 μM | SANGER | |||
| Mo-T | Growth Inhibition Assay | IC50=45.6389 μM | SANGER | |||
| MHH-PREB-1 | Growth Inhibition Assay | IC50=45.7585 μM | SANGER | |||
| ALL-PO | Growth Inhibition Assay | IC50=47.3791 μM | SANGER | |||
| NCI-H510A | Growth Inhibition Assay | IC50=47.9034 μM | SANGER | |||
| ML-2 | Growth Inhibition Assay | IC50=49.7856 μM | SANGER | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 49 mg/mL
(200.58 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 244.29 | Formula | C13H16N4O |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 912444-00-9 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | NSC 737664 | Smiles | CC1(CCCN1)C2=NC3=C(C=CC=C3N2)C(=O)N | ||
Mechanism of Action
| Features |
Increases the efficacy of common cancer therapies such as radiation and alkylating agents.
|
|---|---|
| Targets/IC50/Ki |
PARP2
(Cell-free assay) 2.9 nM(Ki)
PARP1
(Cell-free assay) 5.2 nM(Ki)
|
| In vitro |
Veliparib (ABT-888) is inactive to SIRT2 (>5 μM). It inhibits the PARP activity with EC50 of 2 nM in C41 cells. This compound could decrease the PAR levels in both irradiated and nonirradiated H460 cells. It also reduces clonogenic survival and inhibits DNA repair by PARP-1 inhibition in H460 cells. When combined with radiation, it increases apoptosis and autophagy in H460 cells. It also inhibits PARP activity in H1299, DU145 and 22RV1 cells and the inhibition is independent of p53 function. At 10 μM, it suppresses the surviving fraction (SF) by 43% in the clonogenic H1299 cells. It shows effective radiosensitivity in oxic H1299 cells. Furthermore, it could attenuate the SF of hypoxic-irradiated cells including H1299, DU145 and 22RV1. |
| Kinase Assay |
In vitro PARP assays
|
|
PARP assays for Veliparib (ABT-888) are conducted in a buffer containing 50 mM Tris (pH 8.0), 1 mM DTT, 1.5 μM [3H]NAD+ (1.6 μCi/mmol), 200 nM biotinylated histone H1, 200 nM slDNA, and 1 nM PARP-1 or 4 nM PARP-2 enzyme. Reactions are terminated with 1.5 mM benzamide, transferred to streptavidin Flash plates, and counted using a TopCount microplate scintillation counter.
|
|
| In vivo |
Veliparib (ABT-888) has an oral bioavailability of 56%-92% in mice, Sprague-Dawley rats, beagle dogs, and cynomolgus monkeys after oral administration. This compound (25 mg/kg i.p.) could improve tumor growth delay in a NCI-H460 xenograft model with well tolerated. Combination with radiation, it decreases the tumor vessel formation. It reduces intratumor PAR levels by more than 95% at a dose of 3 and 12.5 mg/kg in A375 and Colo829 xenograft models and the suppression could be maintained over time. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | p-STAT3 / STAT3 / p-AKT(S473) / p-AKT(T308) / p-ERK / p-p38 |
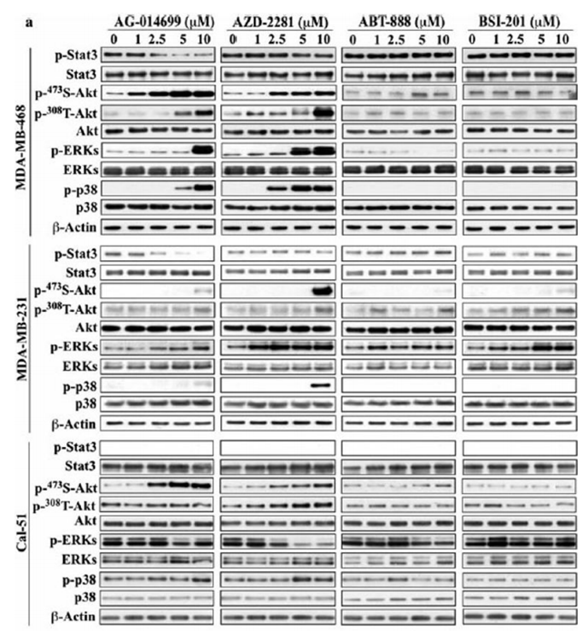
|
22678161 |
| Immunofluorescence | HuR BRCA1 |
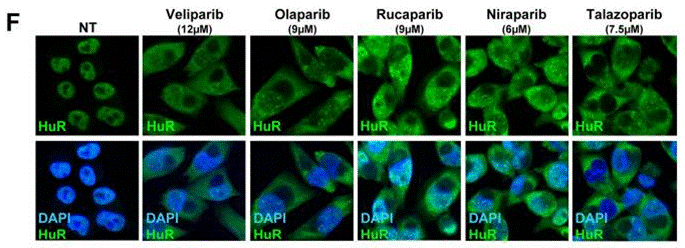
|
28687616 |
| Growth inhibition assay | Cell viability (TNBC cell lines) Cell viability (melanoma cells) |
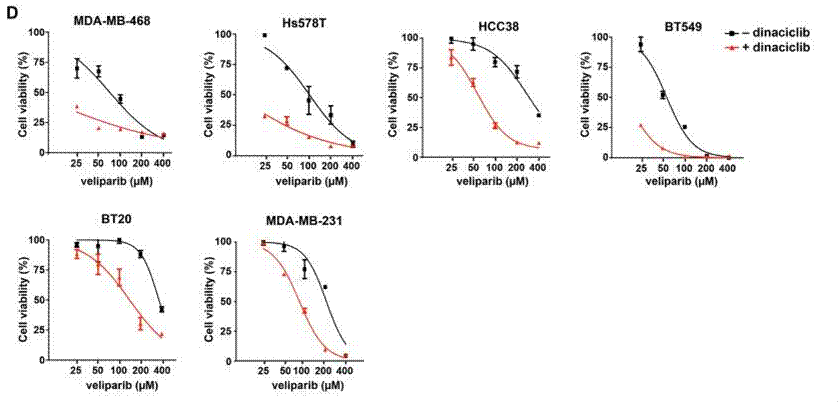
|
27880910 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT03044795 | Withdrawn | Cancer |
University Medical Center Groningen|AbbVie|Dutch Cancer Society |
November 2019 | Phase 2 |
| NCT02723864 | Completed | Neoplasms |
National Cancer Institute (NCI)|National Institutes of Health Clinical Center (CC) |
August 9 2017 | Phase 1 |
| NCT02483104 | Completed | Ovarian Cancer |
AbbVie |
July 2015 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































