research use only
A-1155463 Dihydrochloride Bcl-2 inhibitor
Cat.No.S7800

Chemical Structure
Molecular Weight: 742.71
Quality Control
| Related Targets | Caspase PD-1/PD-L1 Ferroptosis p53 Apoptosis related Synthetic Lethality STAT TNF-alpha Ras KRas |
|---|---|
| Other Bcl-2 Inhibitors | Navitoclax (ABT-263) S63845 ABT-737 Obatoclax Mesylate (GX15-070) A-1331852 A-1210477 TW-37 AZD5991 UMI-77 (R)-(-)-Gossypol (AT-101) acetic acid |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| NCI-H146 cells | Cytotoxicity assay | 48 hrs | Cytotoxicity against BCL-XL-dependent human NCI-H146 cells in presence of 10% human serum incubated for 48 hrs by Cell titerGlo reagent based assay, EC50=0.065 μM | 25313317 | ||
| MOLT4 | Cytotoxicity assay | Cytotoxicity against human Bcl-XL dependant MOLT4 cells by cell based assay, EC50=0.07μM | 26988306 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 100 mg/mL
(134.64 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 742.71 | Formula | C35H34Cl2FN5O4S2 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1235034-55-5(free base) | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | CN(C)CC#CC1=CC(=C(C=C1)OCCCC2=C(N=C(S2)N3CCC4=C(C3)C(=CC=C4)C(=O)NC5=NC6=CC=CC=C6S5)C(=O)O)F | ||
Mechanism of Action
| Targets/IC50/Ki |
BCL-XL
(Cell-free assay) <0.01 nM(Ki)
|
|---|---|
| In vitro |
A-1155463 Dihydrochloride disrupts BCL-XL-BIM but not BCL-2-BIM complexes in cells. A-1155463 kills BCL-XL-dependent Molt-4 cells (EC50=70 nM) but has no measurable cytotoxicity against BCL-2-dependent RS4;11 cells (EC50>5 mM). A-1155463 induces the hallmarks of apoptosis, as evidenced by the release of cytochrome c from mitochondria, caspase activation, and the accumulation of caspase-dependent sub-G0-G1 DNA content in BCL-XL-dependent H146 cells. |
| In vivo |
Following a single 5 mg/kg IP dose of A-1155463 in nontumor bearing SCID-Beige mice, platelet counts fall dramatically as measured at 6 h postdose and then rebound to normal levels within 72 h. Daily Dosing at 5 mg/kg IP to SCID-Beige mice that had been inoculated with BCL-XL-dependent H146 tumor cells for 14 days causes a statistically significant inhibition of tumor growth (maximum tumor growth inhibition = 44%), which is alleviated upon cessation of dosing. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Growth inhibition assay | EC50 |
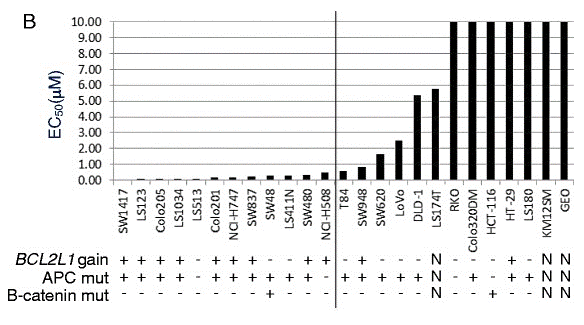
|
26134786 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































