research use only
Butein EGFR inhibitor
Cat.No.S8036
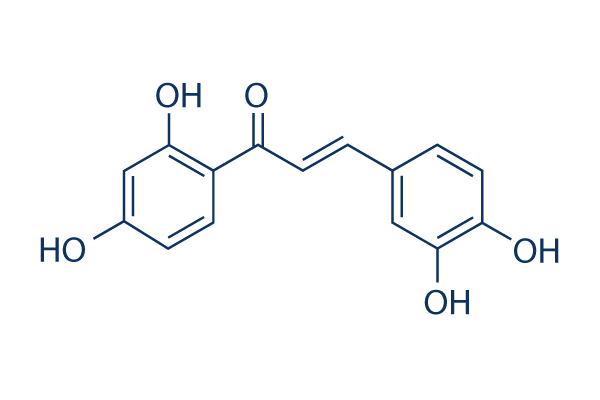
Chemical Structure
Molecular Weight: 272.25
Quality Control
| Related Targets | VEGFR PDGFR FGFR c-Met Src MEK CSF-1R FLT3 HER2 c-Kit |
|---|---|
| Other EGFR Inhibitors | Lazertinib (YH25448) Icotinib Hydrochloride Sunvozertinib AG-490 AG-1478 Canertinib (CI-1033) Genistein Rociletinib (CO-1686) Poziotinib (NOV120101, HM781-36B) WZ4002 |
Solubility
|
In vitro |
DMSO
: 55 mg/mL
(202.02 mM)
Ethanol : 55 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 272.25 | Formula | C15H12O5 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 487-52-5 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | C1=CC(=C(C=C1C=CC(=O)C2=C(C=C(C=C2)O)O)O)O | ||
Mechanism of Action
| Targets/IC50/Ki |
EGFR
|
|---|---|
| In vitro |
Butein inhibits the epidermal growth factor (EGF)-stimulated auto-phosphotyrosine level of EGF receptor in HepG2 cells, and also inhibits tyrosine-specific protein kinase activities of EGF receptor and p60c-src with IC50 of 65 μM in vitro. The inhibition is competitive to ATP and non-competitive to the phosphate acceptor, poly (Glu, Ala, Tyr) 6:3:1 for EGF receptor tyrosine kinase. In contrast, this compound non-significantly inhibits the activities of serine- and threonine-specific protein kinases such as PKC or PKA.
This chemical inhibits Nuclear Factor(NF)-κB and NF-κB-regulated gene expression through direct inhibition of IκBα Kinase β on Cysteine 179 Residue. It (10 μM) inhibits over 90% iNOS and COX-2 expression, as well as nitrite and TNF-α production in LPS-stimulated RAW 264.7 cells. This compound (10 μM) inhibits LPS-induced DNA binding activity of NF-κB, which is mediated through inhibition of the degradation of inhibitory factor-κB and phosphorylation of Erk1/2 MAP kinase, as well as increases binding of the osteopontin a vb3 integrin receptor. It (20 μM) treatment induces morphologic changes of bladder cancer cells BLS(M) from elongated morphology to rounded epithelial-like cells, accompanied by downregulation of vimentin, and gaining of E-cadherin compared to untreated control cells, indicating the reversal of mesenchymal-like phenotype. This chemical (20 μM) suppresses motility and invasion capacity of BLS(M) cells, and reverts EMT-like phenotype induced by TNF-α, through the ERK1/2 and NF-κB signaling pathways.
It inhibits the constitutive activation of STAT3 in HepG2 cells in a dose-dependent manner, with maximum inhibition occurring at 50 μM, mediated through the inhibition of activation of upstream kinases c-Src and Janus-activated kinase2. This compound (50 μM) also could completely inhibit IL-6-induced STAT3 phosphorylation in SNU-387 cells. It downregulates the expression of cyclin D1, Bcl-2, Bcl-xL, survivin, and VEGF, markers of STAT3 activation. This chemical (50 μM) significantly enhance the apoptotic effects of doxorubicin from 18% to 55% and of paclitaxel from 15% to 42%.
It is as a powerful antioxidant against lipid and LDL peroxidation. This compound inhibits iron-induced lipid peroxidation in rat brain homogenate with an IC50 of 3.3 μM. It is as potent α-tocopherol in reducing the stable free radical diphenyl-2-picrylhydrazyl (DPPH) with IC0.2 of 9.2 μM. This chemical also inhibits the activity of xanthine oxidase with an IC50 of 5.9 μM. It scavenges the peroxyl radical derived from 2,2-azobis(2-amidinopropane) dihydrochloride (AAPH) in aqueous phase. Furthermore, this compound inhibits copper-catalyzed oxidation of human low-density lipoprotein (LDL) in a concentration-dependent manner. It is a chelator of ferrous and copper ions.
|
| In vivo |
Butein at 2 mg/kg induces significant inhibition of hepatocellular tumor growth compared with the corn oil-treated controls. At necropsy on day 22 after initial treatment, there is more than 2-fold decrease in tumor growth in this compound-treated group (mean relativetumor burden, 3.90) compared with the control group (8.46), associated with reduced constitutive p-STAT3 (9% vs 81% of vehicle group), Bcl-2 levels (26% vs 96% of vehicle group), and increased caspase-3 level (98% vs 21% of vehicle group) in HCC tumor tissues.
This compound shows antifibrogenic activity. It (25 mg/kg/day) reduces serum AST and ALT activation to 35% and 69%, respectively, of control CCl4-induced rat levels. This chemical (25 mg/kg/day) reduces liver hydroxyproline contents and TBAR4 concentration to 54% and 54%, respectively. α1(I) collagen and TIMP-1 expression in this compound-treated rats is 28% and 20.3% compared with the values for the respective CCl4-treated control.
|
References |
|
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































