research use only
Tamoxifen Estrogen Receptor modulator
Cat.No.S1238
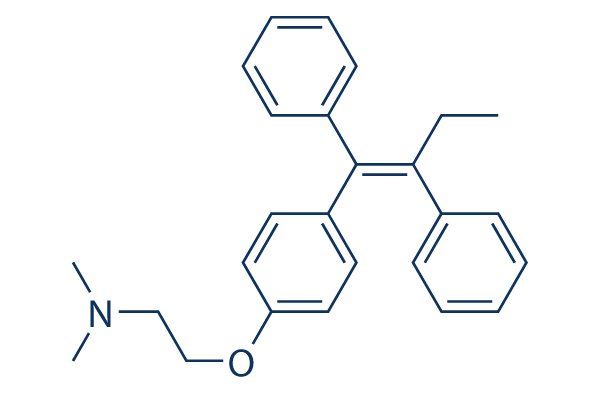
Chemical Structure
Molecular Weight: 371.51
Quality Control
| Related Targets | EGFR VEGFR JAK PDGFR FGFR Src HIF FLT FLT3 HER2 |
|---|---|
| Other Antineoplastic and Immunosuppressive Antibiotics Inhibitors | Staurosporine (STS) Cyclosporin A Oligomycin A (MCH 32) Puromycin Dihydrochloride Nigericin sodium salt Geldanamycin (NSC 122750) Honokiol Streptozotocin (STZ) Sodium Monensin (NSC 343257) Cephalomannine |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| MDA-MB-435 cells | Function assay | Inhibition of MDA-MB-435 breast cancer cell proliferation, IC50=0.05 μM | 14971899 | |||
| MDA-MB-231 cells | Function assay | Inhibition of estrogen receptor negative MDA-MB-231 breast cancer cell proliferation, IC50=0.08 μM | 14971899 | |||
| MCF7 | Antiproliferative assay | 72 hrs | Antiproliferative activity against MCF7 cells after 72 hrs by MTT assay, Activity = 4.6 μM. | 17918820 | ||
| MCF7 | Antiestrogenic assay | 96 hrs | Antiestrogenic activity in human MCF7 cells assessed as drug level causing inhibition of 100 pM E2-enhanced cell proliferation to level equivalent to 50 pM E2 after 96 hrs by alamar blue assay, Activity = 0.1 μM. | 19943620 | ||
| T47D | Antiestrogenic assay | 96 hrs | Antiestrogenic activity in human T47D cells assessed as drug level causing inhibition of 100 pM E2-enhanced cell proliferation to level equivalent to 50 pM E2 after 96 hrs by alamar blue assay, Activity = 0.1 μM. | 19943620 | ||
| MCF7 | Antiestrogenic assay | 96 hrs | Antiestrogenic activity in human MCF7 cells assessed as drug level causing inhibition of 100 pM E2-enhanced cell proliferation to level equivalent to 10 pM E2 after 96 hrs by alamar blue assay, Activity = 0.2 μM. | 19943620 | ||
| MCF7 | Antiestrogenic assay | 96 hrs | Antiestrogenic activity in human MCF7 cells assessed as drug level causing inhibition of 100 pM E2-enhanced cell proliferation to level equivalent to 1 pM E2 after 96 hrs by alamar blue assay, Activity = 0.4 μM. | 19943620 | ||
| T47D | Antiestrogenic assay | 96 hrs | Antiestrogenic activity in human T47D cells assessed as drug level causing inhibition of 100 pM E2-enhanced cell proliferation to level equivalent to 10 pM E2 after 96 hrs by alamar blue assay, Activity = 0.7 μM. | 19943620 | ||
| T47D | Antiestrogenic assay | 96 hrs | Antiestrogenic activity in human T47D cells assessed as drug level causing inhibition of 100 pM E2-enhanced cell proliferation to level equivalent to 1 pM E2 after 96 hrs by alamar blue assay, Activity = 9 μM. | 19943620 | ||
| HepG2 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human HepG2 cells after 24 hrs by resazurin dye-based fluorescence analysis, CC50 = 24.7 μM. | 30322754 | ||
| HEK293 | Cytotoxicity assay | 24 hrs | Cytotoxicity against HEK293 cells after 24 hrs by resazurin dye-based fluorescence analysis, CC50 = 39.4 μM. | 30322754 | ||
| MCF-7 | Cytotoxicity assay | Cytotoxicity against MCF-7 cells in the absence of 1 uM E2 (estradiol), Cytotoxicity = 9.8 μM. | 9089332 | |||
| MCF-7 | Cytotoxicity assay | Cytotoxicity against MCF-7 cells in presence of 1 uM E2 (estradiol), Cytotoxicity = 13 μM. | 9089332 | |||
| MCF-7 | Function assay | Effective dose for [3H]- estradiol against proliferation of MCF-7 cells, EC50 = 0.00011 μM. | 2769681 | |||
| Ishikawa | Function assay | Concentration of compound required to induce 50 % of the maximum stimulation of alkaline phosphatase activity in Ishikawa cells, EC50 = 0.033 μM. | 8201587 | |||
| MCF-7 | Function assay | Effective concentration against MCF-7 breast tumor cells using MCF-7 assay., EC50 = 0.01 μM. | 12825935 | |||
| ScN2a-cl3 | Function assay | 5 days | Inhibition of RML prion protein infected in mouse dividing ScN2a-cl3 cells expressing full length mouse PrP assessed as reduction of PrPsc level after 5 days by ELISA, EC50 = 4.39 μM. | 24183589 | ||
| NCI-H460 | Anticancer assay | 24 hrs | Anticancer activity against human NCI-H460 cells after 24 hrs by SRB assay, GI50 = 4.48 μM. | 19733085 | ||
| DU145 | Anticancer assay | 24 hrs | Anticancer activity against human DU145 cells after 24 hrs by SRB assay, GI50 = 6.07 μM. | 19733085 | ||
| SKOV3 | Anticancer assay | 24 hrs | Anticancer activity against human SKOV3 cells after 24 hrs by SRB assay, GI50 = 6.4 μM. | 19733085 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells expressing estrogen receptor incubated with 0.003 nM of estradiol and 0.01 nM of testosterone after 48 hrs by sulforhodamine B assay in presence of 5% fetal bovine serum, GI50 = 2.29087 μM. | 21513275 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells expressing estrogen receptor incubated with 0.06 nM of estradiol and 0.07 nM of testosterone after 48 hrs by sulforhodamine B assay in presence of fetal bovine serum and NuSerum, GI50 = 2.95121 μM. | 21513275 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells expressing estrogen receptor incubated with 100 nM of estradiol and 0.07 nM of testosterone after 48 hrs by sulforhodamine B assay in presence of fetal bovine serum and NuSerum, GI50 = 5.62341 μM. | 21513275 | ||
| MCF12A | Antiproliferative assay | 48 hrs | Antiproliferative activity against estrogen receptor-deficient human MCF12A cells incubated with 0.003 nM of estradiol and 0.01 nM of testosterone after 48 hrs by sulforhodamine B assay in presence of 5% fetal bovine serum, GI50 = 5.7544 μM. | 21513275 | ||
| MCF12A | Antiproliferative assay | 48 hrs | Antiproliferative activity against estrogen receptor-deficient human MCF12A cells incubated with 100 nM of estradiol and 0.07 nM of testosterone after 48 hrs by sulforhodamine B assay in presence of fetal bovine serum and NuSerum, GI50 = 5.7544 μM. | 21513275 | ||
| NCI-ADR-RES | Antiproliferative assay | 48 hrs | Antiproliferative activity against estrogen receptor-deficient human NCI-ADR-RES cells incubated with 0.003 nM of estradiol and 0.01 nM of testosterone after 48 hrs by sulforhodamine B assay in presence of 5% fetal bovine serum, GI50 = 5.7544 μM. | 21513275 | ||
| MCF12A | Antiproliferative assay | 48 hrs | Antiproliferative activity against estrogen receptor-deficient human MCF12A cells incubated with 0.06 nM of estradiol and 0.07 nM of testosterone after 48 hrs by sulforhodamine B assay in presence of fetal bovine serum and NuSerum, GI50 = 6.91831 μM. | 21513275 | ||
| NCI-ADR-RES | Antiproliferative assay | 48 hrs | Antiproliferative activity against estrogen receptor-deficient human NCI-ADR-RES cells incubated with 0.06 nM of estradiol and 0.07 nM of testosterone after 48 hrs by sulforhodamine B assay in presence of fetal bovine serum and NuSerum, GI50 = 9.54993 μM. | 21513275 | ||
| LNCAP | Antiproliferative assay | 48 hrs | Antiproliferative activity against human LNCAP cells expressing androgen receptor incubated with 0.6 nM of estradiol and 0.7 nM of testosterone after 48 hrs by sulforhodamine B assay, GI50 = 10 μM. | 21513275 | ||
| PC3 | Antiproliferative assay | 48 hrs | Antiproliferative activity against androgen receptor-deficient human PC3 cells incubated with 0.6 nM of estradiol and 0.7 nM of testosterone after 48 hrs by sulforhodamine B assay, GI50 = 10.9648 μM. | 21513275 | ||
| NCI-ADR-RES | Antiproliferative assay | 48 hrs | Antiproliferative activity against estrogen receptor-deficient human NCI-ADR-RES cells incubated with 100 nM of estradiol and 0.07 nM of testosterone after 48 hrs by sulforhodamine B assay in presence of fetal bovine serum and NuSerum, GI50 = 11.4815 μM. | 21513275 | ||
| DU145 | Antiproliferative assay | 48 hrs | Antiproliferative activity against androgen receptor-deficient human DU145 cells incubated with 0.6 nM of estradiol and 0.7 nM of testosterone after 48 hrs by sulforhodamine B assay, GI50 = 13.8038 μM. | 21513275 | ||
| HT-29 | Growth inhibition assay | Growth inhibition of human HT-29 cells, GI50 = 9.5 μM. | 23403084 | |||
| M21 | Growth inhibition assay | Growth inhibition of human M21 cells, GI50 = 11 μM. | 23403084 | |||
| MCF7 | Growth inhibition assay | Growth inhibition of estrogen-dependent human MCF7 cells, GI50 = 11.2 μM. | 23403084 | |||
| MDA-MB-231 | Growth inhibition assay | Growth inhibition of estrogen-independent human MDA-MB-231 cells, GI50 = 18.8 μM. | 23403084 | |||
| MCF7 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MCF7 cells after 72 hrs by trypan blue assay, GI50 = 12 μM. | 23735829 | ||
| MSTO-211H | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MSTO-211H cells after 72 hrs by trypan blue assay, GI50 = 23.3 μM. | 23735829 | ||
| HeLa | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HeLa cells after 72 hrs by trypan blue assay, GI50 = 32.6 μM. | 23735829 | ||
| K562 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human K562 cells after 72 hrs, GI50 = 5.2 μM. | 25420175 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Anti-proliferative activity against human MCF7 cells incubated for 48 hrs by SRB assay, GI50 = 8 μM. | 26163220 | ||
| DU145 | Antiproliferative assay | 48 hrs | Anti-proliferative activity against human DU145 cells incubated for 48 hrs by SRB assay, GI50 = 19.3 μM. | 26163220 | ||
| HepG2 | Antiproliferative assay | 48 hrs | Anti-proliferative activity against human HepG2 cells incubated for 48 hrs by SRB assay, GI50 = 21.7 μM. | 26163220 | ||
| A549 | Antiproliferative assay | 48 hrs | Anti-proliferative activity against human A549 cells incubated for 48 hrs by SRB assay, GI50 = 24.4 μM. | 26163220 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells assessed as growth inhibition after 48 hrs by SRB assay, GI50 = 1.58 μM. | 26896706 | ||
| T47D | Cytotoxicity assay | 48 hrs | Cytotoxicity against human T47D cells assessed as growth inhibition after 48 hrs by SRB assay, GI50 = 1.99 μM. | 26896706 | ||
| MOLT4 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MOLT4 cells assessed as growth inhibition after 48 hrs by SRB assay, GI50 = 2.5 μM. | 26896706 | ||
| MDA-MB-468 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MDA-MB-468 cells assessed as growth inhibition after 48 hrs by SRB assay, GI50 = 2.5 μM. | 26896706 | ||
| HCT15 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HCT15 cells assessed as growth inhibition after 48 hrs by SRB assay, GI50 = 3.16 μM. | 26896706 | ||
| MDA-MB-435 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MDA-MB-435 cells assessed as growth inhibition after 48 hrs by SRB assay, GI50 = 3.16 μM. | 26896706 | ||
| SNB75 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human SNB75 cells assessed as growth inhibition after 48 hrs by SRB assay, GI50 = 5.01 μM. | 26896706 | ||
| EKVX | Cytotoxicity assay | 48 hrs | Cytotoxicity against human EKVX cells assessed as growth inhibition after 48 hrs by SRB assay, GI50 = 6.3 μM. | 26896706 | ||
| UO31 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human UO31 cells assessed as growth inhibition after 48 hrs by SRB assay, GI50 = 6.3 μM. | 26896706 | ||
| SKOV3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human SKOV3 cells assessed as growth inhibition after 48 hrs by SRB assay, GI50 = 10 μM. | 26896706 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells assessed as cell growth inhibition after 72 hrs by trypan blue assay, GI50 = 6.5 μM. | 27128175 | ||
| HeLa | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HeLa cells assessed as cell growth inhibition after 72 hrs by trypan blue assay, GI50 = 12 μM. | 27128175 | ||
| A2780 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A2780 cells assessed as cell growth inhibition after 72 hrs by MTS assay, GI50 = 30.3 μM. | 27128175 | ||
| OVCAR5 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human OVCAR5 cells assessed as cell growth inhibition after 72 hrs by MTS assay, GI50 = 32 μM. | 27128175 | ||
| MCF7 | Cytotoxicity assay | 120 hrs | Cytotoxicity against human MCF7 cells assessed as growth inhibition after 120 hrs by MTT assay, GI50 = 6.8 μM. | 28152429 | ||
| T47D | Cytotoxicity assay | 120 hrs | Cytotoxicity against human T47D cells assessed as growth inhibition after 120 hrs by MTT assay, GI50 = 10.6 μM. | 28152429 | ||
| SKBR3 | Cytotoxicity assay | 120 hrs | Cytotoxicity against human SKBR3 cells assessed as growth inhibition after 120 hrs by MTT assay, GI50 = 12.5 μM. | 28152429 | ||
| MDA-MB-231 | Cytotoxicity assay | 120 hrs | Cytotoxicity against human MDA-MB-231 cells assessed as growth inhibition after 120 hrs by MTT assay, GI50 = 15.1 μM. | 28152429 | ||
| BT-20 | Function assay | 10 nM | Inhibitory activity against proliferation of BT-20 cells in presence of TPE at 10e-5 M concentration., IC30 = 5.7 μM. | 2769681 | ||
| MCF-7 | Function assay | 1 nM | Inhibitory activity against proliferation of MCF-7 cells in presence of 10e-6 M concentration of [3H]- estradiol (E2), IC30 = 6.5 μM. | 2769681 | ||
| MCF-7 | Function assay | Inhibition of [3H]thymidine incorporation into MCF-7 cells, IC50 = 7.2 μM. | 1573630 | |||
| MCF-7 | Cytotoxicity assay | Cytostaticity against MCF-7 cells in the presence of 1 uMolar E2 (estradiol), IC50 = 8 μM. | 9089332 | |||
| MCF-7 | Function assay | Inhibition of estrogen-induced proliferation in human MCF-7 breast cancer cells, IC50 = 0.481 μM. | 9154963 | |||
| Ishikawa | Antiestrogenic assay | 4 days | Antiestrogenic activity in human Ishikawa cells assessed as inhibition of estrogen-induced alkaline phosphatase activity after 4 days by ELISA, IC50 = 0.42 μM. | 9784163 | ||
| NIH-3T3-G185 | Function assay | TP_TRANSPORTER: inhibition of Daunorubicin efflux in NIH-3T3-G185 cells, IC50 = 6.4 μM. | 11716514 | |||
| NIH-3T3-G185 | Function assay | TP_TRANSPORTER: inhibition of LDS-751 efflux in NIH-3T3-G185 cells, IC50 = 12.1 μM. | 11716514 | |||
| NIH-3T3-G185 | Function assay | TP_TRANSPORTER: inhibition of Rhodamine 123 efflux in NIH-3T3-G185 cells, IC50 = 31.4 μM. | 11716514 | |||
| MCF-7-2a | Antiestrogenic assay | Antiestrogenic activity in MCF-7-2a cells as concentration required to reduce estradiol effect by 50%, IC50 = 0.5 μM. | 12431063 | |||
| MCF-7-2a | Function assay | Inhibition of estradiol induced estrogen receptor transcriptional activation in MCF-7-2a cells, IC50 = 0.5 μM. | 12672249 | |||
| SK-E2 | Function assay | TP_TRANSPORTER: increase in bodipy intracellular accumulation (Bodipy: 0.2 uM) in SK-E2 cells (expressing BSEP), IC50 = 15.4 μM. | 12739759 | |||
| SK-E2 | Function assay | TP_TRANSPORTER: increase in dihydrofluorescein intracellular accumulation (dihydrofluorescein: 1 uM) in SK-E2 cells (expressing BSEP), IC50 = 23.3 μM. | 12739759 | |||
| MCF-7 | Function assay | Inhibitory concentration against MCF-7 breast tumor cells using MCF-7 assay., IC50 = 0.58 μM. | 12825935 | |||
| Caco-2 | Function assay | TP_TRANSPORTER: transepithelial transport of Rhodamine 123 (basal to apical) in Caco-2 cells, IC50 = 29 μM. | 15386482 | |||
| HeLa | Function assay | Inhibition of 17-beta-estradiol mediated luciferase transcription in HeLa cells expressing human estrogen receptor alpha; ERE assay, IC50 = 0.622 μM. | 15658851 | |||
| MCF-7 | Antiproliferative assay | Antiproliferative activity in MCF-7 human breast cancer cells, IC50 = 2.3 μM. | 15658875 | |||
| MDA-MB 231 | Antiproliferative assay | Antiproliferative activity in MDA-MB 231 human breast cancer cells, IC50 = 10.6 μM. | 15658875 | |||
| MCF7 | Antiproliferative assay | 3 mg/kg/day | 48 hrs | Antiproliferative activity against MCF7 cells at 3 mg/kg/day after 48 hrs, IC50 = 5.09 μM. | 16979337 | |
| MCF7 | Antiproliferative assay | 24 hrs | Antiproliferative activity against human MCF7 cells after 24 hrs by MTS assay, IC50 = 0.58 μM. | 18272256 | ||
| insect cells | Function assay | Displacement of fluorescein labeled estradiol from human recombinant ERalpha expressed in baculovirus infected insect cells by fluorescence polarization assay, IC50 = 0.07 μM. | 18835176 | |||
| insect cells | Function assay | Displacement of fluorescein labeled estradiol from human recombinant ERbeta expressed in baculovirus infected insect cells by fluorescence polarization assay, IC50 = 0.17 μM. | 18835176 | |||
| Ishikawa | Antiestrogenic assay | 72 hrs | Antiestrogenic activity in human Ishikawa cells assessed as inhibition of estrogen-induced alkaline phosphatase activity after 72 hrs, IC50 = 0.17 μM. | 18835176 | ||
| MCF7 | Antiproliferative assay | 72 hrs | Antiproliferative activity against estrogen receptor dependent human MCF7 cells after 72 hrs by MTT assay, IC50 = 4.12 μM. | 18835176 | ||
| T47D | Antiestrogenic assay | Antiestrogenic activity in human T47D cells expressing estrogen receptor assessed as estrogen-dependent transcription by luciferase reporter gene assay, IC50 = 0.1 μM. | 19131248 | |||
| T47D | Growth inhibition assay | 72 hrs | Growth inhibition of human estrogen-dependent T47D cells after 72 hrs, IC50 = 1 μM. | 19131248 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells expressing estrogen receptor after 48 hrs by MTT assay, IC50 = 10 μM. | 19423356 | ||
| MDA-MB-231 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human estrogen receptor deficient MDA-MB-231 cells after 48 hrs by MTT assay, IC50 = 20 μM. | 19423356 | ||
| MCF7 | Anticancer assay | 18 hrs | Anticancer activity against estrogen-positive human MCF7 cells after 18 hrs by MTT assay, IC50 = 12 μM. | 19446930 | ||
| MDA-MB-231 | Anticancer assay | 18 hrs | Anticancer activity against estrogen-positive human MDA-MB-231 cells after 18 hrs by MTT assay, IC50 = 24 μM. | 19446930 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 7.65 μM. | 19740667 | ||
| MDA-MB-231 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MDA-MB-231 cells after 48 hrs by MTT assay, IC50 = 9.86 μM. | 19740667 | ||
| MCF7 | Cytotoxicity assay | Cytotoxicity against human adriamycin- resistant MCF7 cells by WST-1 assay, IC50 = 10.9 μM. | 19836230 | |||
| MCF7 | Cytotoxicity assay | Cytotoxicity against human tamoxifen-resistant MCF7 cells by WST-1 assay, IC50 = 11.1 μM. | 19836230 | |||
| MCF7 | Cytotoxicity assay | Cytotoxicity against human MCF7 cells by WST-1 assay, IC50 = 11.4 μM. | 19836230 | |||
| MDA-MB-231 | Cytotoxicity assay | Cytotoxicity against human MDA-MB-231 cells by WST-1 assay, IC50 = 12.4 μM. | 19836230 | |||
| MDA-MB-231 | Antiproliferative assay | Antiproliferative activity against human MDA-MB-231 cells by MTT assay, IC50 = 0.66 μM. | 20451380 | |||
| MCF7 | Antiproliferative assay | Antiproliferative activity against human MCF7 cells by MTT assay, IC50 = 1 μM. | 20451380 | |||
| HepG2 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human HepG2 cells after 24 hrs by CellTiter-BlueCell viability assay, IC50 = 23.4 μM. | 20545334 | ||
| HT-29 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human HT-29 cells after 24 hrs by CellTiter-BlueCell viability assay, IC50 = 38.6 μM. | 20545334 | ||
| MCF7 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human MCF7 cells after 24 hrs by MTT assay, IC50 = 8.9 μM. | 20605470 | ||
| HEK293 | Cytotoxicity assay | 24 hrs | Cytotoxicity against HEK293 cells after 24 hrs by MTT assay, IC50 = 10 μM. | 20605470 | ||
| MDA-MB-231 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human MDA-MB-231 cells after 24 hrs by MTT assay, IC50 = 10 μM. | 20605470 | ||
| HeLa | Cytotoxicity assay | 24 hrs | Cytotoxicity against human HeLa cells after 24 hrs by MTT assay, IC50 = 12.5 μM. | 20605470 | ||
| MCF7 | Anticancer assay | Anticancer activity against human MCF7 cells expressing estrogen receptor by MTT assay, IC50 = 11.8 μM. | 20951035 | |||
| MCF7 | Cytotoxicity assay | Cytotoxicity activity against human MCF7 cells by MTS assay, IC50 = 31 μM. | 21680064 | |||
| HEK293 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HEK293 cells after 48 hrs by MTT assay, IC50 = 10 μM. | 21871812 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells expressing estrogen receptor after 48 hrs by MTT assay, IC50 = 12.48 μM. | 21871812 | ||
| MDA-MB-231 | Antiproliferative assay | 48 hrs | Antiproliferative activity against estrogen receptor-deficient human MDA-MB-231 cells expressing estrogen receptor after 48 hrs by MTT assay, IC50 = 18.9 μM. | 21871812 | ||
| Ishikawa | Antiproliferative assay | 48 hrs | Antiproliferative activity against human Ishikawa cells expressing estrogen receptor after 48 hrs by MTT assay, IC50 = 22.5 μM. | 21871812 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells after 48 hrs by SRB assay, IC50 = 0.027 μM. | 22472045 | ||
| AH109 | Function assay | 24 hrs | Antagonist activity at ERbeta receptor LBD expressed in yeast AH109 cells assessed as inhibition of interaction with SRC1 after 24 hrs by alpha-galactosidase assay, IC50 = 1.66 μM. | 22647217 | ||
| AH109 | Function assay | 24 hrs | Antagonist activity at ERalpha receptor LBD expressed in yeast AH109 cells assessed as inhibition of interaction with SRC1 after 24 hrs by alpha-galactosidase assay, IC50 = 2.54 μM. | 22647217 | ||
| MCF7 | Antiproliferative assay | 24 hrs | Antiproliferative activity against human MCF7 cells assessed as inhibition of cell viability after 24 hrs by MTT assay, IC50 = 19.54 μM. | 22647217 | ||
| DLD1 | Cytotoxicity assay | 96 hrs | Cytotoxicity against human DLD1 cells after 96 hrs by SRB assay, IC50 = 4.78 μM. | 22749392 | ||
| MCF7 | Cytotoxicity assay | 96 hrs | Cytotoxicity against human MCF7 cells after 96 hrs by SRB assay, IC50 = 7.1 μM. | 22749392 | ||
| NIH/3T3 | Cytotoxicity assay | 96 hrs | Cytotoxicity against mouse NIH/3T3 cells after 96 hrs by SRB assay, IC50 = 7.26 μM. | 22749392 | ||
| 518A2 | Cytotoxicity assay | 96 hrs | Cytotoxicity against human 518A2 cells after 96 hrs by SRB assay, IC50 = 7.62 μM. | 22749392 | ||
| A2780 | Cytotoxicity assay | 96 hrs | Cytotoxicity against human A2780 cells after 96 hrs by SRB assay, IC50 = 7.77 μM. | 22749392 | ||
| Lipo | Cytotoxicity assay | 96 hrs | Cytotoxicity against human Lipo cells after 96 hrs by SRB assay, IC50 = 8.64 μM. | 22749392 | ||
| A253 | Cytotoxicity assay | 96 hrs | Cytotoxicity against human A253 cells after 96 hrs by SRB assay, IC50 = 8.92 μM. | 22749392 | ||
| A549 | Cytotoxicity assay | 96 hrs | Cytotoxicity against human A549 cells after 96 hrs by SRB assay, IC50 = 9.66 μM. | 22749392 | ||
| 850C | Cytotoxicity assay | 96 hrs | Cytotoxicity against human 850C cells after 96 hrs by SRB assay, IC50 = 11.09 μM. | 22749392 | ||
| KIF5 | Toxicity assay | 24 hrs | Toxicity against human KIF5 cells incubated for 24 hrs by CellTiter Blue cell viability assay, IC50 = 23.75 μM. | 22775474 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells after 72 hrs by WST1 assay in presence of 20% FBS, IC50 = 8.6 μM. | 22901895 | ||
| HL60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60 cells after 72 hrs by WST1 assay in presence of 20% FBS, IC50 = 14.3 μM. | 22901895 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 11.44 μM. | 23022281 | ||
| MDA-MB-231 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MDA-MB-231 cells after 48 hrs by MTT assay, IC50 = 12.41 μM. | 23022281 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells after 48 hrs by sulforhodamine B assay, IC50 = 1.82 μM. | 23123017 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 5.5 μM. | 23287057 | ||
| DU145 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human DU145 cells after 48 hrs by MTT assay, IC50 = 5.5 μM. | 23287057 | ||
| HepG2 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HepG2 cells after 48 hrs by MTT assay, IC50 = 6.3 μM. | 23287057 | ||
| PC3 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human PC3 cells after 48 hrs by MTT assay, IC50 = 10 μM. | 23287057 | ||
| NIH/3T3 | Antiproliferative assay | 48 hrs | Antiproliferative activity against mouse NIH/3T3 cells after 48 hrs by MTT assay, IC50 = 11.4 μM. | 23287057 | ||
| MDA-MB-231 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MDA-MB-231 cells after 48 hrs by MTT assay, IC50 = 11.4 μM. | 23287057 | ||
| A549 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human A549 cells after 48 hrs by MTT assay, IC50 = 12 μM. | 23287057 | ||
| T47D | Antiproliferative assay | 48 hrs | Antiproliferative activity against human T47D cells after 48 hrs by MTT assay, IC50 = 13 μM. | 23287057 | ||
| MCF7 | Growth inhibition assay | 6 days | Antagonist activity at ERalpha receptor in human MCF7 cells assessed as inhibition of cell growth after 6 days by crystal violet staining method, IC50 = 0.2 μM. | 23448346 | ||
| HEK293T | Function assay | 20 hrs | Antagonist activity at Gal4 DBD-fused human ERalpha LBD expressed in HEK293T cells assessed as inhibition of estradiol-induced transcriptional activation after 20 hrs by luciferase reporter gene assay, IC50 = 0.039 μM. | 23786452 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells expressing ERalpha and ERbeta assessed as growth inhibition after 72 hrs by MTT assay, IC50 = 15.6 μM. | 23786452 | ||
| Vero | Cytotoxicity assay | 72 hrs | Cytotoxicity against african green monkey Vero cells assessed as growth inhibition after 72 hrs by MTT assay, IC50 = 15.9 μM. | 23786452 | ||
| MDA-MB-231 | Cytotoxicity assay | 72 hrs | Cytotoxicity against ERalpha and ERbeta-deficient human MDA-MB-231 cells assessed as growth inhibition after 72 hrs by MTT assay, IC50 = 17 μM. | 23786452 | ||
| DU145 | Cytotoxicity assay | 72 hrs | Cytotoxicity against ERalpha-deficient human DU145 cells expressing ERbeta assessed as growth inhibition after 72 hrs by MTT assay, IC50 = 28.9 μM. | 23786452 | ||
| Jurkat | Cytotoxicity assay | 48 hrs | Cytotoxicity against human Jurkat cells after 48 hrs by MTT assay, IC50 = 8.3 μM. | 23792352 | ||
| MCF7 | Cytotoxicity assay | Cytotoxicity against human MCF7 cells by SRB assay, IC50 = 5.19 μM. | 23806014 | |||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 12.35 μM. | 23830503 | ||
| MCF7 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MCF7 cells after 72 hrs by MTT assay, IC50 = 27.96 μM. | 23860592 | ||
| MCF7 | Cytotoxicity assay | 5 days | Cytotoxicity against human MCF7 cells after 5 days, IC50 = 0.794 μM. | 23864928 | ||
| T47D | Cytotoxicity assay | 5 days | Cytotoxicity against human T47D cells after 5 days, IC50 = 1.13 μM. | 23864928 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 15.2 μM. | 23880359 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 13.45 μM. | 23902804 | ||
| MCF7 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human MCF7 cells after 24 hrs by MTT assay, IC50 = 9 μM. | 24457094 | ||
| MDA-MB-231 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human MDA-MB-231 cells after 24 hrs by MTT assay, IC50 = 10 μM. | 24457094 | ||
| HEK293 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human HEK293 cells after 24 hrs by MTT assay, IC50 = 26 μM. | 24457094 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 1.89 μM. | 24721727 | ||
| MCF7 | Antiproliferative assay | 24 hrs | Antiproliferative activity against human MCF7 cells after 24 hrs by MTT assay, IC50 = 6 μM. | 24880230 | ||
| PC3 | Antiproliferative assay | 24 hrs | Antiproliferative activity against human PC3 cells after 24 hrs by MTT assay, IC50 = 7 μM. | 24880230 | ||
| MDA-MB-231 | Antiproliferative assay | 24 hrs | Antiproliferative activity against human MDA-MB-231 cells after 24 hrs by MTT assay, IC50 = 14 μM. | 24880230 | ||
| MCF7 | Antiproliferative assay | 2 days | Antiproliferative activity human ER-positive MCF7 cells after 2 days by MTT assay, IC50 = 4.67 μM. | 25222876 | ||
| MIAPaCa2 | Antiproliferative assay | 2 days | Antiproliferative activity human MIAPaCa2 cells after 2 days by MTT assay, IC50 = 33.12 μM. | 25222876 | ||
| MDA-MB-231 | Antiproliferative assay | 2 days | Antiproliferative activity human ER-negative MDA-MB-231 cells after 2 days by MTT assay, IC50 = 38.97 μM. | 25222876 | ||
| MCF7 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MCF7 cells after 72 hrs by MTT assay, IC50 = 4.1 μM. | 25369367 | ||
| MCF7 | Antiproliferative assay | 5 days | Antiproliferative activity against human MCF7 cells after 5 days by WST-8 assay, IC50 = 2.1 μM. | 25614118 | ||
| MCF7 | Antiproliferative assay | 3 days | Antiproliferative activity against human MCF7 cells assessed as reduction in cell growth incubated at 37 degC for 3 days by resazurin microplate assay, IC50 = 18.71 μM. | 25734623 | ||
| MCF7 | Cytotoxicity assay | Cytotoxicity against human MCF7 cells assessed as cell viability by MTT assay, IC50 = 3.8 μM. | 26079090 | |||
| MDA-MB-435S | Cytotoxicity assay | Cytotoxicity against human MDA-MB-435S cells assessed as cell viability by MTT assay, IC50 = 5.5 μM. | 26079090 | |||
| Vero | Cytotoxicity assay | Cytotoxicity against African green monkey Vero cells assessed as cell viability by MTT assay, IC50 = 5.7 μM. | 26079090 | |||
| A549 | Cytotoxicity assay | Cytotoxicity against human A549 cells assessed as cell viability by MTT assay, IC50 = 8.4 μM. | 26079090 | |||
| GBM2 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human GBM2 cells assessed as reduction in cell viability incubated for 72 hrs by WST-1 method, IC50 = 3.5 μM. | 26355532 | ||
| GBM3 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human GBM3 cells assessed as reduction in cell viability incubated for 72 hrs by WST-1 method, IC50 = 4.7 μM. | 26355532 | ||
| GBM1 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human GBM1 cells assessed as reduction in cell viability incubated for 72 hrs by WST-1 method, IC50 = 7.5 μM. | 26355532 | ||
| MCF7 | Antiproliferative assay | Antiproliferative activity against human MCF7 cells, IC50 = 0.05754 μM. | 26407012 | |||
| MCF | Function assay | Antagonist activity at progesterone receptor in human MCF cells assessed as estradiol-induced receptor response, IC50 = 0.12882 μM. | 26407012 | |||
| MCF7 | Function assay | Antagonist activity at ERalpha receptor in human MCF7 cells, IC50 = 10 μM. | 26407012 | |||
| MCF7 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MCF7 cells after 72 hrs by MTT assay, IC50 = 4.4 μM. | 26896706 | ||
| MRC5 | Cytotoxicity assay | 4 to 7 days | Cytotoxicity against human MRC5 cells assessed as cell viability after 4 to 7 days by Alamar Blue staining based fluorometric analysis, IC50 = 10.9 μM. | 26922226 | ||
| MCF7 | Antiproliferative assay | Antiproliferative activity against human MCF7 cells, IC50 = 13.7 μM. | 27155469 | |||
| MDA-MB-231 | Antiproliferative assay | Antiproliferative activity against human MDA-MB-231 cells, IC50 = 17.9 μM. | 27155469 | |||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells incubated for 48 hrs by MTT assay, IC50 = 5.3 μM. | 27176944 | ||
| Ishikawa | Antiproliferative assay | 48 hrs | Antiproliferative activity against human Ishikawa cells incubated for 48 hrs by MTT assay, IC50 = 6 μM. | 27176944 | ||
| MDA-MB-231 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MDA-MB-231 cells incubated for 48 hrs by MTT assay, IC50 = 13.9 μM. | 27176944 | ||
| MCF7 | Cytotoxicity assay | Cytotoxicity against human MCF7 cells by resazurin microplate assay, IC50 = 6.99 μM. | 27228159 | |||
| MCF7 | Cytotoxicity assay | Cytotoxicity against human MCF7 cells assessed as reduction in cell viability by resazurin microplate assay, IC50 = 19.33 μM. | 27311894 | |||
| MCF7 | Antiproliferative assay | 72 hrs | Antiproliferative activity against ER-positive human MCF7 cells assessed as reduction in cell viability measured after 72 hrs by AlamarBlue assay, IC50 = 4.1 μM. | 27407030 | ||
| insect cells | Function assay | 4 hrs | Displacement of Fluormone ES2 Green from human recombinant full length ERalpha expressed in insect cells measured up to 4 hrs by fluorescence polarization assay, IC50 = 0.222 μM. | 27647375 | ||
| SH-SY5Y | Function assay | 1 hr | Inhibition of PKC in human SH-SY5Y cells assessed as inhibition of PMA-stimulated MARCKS phosphorylation preincubated for 1 hr followed by PMA-stimulation for 15 mins by Western blot analysis, IC50 = 20 μM. | 27647375 | ||
| FADU | Anticancer assay | 48 hrs | Anticancer activity against human FADU cells measured after 48 hrs by SRB assay, IC50 = 5.39 μM. | 27671497 | ||
| DLD1 | Anticancer assay | 48 hrs | Anticancer activity against human DLD1 cells measured after 48 hrs by SRB assay, IC50 = 11.06 μM. | 27671497 | ||
| A549 | Anticancer assay | 48 hrs | Anticancer activity against human A549 cells measured after 48 hrs by SRB assay, IC50 = 17.3 μM. | 27671497 | ||
| DU145 | Anticancer assay | 48 hrs | Anticancer activity against human DU145 cells measured after 48 hrs by SRB assay, IC50 = 18.07 μM. | 27671497 | ||
| MCF7 | Anticancer assay | 48 hrs | Anticancer activity against human ER-positive MCF7 cells measured after 48 hrs by SRB assay, IC50 = 18.18 μM. | 27671497 | ||
| MCF7 | Function assay | 24 hrs | Antagonist activity at ERalpha in human MCF7 cells assessed as inhibition of estrogen-induced transcription preincubated overnight followed by estrogen addition measured after 24 hrs by dual luciferase reporter gene assay, IC50 = 1.605 μM. | 28296398 | ||
| insect cells | Function assay | Displacement of fluorescein-labeled estradiol (fluoromone) from human recombinant full-length untagged estrogen receptor beta expressed in insect cells by fluorescence polarization assay, IC50 = 0.188 μM. | 28426931 | |||
| Ishikawa | Function assay | 72 hrs | Antagonist activity at estrogen receptor alpha/beta in human Ishikawa cells after 72 hrs by alkaline phosphatase assay, IC50 = 0.28 μM. | 28426931 | ||
| MCF7 | Antiproliferative assay | 72 hrs | Antiproliferative activity against ER-dependent human MCF7 cells after 72 hrs by MTT assay, IC50 = 4.1 μM. | 28426931 | ||
| MCF7 | Cytotoxicity assay | 5 days | Cytotoxicity against human MCF7 cells assessed as reduction in cell viability after 5 days by SRB assay, IC50 = 4.4 μM. | 28426931 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against ER positive human MCF7 cells after 48 hrs by MTT assay, IC50 = 11.35 μM. | 28460819 | ||
| Ishikawa | Antiproliferative assay | 48 hrs | Antiproliferative activity against human Ishikawa cells after 48 hrs by MTT assay, IC50 = 16.47 μM. | 28460819 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells assessed as reduction in cell viability after 48 hrs by SRB assay, IC50 = 21.57 μM. | 28505536 | ||
| HeLa | Cytotoxicity assay | 24 hrs | Cytotoxicity against human HeLa cells assessed as reduction in cell viability after 24 hrs by MTT assay, IC50 = 8.85 μM. | 28838694 | ||
| HeLa | Function assay | 24 hrs | Antagonist activity at full length ERalpha (unknown origin) expressed in human HeLa cells incubated for 24 hrs by ERE-driven luciferase reporter gene assay, IC50 = 0.341 μM. | 28882502 | ||
| HeLa | Function assay | 24 hrs | Antagonist activity at full length ERbeta (unknown origin) expressed in human HeLa cells incubated for 24 hrs by ERE-driven luciferase reporter gene assay, IC50 = 1.53 μM. | 28882502 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells after 72 hrs by resazurin based fluorescence assay, IC50 = 7.94 μM. | 28927795 | ||
| insect cells | Function assay | 2 hrs | Displacement of fluorescent estrogen ligand from recombinant human ERalpha expressed in insect cells incubated for 2 hrs by polarization, IC50 = 2.67 μM. | 28942113 | ||
| MCF7 | Cytotoxicity assay | Cytotoxicity in human MCF7 cells assessed as inhibition of cell growth incubated for 48 hrs by MTT assay, IC50 = 14.35 μM. | 28942113 | |||
| Ishikawa | Cytotoxicity assay | Cytotoxicity in human Ishikawa cells assessed as inhibition of cell growth incubated for 48 hrs by MTT assay, IC50 = 20.65 μM. | 28942113 | |||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 8.12 μM. | 29054359 | ||
| MDA-MB-231 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MDA-MB-231 cells after 48 hrs by MTT assay, IC50 = 9.96 μM. | 29054359 | ||
| HEK293 | Antiproliferative assay | 48 hrs | Antiproliferative activity against HEK293 cells after 48 hrs by MTT assay, IC50 = 20.93 μM. | 29054359 | ||
| MCF7 | Cytotoxicity assay | Cytostatic activity against human MCF7 cells, IC50 = 2.7 μM. | 29207335 | |||
| MCF7 | Growth inhibition assay | 72 hrs | Growth inhibition of human MCF7 cells after 72 hrs by MTT assay, IC50 = 5.3 μM. | 29220789 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 19.5 μM. | 29482944 | ||
| MCF7 | Function assay | 24 hrs | Induction of selective estrogen receptor alpha degradation in human MCF7 cells harboring TK-ERE-Luc assessed as reduction in estradiol-induced transcriptional activity after 24 hrs by luciferase reporter gene assay, IC50 = 1.605 μM. | 29562737 | ||
| insect cells | Function assay | 2 hrs | Displacement of Fluormone ES2 Green from full length human ER-alpha expressed in insect cells after 2 hrs by fluorescence polarization assay, IC50 = 1.42 μM. | 29587221 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 15.46 μM. | 29587221 | ||
| Ishikawa | Antiproliferative assay | 48 hrs | Antiproliferative activity against human Ishikawa cells after 48 hrs by MTT assay, IC50 = 26.52 μM. | 29587221 | ||
| MRC5 SV2 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MRC5 SV2 cells after 72 hrs by resazurin dye-based fluorimetric analysis, IC50 = 10.2 μM. | 29604541 | ||
| MCF7 | Cytotoxicity assay | 4 hrs | Cytotoxicity against human MCF7 cells preincubated for 4 hrs followed by incubation in compound free media for 24 hrs by MTT assay, IC50 = 8.61 μM. | 29605808 | ||
| DLD1 | Cytotoxicity assay | 4 hrs | Cytotoxicity against human DLD1 cells preincubated for 4 hrs followed by incubation in compound free media for 24 hrs by MTT assay, IC50 = 16.3 μM. | 29605808 | ||
| A549 | Cytotoxicity assay | 4 hrs | Cytotoxicity against human A549 cells preincubated for 4 hrs followed by incubation in compound free media for 24 hrs by MTT assay, IC50 = 17.3 μM. | 29605808 | ||
| HeLa | Function assay | 4.5 hrs | Inhibition of Ebolavirus glycoprotein/matrix protein VP40 entry in human HeLa cells after 4.5 hrs beta-lactamase reporter assay, IC50 = 0.73 μM. | 29624387 | ||
| MRC5 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MRC5 cells assessed as decrease in cell viability after 72 hrs by resazurin assay, IC50 = 10 μM. | 29672041 | ||
| MCF7 | Antiproliferative assay | 24 hrs | Antiproliferative activity against human MCF7 cells after 24 hrs by MTT assay, IC50 = 7.09 μM. | 30360952 | ||
| MDA-MB-231 | Antiproliferative assay | 24 hrs | Antiproliferative activity against human MDA-MB-231 cells after 24 hrs by MTT assay, IC50 = 47.1 μM. | 30360952 | ||
| HeLa | Function assay | 18 hrs | Displacement of [2,4,6,7-3H]estradiol from human ERalpha expressed in HeLa cells after 18 hrs by liquid scintillation counting, Ki = 0.03119 μM. | 20621492 | ||
| HEK293 | Function assay | 1.5 hrs | Displacement of [3H]LSD from human 5HT6 receptor expressed in HEK293 cells after 1.5 hrs by liquid scintillation counting, Ki = 1.041 μM. | 22537153 | ||
| ScN2a-cl3 | Cytotoxicity assay | 5 days | Cytotoxicity against mouse dividing ScN2a-cl3 cells assessed as cell viability after 5 days by calcein-AM staining-based fluorescence assay, LC50 = 4.89 μM. | 24183589 | ||
| MCF7 | Function assay | >10 uM | Agonist activity at ERalpha assessed as inhibition of E2-induced luciferase gene transactivation in MCF7 cells at >10 uM | 16735120 | ||
| HEK293 | Function assay | 30 mins | Stimulation of GFP-tagged human PLD2 expressed in human HEK293 cells assessed as phosphatidylbutanol-[d9] production after 30 mins by mass spectrometric analysis | 19136975 | ||
| MDA-MB-231 | Cytotoxicity assay | 3 days | Cytostatic activity against human MDA-MB-231 cells after 3 days by microtiter assay | 20598555 | ||
| HeLa | Function assay | 1 uM | 48 hrs | Antagonist activity at human ERalpha expressed in human HeLa cells coexpressing ERE-E1b-Luc assessed as inhibition of estradiol-induced transcriptional activation at 1 uM after 48 hrs by luciferase reporter gene assay | 20621492 | |
| MCF7 | Cytotoxicity assay | 6 uM | 96 hrs | Cytotoxicity against human MCF7 cells assessed as reduction of cell proliferation at 6 uM after 96 hrs by MTT assay | 20621492 | |
| HeLa | Function assay | Antagonist activity at human wild type ERalpha expressed in human HeLa cells co-expressing ERE assessed as inhibition of transactivation activity by luciferase reporter gene assay | 20659801 | |||
| HeLa | Function assay | Transactivation of human wild type ERalpha expressed in human HeLa cells co-expressing AP-1 by luciferase reporter gene assay | 20659801 | |||
| HeLa | Function assay | Antagonist activity at human wild type ERbeta expressed in human HeLa cells co-expressing ERE assessed as inhibition of transactivation activity by luciferase reporter gene assay | 20659801 | |||
| HeLa | Function assay | Agonist activity at human wild type ERbeta expressed in human HeLa cells co-expressing AP-1 assessed as transactivation activity by luciferase reporter gene assay | 20659801 | |||
| MCF7 | Antiestrogenic assay | 20 nM | 48 hrs | Antiestrogenic activity in human ER-positive MCF7 cells assessed as inhibition of 17beta estradiol-induced secreted alkaline phosphatase activity at 20 nM after 48 hrs by phospha-light reporter chemiluminescence assay | 21800859 | |
| Escherichia coli C41 DE3 | Function assay | 60 uM | 10 mins | Inactivation of CYP2B6 (unknown origin) expressed in Escherichia coli C41 DE3 cells assessed as modified apo-P450 2B6 adduct formation at 60 uM after 10 mins by ESI-LC-MS analysis in presence of NADPH | 22942317 | |
| Escherichia coli C41 DE3 | Function assay | 100 uM | 30 mins | Inactivation of CYP2B6 (unknown origin) expressed in Escherichia coli C41 DE3 cells assessed as tryptic peptide 188FHYQDQE194 with site of adduct formation localized to Gln193 at 100 uM after 30 mins by LC-MS/MS analysis in presence of NADPH | 22942317 | |
| Escherichia coli C41 DE3 | Function assay | 60 uM | Inactivation of CYP2B6 (unknown origin) expressed in Escherichia coli C41 DE3 cells assessed as modified apo-P450 2B6 adduct formation at 60 uM by ESI-LC-MS analysis in presence of NADPH and GSHEE | 22942317 | ||
| MCF7 | Cytotoxicity assay | 5 uM | 48 hrs | Cytotoxicity against human MCF7 cells assessed as change in cell viability at 5 uM after 48 hrs by MTT assay | 23287057 | |
| HL60 | Function assay | Inhibition of Delta(8)-Delta(7) sterol isomerase/Delta(24)-sterol reductase-mediated cholesterol biosynthesis in human HL60 cells assessed as increase in zymostenol and zymosterol accumulation | 23433667 | |||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells assessed as reduction in cell viability in presence of 10 mM NAC after 48 hrs by WST8 assay | 25163667 | ||
| Neuro2a | Function assay | 1 uM | Inhibition of delta 8-7 isomerase in Dhcr7-deficient mouse Neuro2a cells assessed as increase in zymostenol levels at 1 uM by LC-MS/GC-MS analysis | 26789657 | ||
| Neuro2a | Function assay | 1 uM | Inhibition of delta 8-7 isomerase in mouse Neuro2a cells assessed as decrease in 7-DHC levels at 1 uM by LC-MS/GC-MS analysis | 26789657 | ||
| Neuro2a | Function assay | 1 uM | Inhibition of delta 8-7 isomerase in Dhcr7-deficient mouse Neuro2a cells assessed as increase in zymosterol levels at 1 uM by LC-MS/GC-MS analysis | 26789657 | ||
| Neuro2a | Function assay | 1 uM | Inhibition of delta 8-7 isomerase in Dhcr7-deficient mouse Neuro2a cells assessed as decrease in cholesterol levels at 1 uM by LC-MS/GC-MS analysis | 26789657 | ||
| Neuro2a | Function assay | 1 uM | Inhibition of delta 8-7 isomerase in Dhcr7-deficient mouse Neuro2a cells assessed as decrease in 7-DHC levels at 1 uM by LC-MS/GC-MS analysis | 26789657 | ||
| Neuro2a | Function assay | 1 uM | Inhibition of DR24 in mouse Neuro2a cells assessed as decrease in 7-DHC levels at 1 uM by LC-MS/GC-MS analysis | 26789657 | ||
| Neuro2a | Function assay | 1 uM | Inhibition of DR24 in Dhcr7-deficient mouse Neuro2a cells assessed as decrease in 7-DHC levels at 1 uM by LC-MS/GC-MS analysis | 26789657 | ||
| AsPC1 | Antiproliferative assay | 10 uM | 48 hrs | Antiproliferative activity against human AsPC1 cells assessed as cell viability at 10 uM after 48 hrs by MTS assay | 26803204 | |
| BxPC3 | Antiproliferative assay | 10 uM | 48 hrs | Antiproliferative activity against human BxPC3 cells assessed as cell viability at 10 uM after 48 hrs by MTS assay | 26803204 | |
| MIAPaCa2 | Antiproliferative assay | 10 uM | 48 hrs | Antiproliferative activity against human MIAPaCa2 cells assessed as cell viability at 10 uM after 48 hrs by MTS assay | 26803204 | |
| MCF7 | Function assay | 3 to 10 uM | 24 hrs | Inhibition of Set7/9 in human MCF7 cells assessed as suppression of beta-estradiol-induced ERE promoter activation at 3 to 10 uM preincubated for 18 hrs followed by beta-estradiol addition measured after 24 hrs by luciferase reporter gene assay | 27088648 | |
| MCF7 | Function assay | 3 to 10 uM | 24 hrs | Inhibition of Set7/9 in human MCF7 cells assessed as suppression of beta-estradiol-induced pS2 mRNA expression at 3 to 10 uM preincubated for 18 hrs followed by beta-estradiol addition measured after 24 hrs by RT-PCR analysis | 27088648 | |
| MCF7 | Function assay | 1 uM | Inhibition of ERalpha in human MCF7 cells assessed as reduction in 10 nM estrogen-induced progesterone receptor expression at 1 uM incubated for 24 hrs by Western blot method | 28942113 | ||
| MCF7 | Function assay | 1 uM | 16 hrs | Inhibition of ERalpha in human MCF7 cells assessed as downregulation of 17beta-estradiol-mediated pS2 mRNA expression at 1 uM preincubated for 4 hrs followed by 17beta-estradiol addition measured after 16 hrs by quantitative RT-PCR analysis | 29045135 | |
| MCF7 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human MCF7 cells assessed as cell viability up to 100 uM after 24 hrs by MTT assay | 29045135 | ||
| U2O2 | Function assay | 1 uM | 20 hrs | Inhibition of recombinant human GFP-fused ERalpha LBD (301 to 553 residues) expressed in human U2O2 cells assessed as downregulation of 17beta-estradiol-induced transcription at 1 uM preincubated for 4 hrs followed by E2 addition measured after 20 hrs by | 29045135 | |
| MCF7 | Cell cycle arrest assay | 10 uM | 24 to 48 hrs | Cell cycle arrest in human MCF7 cells assessed as accumulation at G0/G1 phase at 10 uM after 24 to 48 hrs by propidium iodide staining based flow cytometry | 29054359 | |
| MCF7 | Function assay | 1 uM | Antagonist activity at ERalpha in human MCF7 cells assessed as reduction in estradiol-induced PgR mRNA expression at 1 uM by SYBR Green dye based RT-PCR analysis | 29482944 | ||
| Ishikawa | Antiproliferative assay | >40 uM | 48 hrs | Antiproliferative activity against human Ishikawa cells at >40 uM after 48 hrs by MTT assay | 29482944 | |
| MCF7 | Function assay | 60 mg/kg | 5 days per week for 48 days | Downregulation of progesterone receptor mRNA level in NOD SCID gamma mouse xenografted with human MCF7 cells at 60 mg/kg, po 5 days per week for 48 days post tumor implantation measured 7 hrs post last dose by RT-PCR method | 29562737 | |
| MCF7 | Antitumor assay | 60 mg/kg | 4 weeks | Antitumor activity against human MCF7 cells xenografted in nu/nu mouse model of tamoxifen-sensitive breast cancer xenograft implanted with 17beta-estradiol time release pellets assessed as reduction tumor volume at 60 mg/kg, po qd for 4 weeks | 30086626 | |
| insect cells | Function assay | 0.1 to 10000 uM | Binding affinity to untagged human recombinant full length ERalpha expressed in insect cells 0.1 to 10000 uM by Fluormone ES2 Green displacement based assay | 30091920 | ||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
Ethanol : 74 mg/mL
DMSO
: 10 mg/mL
(26.91 mM)
Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 371.51 | Formula | C26H29NO |
Storage (From the date of receipt) | 2 years 4°C, in dark and seal |
|---|---|---|---|---|---|
| CAS No. | 10540-29-1 | -- | Storage of Stock Solutions |
|
|
| Synonyms | ICI 46474, (Z)-Tamoxifen, trans-Tamoxifen | Smiles | CCC(=C(C1=CC=CC=C1)C2=CC=C(C=C2)OCCN(C)C)C3=CC=CC=C3 | ||
Mechanism of Action
| Targets/IC50/Ki |
estrogen receptor
|
|---|---|
| In vitro |
TAM treatment inhibits significantly MCF7 cell proliferation. Low doses of this compound are able to induce structural chromosomal aberrations (deletions, isochromosomes, translocations, and dicentric chromosomes) in both ER+ and ER- breast cancer cells. This genotoxic effect is higher in those cell lines with HER2 gene amplification. Whereas this chemical at lower concentrations (0.1-1 μM) induces a cell-cycle arrest, pharmacological concentrations (above 5 μM) of TAM have been found to induce apoptosis of breast cancer cells. 5 μM TAM rapidly induced sustained activation of ERK1/2 in ER-positive breast cancer cell lines (MCF-7 and T47D) |
| In vivo |
Tamoxifen (TAM) is widely used for both treatment and prevention of breast cancer. However, it is also carcinogenic in human uterus and rat liver. Tm-inducible Cre-loxP systems are being used in broad areas of research and are providing important biologic insights in tissue development, maintenance, and function. This compound-induced nuclear localization of Cre recombinase is time- and dose-dependent. Higher doses of this chemical induce recombination weeks following administration and lower doses of it induce recombination up to one week following administration. Duration of its-induced gene recombination is also dose-dependent. Administration of high Tm doses leads to extended CreER nuclear localization. Tm treatment induces side effects that may have physiologic consequences in Tm-inducible models. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | p-AKT / AKT ER-α66 / ER-α36 / HER2 / EGFR p-AMPK / AMPK / p-ACC / ACC / p-P70S6K / p-S6 / p-4EBP1 CD36 / p-ERK / ERK |
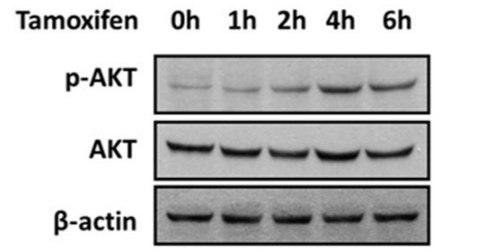
|
30759864 |
| Immunofluorescence | CD36 |
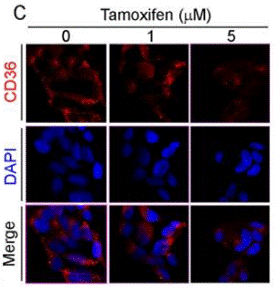
|
30573731 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06154590 | Not yet recruiting | Metastatic Breast Cancer|Tumor|Muscle Neoplasms|Chronic Pain |
DR. DIANE CHISESI NFS. MD. PHD.|IRB|Paradyne Networks A Foundation |
July 2024 | -- |
| NCT05156892 | Recruiting | Ovarian Cancer |
Anthony Joshua FRACP|Royal Prince Alfred Hospital Sydney Australia|Concord Hospital|Prince of Wales Hospital Sydney|St Vincent''s Hospital Sydney |
September 4 2022 | Phase 1 |
| NCT04200066 | Withdrawn | Glioblastoma|Brain Tumor |
University of Rochester |
June 1 2022 | Phase 1 |
| NCT05133674 | Unknown status | Breast Cancer|Breast Carcinoma|Breast Tumors|Cancer of Breast|Malignant Neoplasm of Breast |
Karolinska University Hospital |
April 4 2022 | Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
Frequently Asked Questions
Question 1:
Could you tell me if you have a protocol for its use in vivo in mouse? Is there an administration way that is better than another? This refers to the compound (S1238).
Answer:
It (S1238) can be dissolved in 10% ethanol/10% Tween 80/ddH2O at 5 mg/ml clearly. But precipitation will form if the formulation was left at RT for an hour or longer. So we'd suggest that you use it quickly after formulation. This compound is often administrated via i.p injection.






































