research use only
Semaxanib (SU5416) VEGFR2 inhibitor
Cat.No.S2845
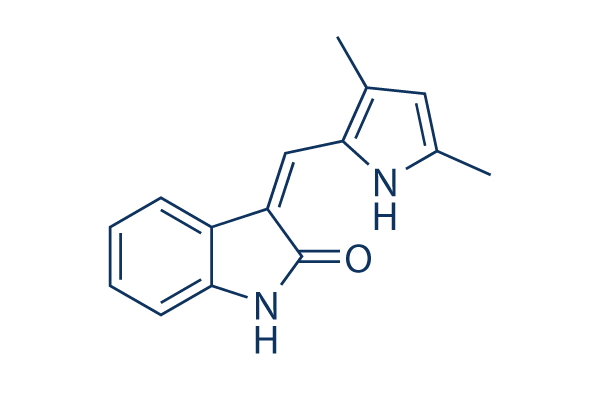
Chemical Structure
Molecular Weight: 238.28
Quality Control
| Related Targets | EGFR PDGFR FGFR c-Met Src MEK CSF-1R FLT3 HER2 c-Kit |
|---|---|
| Other VEGFR Inhibitors | SAR131675 SU 5402 Cediranib (AZD2171) Vatalanib (PTK787) 2HCl Anlotinib (AL3818) Dihydrochloride Linifanib (ABT-869) Apatinib (YN968D1) Apatinib (YN968D1) mesylate Ki8751 ZM 323881 HCl |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| MCF7 cells | Cytotoxicity assay | 48 h | Cytotoxicity against human MCF7 cells after 48 hrs by CCK8 assay, IC50=3.1 nM | |||
| mouse B16F10 cells | Cytotoxicity assay | 48 h | Cytotoxicity against mouse B16F10 cells after 48 hrs by CCK8 assay, IC50=3.6 nM | |||
| human U251 cells | Function assay | Inhibition of VEGFR2 in human U251 cells by phosphotyrosine ELISA, IC50=12.9 nM | ||||
| human A431 cells | Function assay | Antiangiogenic activity against human A431 cells, IC50=0.085 μM | ||||
| CHO cells | Function assay | Inhibition of VEGFR induced autophosphorylation of human Vascular endothelial growth factor receptor 2 (VEGFR2) transfected in CHO cells, IC50=0.884 μM | ||||
| human MCF7 cells | Proliferation assay | 4 days | Antiproliferative activity against human MCF7 cells after 4 days by coulter counter method, IC50=1.9 μM | |||
| human SF539 cells | Function assay | Inhibition of PDGFRbeta in human SF539 cells by phosphotyrosine ELISA, IC50=2.4 μM | ||||
| A431 cells | Function assay | Inhibition of VEGFR2 expressed in human A431 cells, IC50=12.9 μM | ||||
| HUVEC cells | Proliferation assay | 72 h | Antiproliferative activity against human HUVEC cells assessed as reduction in cell viability after 72 hrs by trypan blue assay, GI50=13.6 μM | |||
| human HMEC1 cells | Proliferation assay | 7 days | Antiproliferative activity against human HMEC1 cells after 7 days by coulter counter method, IC50=15 μM | |||
| human HeLa cells | Proliferation assay | 4 days | Antiproliferative activity against human HeLa cells after 4 days by coulter counter method, IC50=20 μM | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 17 mg/mL
(71.34 mM)
Ethanol : 2 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 238.28 | Formula | C15H14N2O |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 204005-46-9 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | semaxinib | Smiles | CC1=CC(=C(N1)C=C2C3=CC=CC=C3NC2=O)C | ||
Mechanism of Action
| Targets/IC50/Ki |
VEGFR2/Flk1
(Cell-free assay) 1.23 μM
|
|---|---|
| In vitro |
Semaxanib (SU5416) inhibits VEGF-dependent phosphorylation of the Flk-1 receptor in Flk-1-overexpressing NIH 3T3 cells with IC50 of 1.04 μM, and PDGF-dependent autophosphorylation in NIH 3T3 cells with IC50 of 20.3 μM. It also inhibits VEGF- and FGF-driven mitogenesis in a dose-dependent manner with IC50 of 0.04 and 50 μM, respectively. Treatment with this compound has no effect on the in vitro growth of C6 glioma, Calu 6 lung carcinoma, A375 melanoma, A431 epidermoid carcinoma, and SF767T glioma cells (all IC50s > 20 μM).
|
| Kinase Assay |
Biochemical kinase assays
|
|
Solubilized membranes from 3T3 Flk-1 cells are added to polystyrene ELISA plates that had been precoated with a monoclonal antibody that recognizes Flk-1. After an overnight incubation with lysate at 4 ℃, serial dilutions of Semaxanib (SU5416) are added to the immunolocalized receptor. To induce autophosphorylation of the receptor, various concentrations of ATP are added to the ELISA plate wells containing serially diluted solutions of this compound. The autophosphorylation is allowed to proceed for 60 min at room temperature and then stopped with EDTA. The amount of phosphotyrosine present on the Flk-1 receptors in the individual wells is determined by incubating the immunolocalized receptor with a biotinylated monoclonal antibody directed against phosphotyrosine. After removal of the unbound anti-phosphotyrosine antibody, avidin-conjugated horseradish pero-idase H is added to the wells. A stabilized form of 3,3 9,5,5 9-tetramethyl benzidine dihydrochloride and H2O2 is added to the wells. The color readout of the assay is allowed to develop for 30 min, and the reaction is stopped with H2SO4.
|
|
| In vivo |
Semaxanib (SU5416) dose-related inhibits growth of A375 tumor in vivo. A >85% inhibition of subcutaneous tumor growth is observed with daily i.p. administration of this compound in DMSO, without measurable toxicity. It shows broad spectrum antitumor activity, significantly inhibiting the subcutaneous growth of 8 of 10 tumor lines tested (A431, Calu-6, C6, LNCAP, EPH4-VEGF, 3T3HER2, 488G2M2 and SF763T cells) with an average mortality rate of 2.5%. At 25 mg/kg/day, it displays potent antiangiogenic activity, resulting in a significant reduction of both the total and functional vascular density of the tumor microvasculature.
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
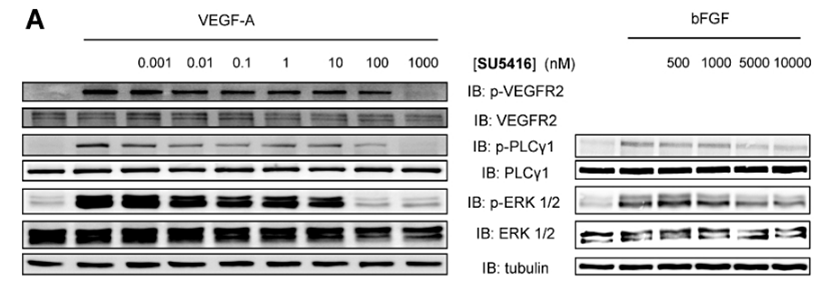
| Western blot | p-VEGFR2 / VEGFR2 / p-PLCγ1 / PLCγ1 / p-ERK / ERK |
|
21699503 |
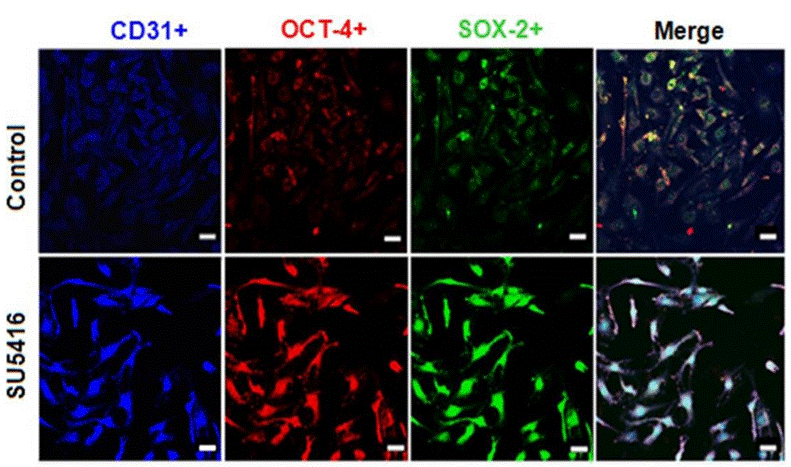
| Immunofluorescence | CD31 / OCT-4 / SOX-2 |
|
25665868 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT00006247 | Terminated | Brain and Central Nervous System Tumors |
Pediatric Brain Tumor Consortium|National Cancer Institute (NCI) |
August 2000 | Phase 1 |
| NCT00005642 | Completed | Unspecified Adult Solid Tumor Protocol Specific |
Case Comprehensive Cancer Center|National Cancer Institute (NCI) |
May 2000 | Phase 1 |
| NCT00005647 | Completed | Head and Neck Cancer |
Case Comprehensive Cancer Center|National Cancer Institute (NCI) |
May 2000 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































