research use only
Cediranib Maleate VEGFR inhibitor
Cat.No.S5242
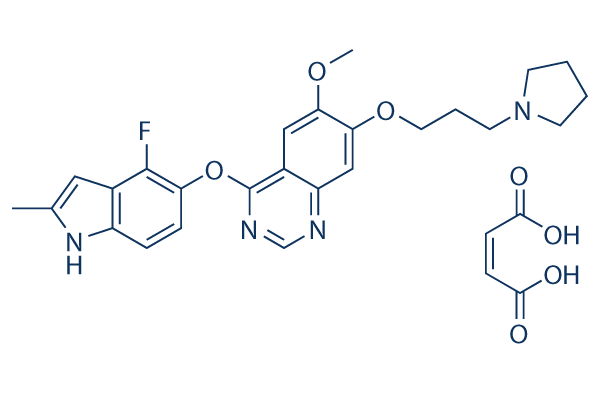
Chemical Structure
Molecular Weight: 566.58
Quality Control
| Related Targets | EGFR PDGFR FGFR c-Met Src MEK CSF-1R FLT3 HER2 c-Kit |
|---|---|
| Other VEGFR Inhibitors | SAR131675 SU 5402 Cediranib (AZD2171) Vatalanib (PTK787) 2HCl Anlotinib (AL3818) Dihydrochloride Linifanib (ABT-869) Apatinib (YN968D1) Apatinib (YN968D1) mesylate Semaxanib (SU5416) Ki8751 |
Solubility
|
In vitro |
DMSO
: 100 mg/mL
(176.49 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 566.58 | Formula | C25H27FN4O3.C4H4O4 |
Storage (From the date of receipt) | 3 years -20°C powder |
|---|---|---|---|---|---|
| CAS No. | 857036-77-2 | -- | Storage of Stock Solutions |
|
|
| Synonyms | AZD2171 maleate | Smiles | CC1=CC2=C(N1)C=CC(=C2F)OC3=NC=NC4=CC(=C(C=C43)OC)OCCCN5CCCC5.C(=CC(=O)O)C(=O)O | ||
Mechanism of Action
| Targets/IC50/Ki |
VEGFR2/KDR
(HUVECs) 0.5 nM
c-Kit
(HUVECs) 2 nM
c-Kit
(HUVECs) 2 nM
VEGFR3/FLT4
(HUVECs) <=3 nM
VEGFR1/FLT1
(HUVECs) 5 nM
PDGFRβ
(HUVECs) 5 nM
FGFR1
(HUVECs) 26 nM
FGFR1
(HUVECs) 26 nM
PDGFRα
(HUVECs) 36 nM
|
|---|---|
| In vitro |
In recombinant kinase assays, cediranib inhibits VEGFR-1 kinase activity (IC50 = 5 nmol/L) within a similar concentration range to VEGFR-2 and VEGFR-3 (IC50 values of <0.1 and ≤3 nmol/L). It shows selectivity against other kinases. Cediranib has similar potency against c-Kit when compared with VEGFR-2 in phosphorylation assays but less potency against PDGFR-α and PDGFR-β, particularly in a PDGF-AA/PDGFR-α–driven tumor cell proliferation assay. Cediranib is inactive against wild-type Flt-3 (IC50 value >1 μmol/L) and had marginal activity versus FGFR-1 and -4 (IC50 values of 0.35 and 2.17 μmol/L, respectively). |
| In vivo |
Phosphorylation of wild-type c-Kit in NCI-H526 tumor xenografts was reduced markedly following oral administration of cediranib (≥1.5 mg/kg/d) to tumor-bearing nude mice. |
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT00458731 | Completed | Adult Grade III Lymphomatoid Granulomatosis|Adult Nasal Type Extranodal NK/T-cell Lymphoma|Anaplastic Large Cell Lymphoma|Angioimmunoblastic T-cell Lymphoma|Childhood Burkitt Lymphoma|Childhood Diffuse Large Cell Lymphoma|Childhood Grade III Lymphomatoid Granulomatosis|Childhood Immunoblastic Large Cell Lymphoma|Childhood Nasal Type Extranodal NK/T-cell Lymphoma|Cutaneous B-cell Non-Hodgkin Lymphoma|Extranodal Marginal Zone B-cell Lymphoma of Mucosa-associated Lymphoid Tissue|Hepatosplenic T-cell Lymphoma|Intraocular Lymphoma|Nodal Marginal Zone B-cell Lymphoma|Noncutaneous Extranodal Lymphoma|Peripheral T-cell Lymphoma|Progressive Hairy Cell Leukemia Initial Treatment|Recurrent Adult Burkitt Lymphoma|Recurrent Adult Diffuse Large Cell Lymphoma|Recurrent Adult Diffuse Mixed Cell Lymphoma|Recurrent Adult Diffuse Small Cleaved Cell Lymphoma|Recurrent Adult Hodgkin Lymphoma|Recurrent Adult Immunoblastic Large Cell Lymphoma|Recurrent Adult Lymphoblastic Lymphoma|Recurrent Adult T-cell Leukemia/Lymphoma|Recurrent Childhood Anaplastic Large Cell Lymphoma|Recurrent Childhood Large Cell Lymphoma|Recurrent Childhood Lymphoblastic Lymphoma|Recurrent Childhood Small Noncleaved Cell Lymphoma|Recurrent Grade 1 Follicular Lymphoma|Recurrent Grade 2 Follicular Lymphoma|Recurrent Grade 3 Follicular Lymphoma|Recurrent Mantle Cell Lymphoma|Recurrent Mycosis Fungoides/Sezary Syndrome|Recurrent/Refractory Childhood Hodgkin Lymphoma|Refractory Hairy Cell Leukemia|Small Intestine Lymphoma|Splenic Marginal Zone Lymphoma|Stage IV Adult Burkitt Lymphoma|Stage IV Adult Diffuse Large Cell Lymphoma|Stage IV Adult Diffuse Mixed Cell Lymphoma|Stage IV Adult Diffuse Small Cleaved Cell Lymphoma|Stage IV Adult Hodgkin Lymphoma|Stage IV Adult Immunoblastic Large Cell Lymphoma|Stage IV Adult Lymphoblastic Lymphoma|Stage IV Adult T-cell Leukemia/Lymphoma|Stage IV Childhood Anaplastic Large Cell Lymphoma|Stage IV Childhood Hodgkin Lymphoma|Stage IV Childhood Large Cell Lymphoma|Stage IV Childhood Lymphoblastic Lymphoma|Stage IV Childhood Small Noncleaved Cell Lymphoma|Stage IV Grade 1 Follicular Lymphoma|Stage IV Grade 2 Follicular Lymphoma|Stage IV Grade 3 Follicular Lymphoma|Stage IV Mantle Cell Lymphoma|Stage IVA Mycosis Fungoides/Sezary Syndrome|Stage IVB Mycosis Fungoides/Sezary Syndrome|T-cell Large Granular Lymphocyte Leukemia|Testicular Lymphoma|Unspecified Adult Solid Tumor Protocol Specific|Unspecified Childhood Solid Tumor Protocol Specific|Waldenström Macroglobulinemia |
National Cancer Institute (NCI) |
May 2007 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































