research use only
Apatinib (YN968D1) mesylate VEGFR inhibitor
Cat.No.S2221
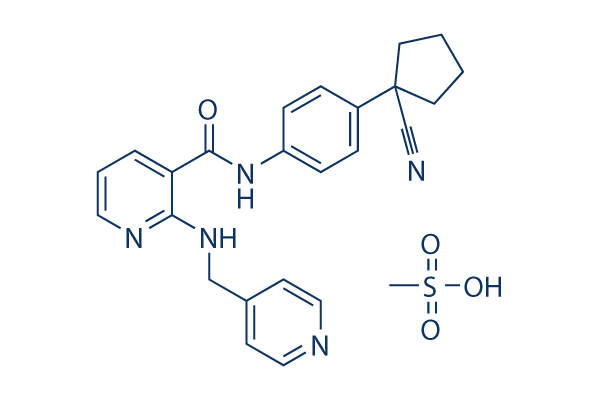
Chemical Structure
Molecular Weight: 493.58
Quality Control
| Related Targets | EGFR PDGFR FGFR c-Met Src MEK CSF-1R FLT3 HER2 c-Kit |
|---|---|
| Other VEGFR Inhibitors | SAR131675 SU 5402 Cediranib (AZD2171) Vatalanib (PTK787) 2HCl Anlotinib (AL3818) Dihydrochloride Linifanib (ABT-869) Apatinib (YN968D1) Semaxanib (SU5416) Ki8751 ZM 323881 HCl |
Solubility
|
In vitro |
DMSO
: 99 mg/mL
(200.57 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 493.58 | Formula | C25H27N5O4S |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1218779-75-9 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | YN968D1, Rivoceranib | Smiles | C[S](O)(=O)=O.O=C(NC1=CC=C(C=C1)C2(CCCC2)C#N)C3=CC=CN=C3NCC4=CC=NC=C4 | ||
Mechanism of Action
| Features |
Good anti-tumor effects for gastric and colorectal cancer compared with NSC-724772 and sunitinib.
|
|---|---|
| Targets/IC50/Ki |
VEGFR2
(Cell-free assay) 1 nM
RET
(Cell-free assay) 13 nM
|
| In vitro |
Apatinib (YN968D1) is a novel, orally bioavailable, selective inhibitor with potential antiangiogenic and antineoplastic activities. Apatinib selectively binds to and inhibits VEGFR2. Apatinib can also potently suppress the activities of Ret, c-kit and c-src with IC50 of 0.013 μM, 0.429 μM and 0.53 μM, respectively. Apatinib inhibits cellular phosphorylation of VEGFR-2, c-kit and PDGFRβ. Apatinib significantly inhibits proliferation stimulated by 20 ng/mL VEGF (IC50 = 0.17μM). Apatinib effectively inhibits proliferation, migration and tube formation of human umbilical vein endothelial cells induced by FBS, and blocked the budding of rat aortic ring. Apatinib reverses ABCB1- and ABCG2-mediated MDR by inhibiting their transport function, but not by blocking the AKT or ERK1/2 pathway or downregulating ABCB1 or ABCG2 expression. Apatinib significantly potentiates the cytotoxicity of established ABCB1 and ABCG2 substrates and increased the accumulation of DOX and Rho 123 in ABCB1- or ABCG2-overexpressing cells. Furthermore, apatinib significantly inhibited the photoaffinity labeling of both ABCB1 and ABCG2 with [125I]iodoarylazidoprazosin in a concentration-dependent manner. |
| Kinase Assay |
Enzyme-linked immunosorbent assay
|
|
A poly(glu, ala, tyr) 6:3:1 random copolymer is used as a tyrosine containing substrate solution. The substrate is stored as a 1 mg/mL stock in PBS at −20 °C and diluted 1 in 500 with PBS in order to coat 96 well plates (100 μL/well). Plates are coated on the day prior to assay, sealed with adhesive seals, and stored overnight at 4 °C. On the day of the assay, the substrate solution is discarded and the assay plate wells are washed once with PBST (PBS containing 0.05% v/v Tween 20) and once with Hepes buffer (50 mM, pH 7.4).Test compounds are diluted with 10% DMSO de-ionized water and 25 μL volumes transferred to wells in the washed assay plates. Manganese chloride solution (40 mM) containing 8 μM ATP is then added (25 μL) to all test wells. Control and blank wells, containing compound diluent and manganese chloride solution with and without ATP, respectively, are also included to determine the dynamic range of the assay. Freshly diluted enzyme (50 μL) is added to each well, and the plates incubated at room temperature for 20 min. The liquid is then discarded and the wells are washed twice with PBST. Mouse IgG anti-phosphotyrosine antibody diluted 1:6000 with PBST containing 0.5% (w/v) bovine serum albumin (BSA) is added (100 μL/well), and the plates incubated for 1h at room temperature before discarding the liquid and washing the wells twice with PBST. Horseradish peroxidase (HRP)-linked sheep anti-mouse Ig antibody diluted 1:500 with PBST containing 0.5% (w/v) BSA, is then added (100 μL/well) and the plates incubated for a further 1 h at room temperature before discarding the liquid and washing the wells twice with PBST. A 1 mg/mL solution of 2,2‘-azino-bis(3-ethylbenzthiazoline-6-sulfonic acid is freshly prepared in 50 mM phosphate-citrate buffer (pH5.0) containing 0.03% (w/v) sodium perborate, and 100 μL added to each well. Plates are then incubated for 20−60 min at room temperature until the optical density value of control wells measured at 405 nm is approximately 1.0. IC50 values for compound enzyme inhibition are interpolated using Microcal Origin following subtraction of blank values.
|
|
| In vivo |
Apatinib inhibits the growth of a broad range of human tumor xenografts in a significant dose-dependent manner. Apatinib reverses ABCB1-mediated MDR in the nude mouse xenograft model. Apatinib significantly enhances the antitumor activity of doxorubicin in nude mice bearing K562/ADR xenografts. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | p-VEGFR2 / VEGFR2 / p-ERK / ERK PI3K / p-PI3K / mTOR / p-mTOR / AKT / p-AKT Beclin 1 / Atg7 / p62 / LC3-I / LC3-II |
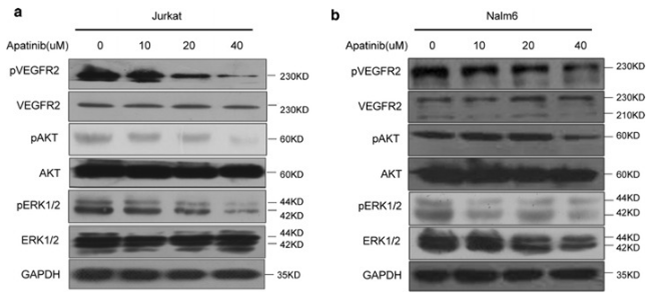
|
29490645 |
| Growth inhibition assay | Cell viability |
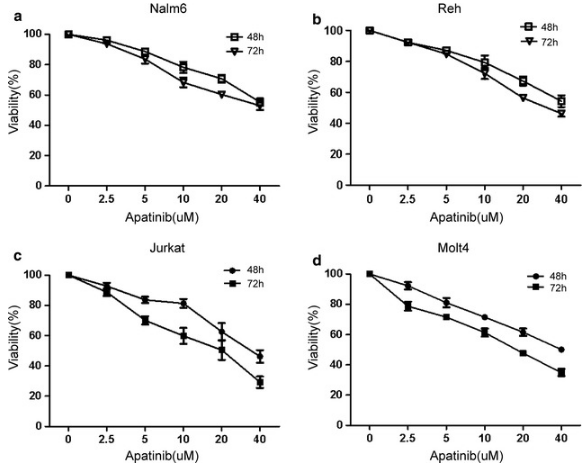
|
29490645 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05839197 | Recruiting | Macrotrabecular Massive Hepatocellular Carcinoma |
Wan-Guang Zhang|Tongji Hospital |
May 5 2023 | Phase 2 |
| NCT05742750 | Not yet recruiting | Locally Advanced Biliary Tract Cancer|Metastatic Biliary Tract Cancer |
Sun Yat-sen University|Jiangsu Hengrui Pharmaceutical Co. Ltd. |
March 1 2023 | Phase 1|Phase 2 |
| NCT05287360 | Completed | Healthy |
Elevar Therapeutics |
December 30 2021 | Phase 1 |
| NCT03743428 | Suspended | Colorectal Neoplasms |
Shenzhen People''s Hospital |
October 22 2020 | Not Applicable |
| NCT04517357 | Unknown status | Relapsed Ovarian Cancer |
Jiangsu HengRui Medicine Co. Ltd. |
October 16 2020 | Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
Frequently Asked Questions
Question 1:
How to reconstitute it for in vivo studies?
Answer:
We suggest the vehicle 0.5% CMC. In this vehicle, it is not fully dissolved. However, the mixture is a stable suspension and can be used for oral gavage feeding.






































