research use only
Triciribine (API-2) Akt inhibitor
Cat.No.S1117
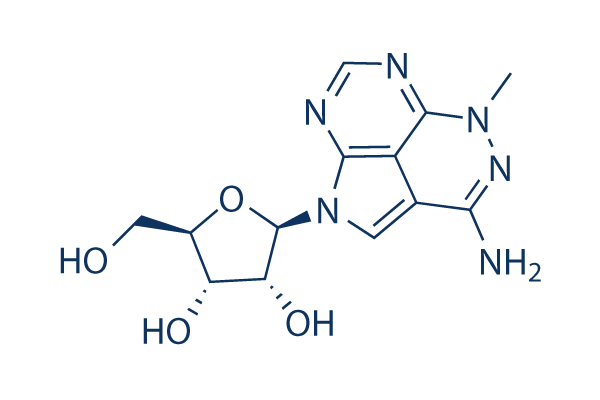
Chemical Structure
Molecular Weight: 320.3
Quality Control
| Related Targets | PI3K mTOR GSK-3 ATM/ATR DNA-PK AMPK PDPK1 PTEN PP2A PDK |
|---|---|
| Other Akt Inhibitors | SC79 AZD5363 (Capivasertib) MK-2206 Dihydrochloride Ipatasertib (GDC-0068) Perifosine GSK690693 Afuresertib (GSK2110183) CCT128930 AKTi-1/2 (AKT Inhibitor VIII) A-674563 HCl |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| L1210 | Function assay | Tested in vitro for cytotoxicity against murine L1210 leukemic cells, IC50=0.035μM | 10882371 | |||
| HFF | Function assay | HCMV plaque assay was performed using HFF cells and effect was calculated as a percentage of reduction in number of plaques, IC50=2.5μM | 10882371 | |||
| BSC-1 | Antiviral assay | Antiviral activity was tested using an enzyme-linked immunosorbent assay (ELISA) to detect HSV-1 (herpes simplex virus type 1) using BSC-1 cells, IC50=23μM | 10882371 | |||
| HFF | Antiviral assay | Antiviral activity against HCMV was determined by plaque reduction assay using HFF cells, IC50=2.5μM | 10882373 | |||
| AA2 | Function assay | 5 hrs | Intracellular phosphorylation (100 uM) in uninfected AA2 cells was studied after 5 hrs of Incubation., Concentration=9μM | 10882373 | ||
| BSC-1 | Antiviral assay | Antiviral activity against HSV-1 was determined using BSC-1 cells by an enzyme-linked immunosorbent assay (ELISA), IC50=23μM | 10882373 | |||
| Huh-7 | Function assay | Compound was tested for its ability to inhibit hepatitis C viral RNA replication in Huh-7 cells (human hepatoma cells), EC50=2μM | 15177464 | |||
| MCF7 | Cytotoxicity assay | Cytotoxicity against human MCF7 cells in presence of 20 uM chloroquine by SRB assay, GI50=0.05μM | 18691894 | |||
| MCF7 | Cytotoxicity assay | Cytotoxicity against human MCF7 cells in presence of 10 uM chloroquine by SRB assay, GI50=0.56μM | 18691894 | |||
| MDA-MB-231 | Cytotoxicity assay | Cytotoxicity against human MDA-MB-231 cells in presence of 20 uM chloroquine by SRB assay, GI50=0.69μM | 18691894 | |||
| MCF7 | Cytotoxicity assay | Cytotoxicity against human MCF7 cells by SRB assay, GI50=3.64μM | 18691894 | |||
| MDA-MB-468 | Cytotoxicity assay | Cytotoxicity against human MDA-MB-468 cells in presence of 20 uM chloroquine by SRB assay, GI50=10.29μM | 18691894 | |||
| 184B5 | Cytotoxicity assay | 20 uM | Cytotoxicity against human 184B5 cells at 20 uM chloroquine by SRB assay, GI50=16.96μM | 18691894 | ||
| MDA-MB-468 | Cytotoxicity assay | 10 uM | Cytotoxicity against human MDA-MB-468 cells in presence of 10 uM chloroquine by SRB assay, GI50=20.45μM | 18691894 | ||
| 184B5 | Cytotoxicity assay | 10 uM | Cytotoxicity against human 184B5 cells at 10 uM chloroquine by SRB assay, GI50=34μM | 18691894 | ||
| MDA-MB-231 | Cytotoxicity assay | 10 uM | Cytotoxicity against human MDA-MB-231 cells in presence of 10 uM chloroquine by SRB assay, GI50=37μM | 18691894 | ||
| 184B5 | Cytotoxicity assay | Cytotoxicity against human 184B5 cells by SRB assay, GI50=40μM | 18691894 | |||
| MDA-MB-468 | Cytotoxicity assay | Cytotoxicity against human MDA-MB-468 cells by SRB assay, GI50=43.53μM | 18691894 | |||
| MDM | Antiviral assay | 6 days | Antiviral activity against Human immunodeficiency virus 1 ADA infected in human MDM cells assessed as expression of p24 antigen after 6 days postinfection by ELISA, IC50=0.006μM | 20086149 | ||
| ACH2 | Cytotoxicity assay | Cytotoxicity against human ACH2 cells infected with latent Human immunodeficiency virus 1 by MTS assay, CC50=0.01μM | 20086149 | |||
| ACH2 | Cytotoxicity assay | Cytotoxicity against human ACH2 cells infected with latent Human immunodeficiency virus 1 by MTS assay in presence of tumor necrosis factor alpha, CC50=0.01μM | 20086149 | |||
| CEM-SS | Antiviral assay | 6 days | Antiviral activity against Human immunodeficiency virus 1 3B infected in human CEM-SS cells infected with 0.78 uL of virus stock assessed as expression of p24 antigen after 6 days postinfection by ELISA, IC50<0.01μM | 20086149 | ||
| MDM | Antiviral assay | 6 days | Antiviral activity against Human immunodeficiency virus 1 BAL infected in human MDM cells assessed as expression of p24 antigen after 6 days postinfection by ELISA, IC50=0.018μM | 20086149 | ||
| H9 | Antiviral assay | Antiviral activity against Human immunodeficiency virus 1 SK1 infected in human H9 cells assessed as inhibition of viral Reverse transcriptase by [3H]TTP incorporation assay, IC50=0.036μM | 20086149 | |||
| CEM-SS | Antiviral assay | Antiviral activity against Human immunodeficiency virus 1 3B expressing nef protein infecting in CEM-SS cells assessed as inhibition of viral Reverse transcriptase by [3H]TTP incorporation assay, IC50=0.04μM | 20086149 | |||
| CEM-SS | Antiviral assay | Antiviral activity against Human immunodeficiency virus 1 3B infected in human CEM-SS cells infected with 0.78 uL of virus stock assessed as inhibition of viral Reverse transcriptase by [3H]TTP incorporation assay, IC50=0.08μM | 20086149 | |||
| H9 | Antiviral assay | Antiviral activity against Human immunodeficiency virus 1 3B infected in human H9 cells infected with 25 uL of virus stock assessed as inhibition of viral Reverse transcriptase by [3H]TTP incorporation assay, IC50<0.1μM | 20086149 | |||
| H9 | Antiviral assay | Antiviral activity against Human immunodeficiency virus 1 3B infected in human H9 cells infected with 12.5 uL of virus stock assessed as inhibition of viral Reverse transcriptase by [3H]TTP incorporation assay, IC50<0.1μM | 20086149 | |||
| H9 | Antiviral assay | Antiviral activity against Human immunodeficiency virus 1 3B infected in human H9 cells infected with 6.25 uL of virus stock assessed as inhibition of viral Reverse transcriptase by [3H]TTP incorporation assay, IC50<0.1μM | 20086149 | |||
| H9 | Antiviral assay | 6 days | Antiviral activity against Human immunodeficiency virus 1 3B infected in human H9 cells infected with 12.5 uL of virus stock assessed as expression of p24 antigen after 6 days postinfection by ELISA, IC50<0.1μM | 20086149 | ||
| H9 | Antiviral assay | 6 days | Antiviral activity against Human immunodeficiency virus 1 3B infected in human H9 cells infected with 6.25 uL of virus stock assessed as expression of p24 antigen after 6 days postinfection by ELISA, IC50<0.1μM | 20086149 | ||
| CEM-SS | Antiviral assay | Antiviral activity against Human immunodeficiency virus 1 SK1 infected in human CEM-SS cells assessed as inhibition of viral Reverse transcriptase by [3H]TTP incorporation assay, IC50=0.13μM | 20086149 | |||
| ACH2 | Antiviral assay | 3 days | Antiviral activity against 5 x 10'3 cells/well Human immunodeficiency virus 1 infected in human ACH2 cells assessed as inhibition of viral Reverse transcriptase after 3 days by [3H]TTP incorporation assay in presence of 5 ng/ml tumor necrosis factor alpha, IC50=0.15μM | 20086149 | ||
| AA5 | Antiviral assay | Antiviral activity against of Human immunodeficiency virus 1 3B infected in AA5 cells infected with 3.13 uL of virus stock assessed as inhibition of viral Reverse transcriptase by [3H]TTP incorporation assay, IC50<0.17μM | 20086149 | |||
| AA5 | Antiviral assay | Antiviral activity against of Human immunodeficiency virus 1 3B infected in AA5 cells infected with 1.56 uL of virus stock assessed as inhibition of viral Reverse transcriptase by [3H]TTP incorporation assay, IC50<0.17μM | 20086149 | |||
| AA5 | Antiviral assay | Antiviral activity against of Human immunodeficiency virus 1 3B infected in AA5 cells infected with 0.78 uL of virus stock assessed as inhibition of viral Reverse transcriptase by [3H]TTP incorporation assay, IC50<0.17μM | 20086149 | |||
| CEM-SS | Antiviral assay | 6 days | Antiviral activity against Human immunodeficiency virus 1 3B infected in human CEM-SS cells infected with 1.56 uL of virus stock assessed as expression of p24 antigen after 6 days postinfection by ELISA, IC50=0.19μM | 20086149 | ||
| CEM-SS | Antiviral assay | Antiviral activity against Human immunodeficiency virus 1 harboring plasmid NL4-3 infected in CEM-SS cells assessed as inhibition of viral Reverse transcriptase by [3H]TTP incorporation assay, IC50=0.2μM | 20086149 | |||
| U1 | Antiviral assay | Antiviral activity against Human immunodeficiency virus 1 infected in human U1 cells assessed as inhibition of viral Reverse transcriptase by [3H]TTP incorporation assay, IC50=0.23μM | 20086149 | |||
| CEM-SS | Antiviral assay | 6 days | Antiviral activity against Human immunodeficiency virus 1 3B infected in human CEM-SS cells infected with 3.13 uL of virus stock assessed as expression of p24 antigen after 6 days postinfection by ELISA, IC50=0.24μM | 20086149 | ||
| U1 | Cytotoxicity assay | Cytotoxicity against human U1 cells infected with latent Human immunodeficiency virus 1 by MTS assay, CC50=0.28μM | 20086149 | |||
| U1 | Cytotoxicity assay | Cytotoxicity against human U1 cells infected with latent Human immunodeficiency virus 1 by MTS assay in presence of tumor necrosis factor alpha, CC50=0.28μM | 20086149 | |||
| CEM-SS | Antiviral assay | Antiviral activity against Human immunodeficiency virus 1 3B infected in human CEM-SS cells infected with 1.56 uL of virus stock assessed as inhibition of viral Reverse transcriptase by [3H]TTP incorporation assay, IC50=0.29μM | 20086149 | |||
| CEM-SS | Antiviral assay | Antiviral activity against Human immunodeficiency virus 1 3B infected in human CEM-SS cells infected with 3.13 uL of virus stock assessed as inhibition of viral Reverse transcriptase by [3H]TTP incorporation assay, IC50=0.3μM | 20086149 | |||
| CEM-SS | Antiviral assay | Antiviral activity against Human immunodeficiency virus 1 D1 harboring Tyr127His mutation in nef protein infecting in CEM-SS cells assessed as inhibition of viral Reverse transcriptase by [3H]TTP incorporation assay, IC50=0.33μM | 20086149 | |||
| CEM-SS | Antiviral assay | Antiviral activity against Human immunodeficiency virus 1 A7 harboring Tyr127His mutation in nef protein infecting in CEM-SS cells assessed as inhibition of viral Reverse transcriptase by [3H]TTP incorporation assay, IC50=0.5μM | 20086149 | |||
| MDM | Cytotoxicity assay | Cytotoxicity against human MDM cells infected with Human immunodeficiency virus 1 BAL by MTS assay, TC50=0.66μM | 20086149 | |||
| CEM-SS | Antiviral assay | Antiviral activity against Human immunodeficiency virus 2 ROD infected in human CEM-SS cells assessed as inhibition of viral Reverse transcriptase by [3H]TTP incorporation assay, IC50=1.4μM | 20086149 | |||
| H9 | Antiviral assay | Antiviral activity against Human immunodeficiency virus 1 3B infected in human H9 cells infected with 50 uL of virus stock assessed as inhibition of viral Reverse transcriptase by [3H]TTP incorporation assay, IC50=1.53μM | 20086149 | |||
| H9 | Cytotoxicity assay | Cytotoxicity against human H9 cells by MTS assay, IC50=16.3μM | 20086149 | |||
| H9 | Cytotoxicity assay | Cytotoxicity against human H9 cells by MTS assay, CC50=19.6μM | 20086149 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 64 mg/mL
(199.81 mM)
Ethanol : 16 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 320.3 | Formula | C13H16N6O4 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 35943-35-2 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | NSC 154020, VD-0002, vqd-002, TCN | Smiles | CN1C2=NC=NC3=C2C(=CN3C4C(C(C(O4)CO)O)O)C(=N1)N | ||
Mechanism of Action
| Targets/IC50/Ki |
HIV-1
(CEM-SS, H9, H9IIIB, U1 cells) 20 nM
Akt
(PC3 cells) 130 nM
|
|---|---|
| In vitro |
Triciribine (API-2) exhibits maximum growth inhibition around 1-10 μM and inhibits phosphorylation of Akt, as well as downstream p70S6K, to basal levels at 100μM (IC50 = 130 nM). It shows particular promise for inhibiting growth in Nf1 and Trp53 mutant astrocytoma cells in a grade-dependent manner. The WHO II K1861-10 line is inhibited, incompletely (69% maximum inhibition), with a GI50 value of 1.7 μM for this compound, whereas higher-grade tumor lines (KR158, KR130, and SF295) are inhibited to a greater extent (>80% maximum inhibition) at lower GI50 values (0.4–1.1 mM). Importantly, it is much less effective at inhibiting primary astrocytes (GI5013.6 mM), suggesting that this inhibitor may show specificity for tumor cells. It inihibits HIV-1with an IC50 of 20 nM. Greater than 90% inhibition is achieved at 0.1μM and complete inhibition of syncytia formation is achieved at 5μM. Associated cell toxicity in the same cell line for Triciribine is 46 μM, resulting in selectivity indices of 2250. It markedly inhibits HIV-1-induced p24 core antigen production, reverse transcriptase, and infectious virus production in a dose-dependent manner using HIV-1 acutedly infected CEM-SS, H9, and persistently infected H9III B and U1 cells. This compound inhibits Akt phosphorylation at Thr308 and Ser473 and Akt activity in the human prostate cancer cell line PC-3. It sensitizes PC-3 cells to TRAIL- and anti-CD95-induced apoptosis, whereas the cells remain resistant to DNA damaging chemotherapeutics. It is highly selective for Akt and does not inhibit the activation of phosphatidylinositol 3-kinase, phosphoinositide-dependent kinase-1, protein kinase C, serum and glucocorticoid-inducible kinase, protein kinase A, signal transducer and activators of transcription 3, extracellular signal-regulated kinase-1/2, or c-Jun NH2-terminal kinase.
|
| Kinase Assay |
Akt Phosphorylation Changes Assay
|
|
Cells are grown to 80%–90% confluency and stimulated for 5–10 minutes with 1–10 ng/mL of epidermal growth factor or platelet derived growth factor (PDGF)–AA with or without 10–20 mM of U0126 or LY-294002. Protein lysates (5–20 μg) are separated by 12%–15% SDS PAGE and analyzed by Western blot for Akt, phosphorylated Akt (phospho-Ser 473), MAPK, and phosphorylated MAPK (p44/42 phospho-Thr202/Tyr204) antibodies (1:1000).
|
|
| In vivo |
Triciribine (API-2), administered at 1 mg/kg/day i.p., inhibits tumor growth by 90%, 88%, and 80% in nude mice bearing OVCAR3, OVCAR8, and PANC1 tumors, respectively, which overexpress Akt. However, this compound has little effect on the growth of OVCAR5 and COLO357 cells.
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | PUMA / p-FoxO3a / p-AKT |
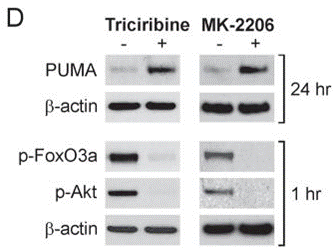
|
20978166 |
| Immunofluorescence | TRF2 / 53BP1 |
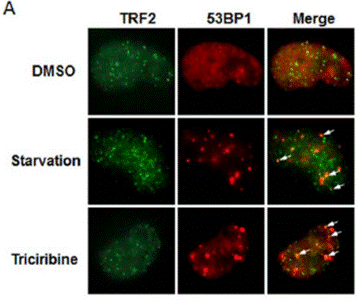
|
23862686 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT02987127 | Unknown status | Mucosa-Associated Lymphoid Tissue Lymphoma |
National Taiwan University Hospital |
February 2016 | -- |
| NCT00642031 | Completed | Hematologic Malignancies|Leukemia |
Prescient Therapeutics Ltd.|VioQuest Pharmaceuticals |
August 2006 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































