research use only
AZD5363 (Capivasertib) Akt inhibitor
Cat.No.S8019
Chemical Structure
Molecular Weight: 428.92
Quality Control
| Related Targets | PI3K mTOR GSK-3 ATM/ATR DNA-PK AMPK PDPK1 PTEN PP2A PDK |
|---|---|
| Other Akt Inhibitors | SC79 MK-2206 Dihydrochloride Ipatasertib (GDC-0068) Perifosine GSK690693 Triciribine (API-2) Afuresertib (GSK2110183) CCT128930 AKTi-1/2 (AKT Inhibitor VIII) A-674563 HCl |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| PC-3 | Function Assay | 10 μM | 12 h | induces autophagy | 23258740 | |
| LNCaP | Cell Viability Assay | 0-1000 nM | 0-4 d | reduced LNCaP cell viability in a dose- and time-dependent manner | 23258740 | |
| DU145 | Function Assay | 0.5/1/10 μM | 48 h | downregulates the phosphorylation of downstream pathway proteins in a dose-dependent manner | 23258740 | |
| PC-3 | Function Assay | 0.5/1/10 μM | 48 h | downregulates the phosphorylation of downstream pathway proteins in a dose-dependent manner | 23258740 | |
| C4-2 | Growth Inhibition Assay | 100-5000 nM | 72 h | increases the fraction of cells undergoing cell death | 23966621 | |
| LNCaP | Growth Inhibition Assay | 100-5000 nM | 72 h | increases the fraction of cells undergoing cell death | 23966621 | |
| C4-2 | Growth Inhibition Assay | 1-10000 nM | 0-3 d | inhibits cell viability dose dependently | 23966621 | |
| LNCaP | Growth Inhibition Assay | 1-10000 nM | 0-3 d | inhibits cell viability dose dependently | 23966621 | |
| C4-2 | Function Assay | 5 μM | 0-24 h | inhibits phosphorylation of the distal AKT-pathway biomarkers including PRAS40, eIF4E, 4E-BP1, mTOR, and P70 S6 kinase in a time-dependent manner | 23966621 | |
| LNCaP | Function Assay | 5 μM | 0-24 h | inhibits phosphorylation of the distal AKT-pathway biomarkers including PRAS40, eIF4E, 4E-BP1, mTOR, and P70 S6 kinase in a time-dependent manner | 23966621 | |
| C4-2 | Function Assay | 5 μM | 0-24 h | induces AKTS473 and AKTT308 phosphorylation in a time dependent manner | 23966621 | |
| LNCaP | Function Assay | 5 μM | 0-24 h | induces AKTS473 and AKTT308 phosphorylation in a time dependent manner | 23966621 | |
| PAMC82 | Growth Inhibition Assay | IC50=30 μM | 24088382 | |||
| MKN74 | Growth Inhibition Assay | IC50=30 μM | 24088382 | |||
| GTL-16 | Growth Inhibition Assay | IC50=30 μM | 24088382 | |||
| SNU-5 | Growth Inhibition Assay | IC50=30 μM | 24088382 | |||
| NUGC-4 | Growth Inhibition Assay | IC50=30 μM | 24088382 | |||
| SNU-216 | Growth Inhibition Assay | IC50=30 μM | 24088382 | |||
| AZ521 | Growth Inhibition Assay | IC50=25.448 μM | 24088382 | |||
| NUGC-3 | Growth Inhibition Assay | IC50=21.873 μM | 24088382 | |||
| OCUM-1 | Growth Inhibition Assay | IC50=14.515 μM | 24088382 | |||
| SNU-16 | Growth Inhibition Assay | IC50=11.097 μM | 24088382 | |||
| SNU-484 | Growth Inhibition Assay | IC50=7.392 μM | 24088382 | |||
| KATO III | Growth Inhibition Assay | IC50=7.267 μM | 24088382 | |||
| HS746T | Growth Inhibition Assay | IC50=6.084 μM | 24088382 | |||
| SNU-668 | Growth Inhibition Assay | IC50=6.003 μM | 24088382 | |||
| SNU-601 | Growth Inhibition Assay | IC50=5.938 μM | 24088382 | |||
| SNU-1 | Growth Inhibition Assay | IC50=5.258 μM | 24088382 | |||
| SNU-638 | Growth Inhibition Assay | IC50=4.523 μM | 24088382 | |||
| SNU-620 | Growth Inhibition Assay | IC50=3.384 μM | 24088382 | |||
| MKN1 | Growth Inhibition Assay | IC50=2.421 μM | 24088382 | |||
| 23132/87 | Growth Inhibition Assay | IC50=1.671 μM | 24088382 | |||
| NCI-N87 | Growth Inhibition Assay | IC50=1.037 μM | 24088382 | |||
| AGS | Growth Inhibition Assay | IC50=0.552 μM | 24088382 | |||
| IM95m | Growth Inhibition Assay | IC50=0.51 μM | 24088382 | |||
| HGC27 | Growth Inhibition Assay | IC50=0.445 μM | 24088382 | |||
| PC-9 | Function Assay | 1/5/10 μM | 4/24 h | increases AKT phosphorylation | 24957682 | |
| NCI-H522 | Function Assay | 1/5/10 μM | 4/24 h | increases AKT phosphorylation | 24957682 | |
| PC-9 | Growth Inhibition Assay | IC50=9.3 (±1.2) μM | 24957682 | |||
| NCI-H522 | Growth Inhibition Assay | IC50=11.3 (±2.7) μM | 24957682 | |||
| MR49F | Growth Inhibition Assay | 0-5 μM | 48 h | inhibits cell growth in a dose dependent manner | 25151012 | |
| MR49C | Growth Inhibition Assay | 0-5 μM | 48 h | inhibits cell growth in a dose dependent manner | 25151012 | |
| SKBR3 | Growth Inhibition Assay | 0-1.35 μM | 5 d | enhances the growth inhibition of AZD8931 | 26095475 | |
| KPL4 | Growth Inhibition Assay | 0-1.35 μM | 5 d | enhances the growth inhibition of AZD8931 | 26095475 | |
| BT474c | Growth Inhibition Assay | 0-1.35 μM | 5 d | enhances the growth inhibition of AZD8931 | 26095475 | |
| HCC1954 | Growth Inhibition Assay | 0-1.35 μM | 5 d | enhances the growth inhibition of AZD8931 | 26095475 | |
| TamR | Growth Inhibition Assay | 400 nM | 6 d | increased drug sensitivity of 4-OHT and fulvestrant | 26351323 | |
| T74D LTED | Growth Inhibition Assay | 100 nM | 6 d | increased drug sensitivity of 4-OHT and fulvestrant | 26351323 | |
| ZR75 LTED | Growth Inhibition Assay | 100 nM | 6 d | increased drug sensitivity of 4-OHT and fulvestrant | 26351323 | |
| MCF7 LTED | Growth Inhibition Assay | 200 nM | 6 d | increased drug sensitivity of 4-OHT and fulvestrant | 26351323 | |
| 1%MCF7 | Growth Inhibition Assay | 400 nM | 6 d | increased drug sensitivity of 4-OHT and fulvestrant | 26351323 | |
| T74D | Growth Inhibition Assay | 100 nM | 6 d | increased drug sensitivity of 4-OHT and fulvestrant | 26351323 | |
| ZR75 | Growth Inhibition Assay | 100 nM | 6 d | increased drug sensitivity of 4-OHT and fulvestrant | 26351323 | |
| MCF7 | Growth Inhibition Assay | 200 nM | 6 d | increased drug sensitivity of 4-OHT and fulvestrant | 26351323 | |
| LNCaP | Function assay | Inhibition of Akt in PTEN-deficient human LNCaP cells assessed as phosphorylation of GSK3beta, IC50 = 0.06 μM. | 23394218 | |||
| MDA-MB-468 | Function assay | 2 hrs | Inhibition of Akt in human MDA-MB-468 cells assessed as inhibition of GSK3beta phosphorylation after 2 hrs by laser scanning cytometry, IC50 = 0.089 μM. | 23394218 | ||
| LNCaP | Function assay | Inhibition of Akt in PTEN-deficient human LNCaP cells assessed as phosphorylation of PRAS40, IC50 = 0.22 μM. | 23394218 | |||
| BT474c | Function assay | Inhibition of Akt in human BT474c cells harboring HER2+/PIK3CA double mutant assessed as phosphorylation of PRAS40, IC50 = 0.31 μM. | 23394218 | |||
| MDA-MB-468 | Function assay | Inhibition of Akt in PTEN-deficient human MDA-MB-468 cells assessed as phosphorylation of GSK3beta, IC50 = 0.38 μM. | 23394218 | |||
| MDA-MB-468 | Function assay | Inhibition of Akt in PTEN-deficient human MDA-MB-468 cells assessed as phosphorylation of PRAS40, IC50 = 0.39 μM. | 23394218 | |||
| BT474c | Function assay | Inhibition of Akt in human BT474c cells harboring HER2+/PIK3CA double mutant assessed as phosphorylation of GSK3beta, IC50 = 0.76 μM. | 23394218 | |||
| RT4 | Function assay | Inhibition of PKA in TSC1 deficient human RT4 cells assessed as S6 phosphorylation, IC50 = 1 μM. | 23394218 | |||
| RT4 | Function assay | Inhibition of P70S6K in TSC1 deficient human RT4 cells assessed as S6 phosphorylation, IC50 = 5 μM. | 23394218 | |||
| PTEN-null LNCAP | Function assay | 1 hr | Inhibition of Akt in human PTEN-null LNCAP cells assessed as suppression in PRAS40 phosphorylation after 1 hr by ELISA analysis, IC50 = 0.3368 μM. | 27089211 | ||
| HCT116 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HCT116 cells after 72 hrs by SRB assay, IC50 = 5.2 μM. | 27089211 | ||
| OVCAR8 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human OVCAR8 cells after 72 hrs by SRB assay, IC50 = 7.27 μM. | 27089211 | ||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | |||
| DAOY | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for DAOY cells | 29435139 | |||
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Saos-2 cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-SH cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | |||
| MG 63 (6-TG R) | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for MG 63 (6-TG R) cells | 29435139 | |||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh41 cells | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | |||
| NB-EBc1 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB-EBc1 cells | 29435139 | |||
| LAN-5 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for LAN-5 cells | 29435139 | |||
| Rh18 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh18 cells | 29435139 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 86 mg/mL
(200.5 mM)
Ethanol : 20 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 428.92 | Formula | C21H25ClN6O2 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1143532-39-1 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | C1CN(CCC1(C(=O)NC(CCO)C2=CC=C(C=C2)Cl)N)C3=NC=NC4=C3C=CN4 | ||
Mechanism of Action
| Features |
Moderate preclinical tolerability, and PD characteristics of an AKT inhibitor. Distinct profile from other AKT inhibitors in clinical development.
|
|---|---|
| Targets/IC50/Ki |
Akt1
(Cell-free assay) 3 nM
Akt2
(Cell-free assay) 8 nM
Akt3
(Cell-free assay) 8 nM
ROCK2
(Cell-free assay) 56 nM
|
| In vitro |
Capivasertib (AZD5363) is a potent Akt inhibitor with IC50 of 3 nM, 8 nM and 8 nM for Akt1, Akt2 and Akt3, respectively. It inhibits phosphorylation of AKT substrates in cells with a potency of approximately 0.3 to 0.8 μM. This compound inhibits the proliferation of 41 of 182 solid and hematologic tumor cell lines with a potency of < 3 μM. Activating mutations in PIK3CA, loss or inactivation of tumor suppressor PTEN, or HER2 amplification all are significantly predictive of responsiveness to AZD5363. Additionally, correlation is also seen between the RAS mutation status of cell lines and resistance to it. |
| Kinase Assay |
Caliper Off-Chip Incubation Mobility Shift assay
|
|
The ability of Capivasertib (AZD5363) and other compounds to inhibit the activity of AKT1, AKT2, and AKT3 is evaluated by the Caliper Off-Chip Incubation Mobility Shift assay. Active recombinant AKT1, AKT2, or AKT3 are incubated with a 5-FAM-labeled custom-synthesized peptide substrate together with increasing concentrations of this compound. Final reactions contained 1 to 3 nM AKT1, AKT2, or AKT3 enzymes; 1.5 mM peptide substrate; ATP at K m for each AKT isoform; 10 mM MgCl2, 4 mM DTT, 100 mM HEPES, and 0.015% Brij-35. The reactions are incubated at room temperature for 1 hour and stopped by the addition of buffer containing 100 mM HEPES, 0.015% Brij-35 solution, 0.1% coating reagent, 40 mM EDTA, and 5% DMSO. Plates are then analyzed using a Caliper LC3000, allowing for separation of peptide substrate and phosphorylated product by electrophoresis with subsequent detection and quantification of laser induced fluorescence.
|
|
| In vivo |
Oral dosing of Capivasertib (AZD5363) (100, 300 mg/kg) to nude mice causes dose- and time-dependent reduction of PRAS40, GSK3β, and S6 phosphorylation in BT474c xenografts, reversible increases in blood glucose concentrations, and dose-dependent decreases in 2[18F]fluoro-2-deoxy-d-glucose (18F-FDG) uptake in U87-MG xenografts. Chronic oral dosing of this compound (130, 200, and 300 mg/kg) causes dose-dependent growth inhibition of xenografts derived from various tumor types, including HER2+ breast cancer models. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | pAKT / AKT / pGSK3β / GSK3β HER3 / pHER3 / HER2 / pHER2 / pPRAS40 / pS6 / p-4EBP1 / pFOXO / pERK / PARP / Cleaved PARP |
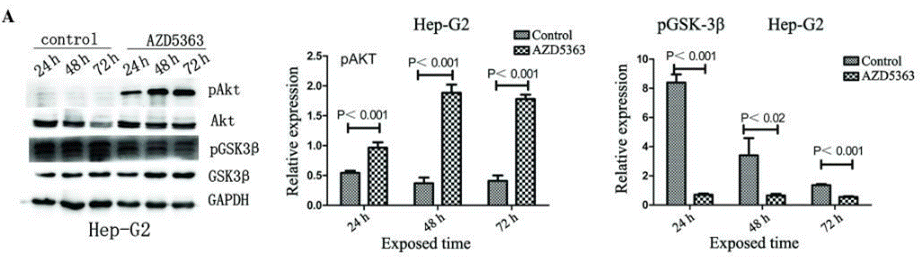
|
26998062 |
| Immunofluorescence | p-Chk2 / γ-H2AX |
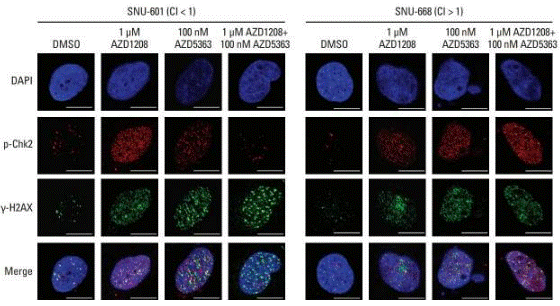
|
29879757 |
| Growth inhibition assay | Cell viability |
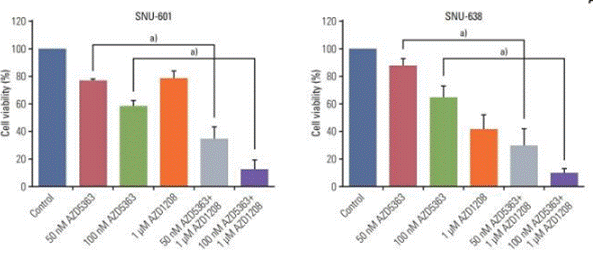
|
29879757 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT03310541 | Completed | Breast Cancer|Prostate Cancer|Advanced Solid Tumors |
Memorial Sloan Kettering Cancer Center |
October 11 2017 | Phase 1 |
| NCT01992952 | Active not recruiting | Estrogen Receptor Positive Breast Cancer |
Velindre NHS Trust|AstraZeneca|Cenduit LLC|Covance|Cardiff and Vale University Health Board |
May 2014 | Phase 1|Phase 2 |
| NCT02338622 | Completed | Advanced Cancer |
Royal Marsden NHS Foundation Trust|Institute of Cancer Research United Kingdom|AstraZeneca |
March 31 2014 | Phase 1 |
| NCT02121639 | Completed | Prostate Cancer |
University Hospital Southampton NHS Foundation Trust|AstraZeneca|Cancer Research UK |
January 29 2014 | Phase 1|Phase 2 |
| NCT02077569 | Completed | Invasive Breast Cancer |
University of Nottingham|AstraZeneca|Cancer Research UK|National Cancer Research Network |
January 2014 | Phase 2 |
| NCT01692262 | Completed | Metastatic Castrate-Resistant Prostate Cancer (mCRPC)|Efficacy|Safety and Tolerability|Pharmacokinetics|Pharmacodynamics|Tumour Response. |
AstraZeneca |
November 2012 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































