research use only
Miransertib (ARQ 092) HCl Akt inhibitor
Cat.No.S8339
Chemical Structure
Molecular Weight: 468.98
Quality Control
| Related Targets | PI3K mTOR GSK-3 ATM/ATR DNA-PK AMPK PDPK1 PTEN PP2A PDK |
|---|---|
| Other Akt Inhibitors | SC79 AZD5363 (Capivasertib) MK-2206 Dihydrochloride Ipatasertib (GDC-0068) Perifosine GSK690693 Triciribine (API-2) Afuresertib (GSK2110183) CCT128930 A-674563 HCl |
Solubility
|
In vitro |
DMSO
: 75 mg/mL
(159.92 mM)
Ethanol : 4 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 468.98 | Formula | C27H25ClN6 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1313883-00-9 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | Miransertib | Smiles | C1CC(C1)(C2=CC=C(C=C2)N3C4=C(C=CC(=N4)C5=CC=CC=C5)N=C3C6=C(N=CC=C6)N)N.Cl | ||
Mechanism of Action
| Targets/IC50/Ki |
Akt2
(Cell-free assay) 4.5 nM
Akt1
(Cell-free assay) 5 nM
Akt3
(Cell-free assay) 16 nM
|
|---|---|
| In vitro |
ARQ 092 blocks membrane translocation of inactive AKT and even dephosphorylates the membrane-associated active form, thereby perturbing AKT activity. Treatment with 50-500 nM ARQ 092 significantly blocks αMβ2 integrin function in neutrophils and reduces P-selectin exposure and glycoprotein Ib/IX/V-mediated agglutination in platelets. In a large panel of diverse cancer cell lines, ARQ 092 inhibits proliferation across multiple tumor types but are most potent in leukemia, breast, endometrial, and colorectal cancer cell lines. Moreover, inhibition by ARQ 092 is more prevalent in cancer cell lines containing PIK3CA/PIK3R1 mutations compared to those with wt-PIK3CA/PIK3R1 or PTEN mutations. ARQ 092 targets the PI3K/AKT pathway and AKT specifically and reduces phosphorylation of GSK3α and GSK3β in mutation-positive cells. |
| In vivo |
ARQ 092 is well tolerated at a continuous daily dose of 60 mg or a dose of 600 mg when administered once a week, for several months. ARQ 092 is likely to inhibit the activity of all AKT isoforms in intravascular cells and thereby attenuates the process of thrombosis and inflammation in SCD patients. ARQ 092 is highly active in a subset of endometrial tumors that harbor PI3K pathway gene mutations. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | p-AKT / AKT / p-PRAS40 / PRAS40 / p-ERK / ERK / p-FOXO1 / p-GSKβ(S9) / pAS160(S318) / pBAD / pS6 / p-4EBP1 |
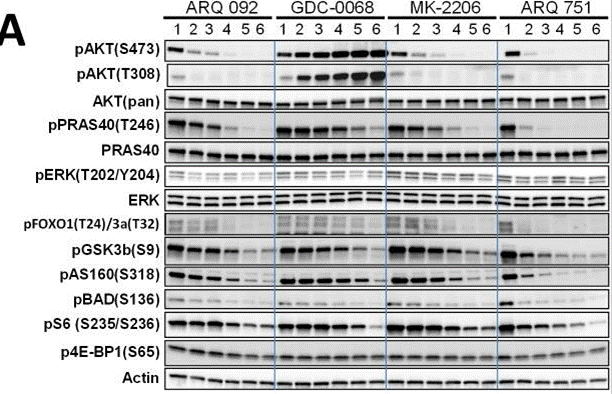
|
26469692 |
| Growth inhibition assay | Cell viability |
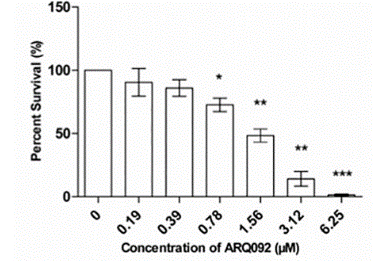
|
30399177 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT01473095 | Completed | Solid Tumor|Malignant Lymphoma|Tumor |
ArQule Inc. a subsidiary of Merck Sharp & Dohme LLC a subsidiary of Merck & Co. Inc. (Rahway NJ USA) |
November 2011 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































