| S1030 |
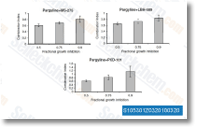
Panobinostat (LBH589)
|
Panobinostat (LBH589, NVP-LBH589) is a novel broad-spectrum HDAC inhibitor with IC50 of 5 nM in a cell-free assay. It induces autophagy and apoptosis, and effectively disrupts HIV latency in vivo. Phase 3.
|
-
J Cell Mol Med, 2026, 30(4):e71053
-
Hematol Oncol, 2026, 44(2):e70175
-
Cancer Cell, 2025, 43(4):776-796.e14
|
|
| S1185 |
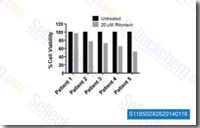
Ritonavir
|
Ritonavir is a Cytochrome P450 3A and Protease Inhibitor; Also inhibits Cytochrome P450 2D6, P-Glycoprotein and induces Cytochrome P450 2C19, Cytochrome P450 1A2, Cytochrome P450 2C9, Cytochrome P450 2B6 and UDP Glucuronosyltransferases. This compound induces apoptosis.
|
-
Nature, 2025, 10.1038/s41586-025-09755-9
-
Vet Parasitol, 2025, 337:110471
-
Cell Chem Biol, 2024, S2451-9456(24)00396-9
|
|
| S1380 |
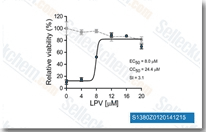
Lopinavir (ABT-378)
|
Lopinavir (ABT-378) is a potent HIV protease inhibitor with Ki of 1.3 pM in a cell-free assay.
|
-
Viruses, 2025, 1391
-
J Microbiol Biotechnol, 2025, 35:e2411063
-
Nat Commun, 2023, 14(1):5034
|
|
| S7381 |
Pepstatin A
|
Pepstatin A (Pepstatin) is a potent aspartic protease inhibitor, and also inhibits HIV replication. Pepstatin A is also an inhibitor of cathepsins D and cathepsins E. Pepstatin A inhibits autophagy by suppressing lysosomal proteases.
|
-
Signal Transduct Target Ther, 2025, 10(1):412
-
Nat Commun, 2025, 16(1):4069
-
Autophagy, 2025, 21(11):2473-2496
|
|
| S1457 |
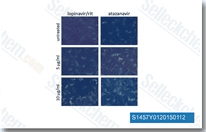
Atazanavir (BMS-232632) Sulfate
|
Atazanavir Sulfate (BMS-232632-05, Reyataz) is a HIV protease inhibitor with Ki of 2.66 nM in a cell-free assay.
|
-
Science Advances, 2025, eads1875
-
bioRxiv, 2024, 2024.05.10.593637
-
bioRxiv, 2023, nan
|
|
| S4282 |
Nelfinavir Mesylate
|
Nelfinavir Mesylate is a potent HIV protease inhibitor with Ki of 2 nM.
|
-
Biochem Biophys Rep, 2025, 42:101987
-
Int J Mol Sci, 2023, 24(7)6062
-
Int. J. Mol. Sci, 2023, 10.3390/ijms24076062
|
|
| S5250 |
Darunavir
|
Darunavir (TMC114,DRV) is a nonpeptidic HIV protease inhibitor, used to treat HIV infection.
|
-
Front Cell Infect Microbiol, 2024, 14:1334126
-
International Journal of Molecular Sciences, 2024, 2220
-
Frontiers in Microbiology, 2024, 1384009
|
|
| S4662 |
Atazanavir
|
Atazanavir (Latazanavir, Zrivada, Reyataz, BMS-232632) is an azapeptide and HIV-protease inhibitor that is used in the treatment of HIV infections and AIDS in combination with other anti-HIV agents. This compound is a substrate and inhibitor of cytochrome P450 isozyme 3A (CYP3A4) and an inhibitor and inducer of P-glycoprotein.
|
-
Anticancer Research, July 15, 2019, 3757-3765
-
Journal of Virology, April 17, 2019, e00077-19
-
Int J Mol Sci, 2022, 23(23)15380
|
|
| S1620 |
Darunavir Ethanolate
|
Darunavir Ethanolate (TMC-114, UIC 94017) is a nonpeptidic HIV protease inhibitor, used to treat HIV infection.
|
-
mBio, November 06, 2018, e01423-18
-
Antimicrobial Agents and Chemotherapy, April 24, 2017, e01776-16
-
J Virol, 2022, JVI0173021
|
|
| S9567 |
Indinavir Sulfate
|
Indinavir sulfate (Crixivan, L-735524, MK-639) is a specific and potent inhibitor of HIV-1 protease and is widely used in the treatment of AIDS.
|
-
Antimicrob Agents Chemother, 2023, 67(7):e0046223
-
J Cell Biochem, 2022, 10.1002/jcb.30213
-
Mol Metab, 2020, 29;101027
|
|










































