research use only
MMAE (Monomethyl auristatin E) Antineoplastic Agent
Cat.No.S7721
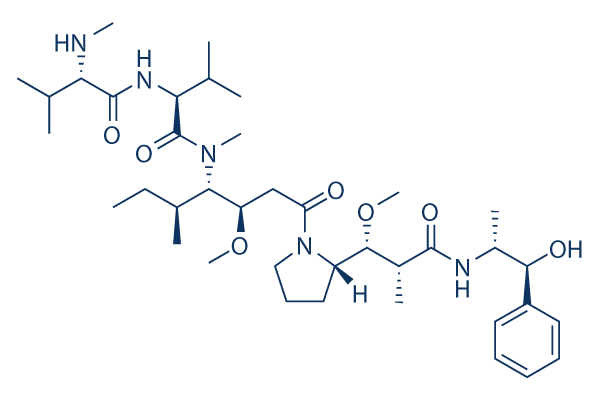
Chemical Structure
Molecular Weight: 717.98
Quality Control
| Related Targets | Akt Wnt/beta-catenin HSP PKC ROCK Microtubule Associated Integrin Bcr-Abl Actin FAK |
|---|---|
| Other ADC Cytotoxin Inhibitors | Triptolide SN-38 Luteolin (+)-Bicuculline Rutin Artemisinin Pinocembrin Psoralen Luteoloside Lycorine hydrochloride |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| KB | Antiproliferative assay | 4 days | Antiproliferative activity against human KB cells after 4 days by XTT assay, IC50=0.00019μM | 23845743 | ||
| BT-12 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-12 cells | 23845743 | |||
| A549 | Antiproliferative assay | 4 days | Antiproliferative activity against human A549 cells after 4 days by XTT assay, IC50=0.00059μM | 23845743 | ||
| non-small cell lung cancer cells | Cytotoxicity assay | 0.5 to 1 nM | 4 days | Cytotoxicity against human non-small cell lung cancer cells at 0.5 to 1 nM after 4 days by XTT assay | 23845743 | |
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 25431858 | |||
| MDA-MB-361 | Cytotoxicity assay | Cytotoxicity against human MDA-MB-361 cells assessed as cell viability incubated fore 4 days by MTS assay, GI50=0.00049μM | 25431858 | |||
| NCI-N87 | Cytotoxicity assay | Cytotoxicity against human NCI-N87 cells assessed as cell viability incubated fore 4 days by MTS assay, GI50=0.00054μM | 25431858 | |||
| OHS-50 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for OHS-50 cells | 28211277 | |||
| DU145 | Growth inhibition assay | 48 hrs | Growth inhibition of human DU145 cells incubated for 48 hrs by MTT assay, GI50=0.000418μM | 28211277 | ||
| SF268 | Growth inhibition assay | 48 hrs | Growth inhibition of human SF268 cells incubated for 48 hrs by MTT assay, GI50=0.000432μM | 28211277 | ||
| KM20L2 | Growth inhibition assay | 48 hrs | Growth inhibition of human KM20L2 cells incubated for 48 hrs by MTT assay, GI50=0.000599μM | 28211277 | ||
| NCI-H460 | Growth inhibition assay | 48 hrs | Growth inhibition of human NCI-H460 cells incubated for 48 hrs by MTT assay, GI50=0.000683μM | 28211277 | ||
| HCT116 | Function assay | 0.5 mM | 24 hrs | Effect on mitochondrial respiration in human HCT116 cells expressing carbonic anhydrase 9 assessed as reduction in oxygen consumption rate at 0.5 mM after 24 hrs under hypoxic condition by Western blot analysis | 28895394 | |
| DU145 | Growth inhibition assay | 48 hrs | Growth inhibition of human DU145 cells after 48 hrs by SRB assay, GI50=0.000418μM | 28895394 | ||
| SF268 | Growth inhibition assay | 48 hrs | Growth inhibition of human SF268 cells after 48 hrs by SRB assay, GI50=0.000432μM | 28895394 | ||
| KM20L2 | Growth inhibition assay | 48 hrs | Growth inhibition of human KM20L2 cells after 48 hrs by SRB assay, GI50=0.000599μM | 28895394 | ||
| NCI-H460 | Growth inhibition assay | 48 hrs | Growth inhibition of human NCI-H460 cells after 48 hrs by SRB assay, GI50=0.000683μM | 28895394 | ||
| MCF7 | Growth inhibition assay | 48 hrs | Growth inhibition of human MCF7 cells after 48 hrs by SRB assay, GI50=0.000404μM | 28926240 | ||
| DU145 | Growth inhibition assay | 48 hrs | Growth inhibition of human DU145 cells after 48 hrs by SRB assay, GI50=0.000418μM | 28926240 | ||
| SF268 | Growth inhibition assay | 48 hrs | Growth inhibition of human SF268 cells after 48 hrs by SRB assay, GI50=0.000432μM | 28926240 | ||
| KM20L2 | Growth inhibition assay | 48 hrs | Growth inhibition of human KM20L2 cells after 48 hrs by SRB assay, GI50=0.000599μM | 28926240 | ||
| NCI-H460 | Growth inhibition assay | 48 hrs | Growth inhibition of human NCI-H460 cells after 48 hrs by SRB assay, GI50=0.000683μM | 28926240 | ||
| DAOY | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for DAOY cells | 28972755 | |||
| A549 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human A549 cells after 72 hrs by MTS assay, EC50=0.0005μM | 28972755 | ||
| SKOV3 | Cytotoxicity assay | 2 days | Cytotoxicity against human SKOV3 cells after 2 days by cell titer 96 aqueous one solution based assay, IC50=0.00066μM | 29454703 | ||
| A549 | Cytotoxicity assay | 2 days | Cytotoxicity against human A549 cells after 2 days by cell titer 96 aqueous one solution based assay, IC50=0.0013μM | 29454703 | ||
| L1210 | Cytotoxicity assay | 2 days | Cytotoxicity against mouse L1210 cells after 2 days by cell titer 96 aqueous one solution based assay, IC50=0.0021μM | 29454703 | ||
| NCI-H524 | Cytotoxicity assay | 2 hrs | Cytotoxicity in human NCI-H524 cells pre-incubated for 2 hrs followed by compound wash out and subsequently incubated for 70 hrs by Cell Titer Glo assay, IC50=0.0037μM | 30735385 | ||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 100 mg/mL
(139.27 mM)
Ethanol : 100 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 717.98 | Formula | C39H67N5O7
|
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 474645-27-7 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | SGD-1010 | Smiles | CCC(C)C(C(CC(=O)N1CCCC1C(C(C)C(=O)NC(C)C(C2=CC=CC=C2)O)OC)OC)N(C)C(=O)C(C(C)C)NC(=O)C(C(C)C)NC | ||
Mechanism of Action
| In vitro |
When coupled to cAC10, MMAE shows selective cytotoxicity in CD30+ cells, and induces G2/M-phase growth arrest and cell death through the induction of apoptosis. When coupled to anti-CD79b antibody, anti–CD79b-vcMMAE has very potent and broad activity across a large panel of NHL cell lines in vitro. When coupled to anti-HER2 antibody, hertuzumab-vc-MMAE can also be effectively internalized and potently kill HER2 over-expressing tumor cells. |
|---|---|
| In vivo |
In the Karpas 299 ALCL model, cAC10-vcMMAE (1 mg/kg, i.v.) induces complete, durable tumor regression, while free MMAE (0.36 mg/kg) doesn’t produce detectable antitumor activity. In mouse xenograft models of NHL, anti–CD79b-vcMMAE (7 mg/kg, p.o.) strikingly results in sustained complete tumor remission. |
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06395103 | Not yet recruiting | B-cell Acute Lymphoblastic Leukemia|Diffuse Large B-cell Lymphoma|Burkitt Lymphoma|Neuroblastoma|Ewing Sarcoma |
Merck Sharp & Dohme LLC |
June 4 2024 | Phase 1|Phase 2 |
| NCT06227156 | Recruiting | Castration-resistant Prostate Cancer |
RemeGen Co. Ltd. |
April 8 2024 | Phase 1 |
| NCT06265727 | Recruiting | Solid Tumor Adult |
Corbus Pharmaceuticals Inc.|CSPC Zhongnuo Pharmaceutical (Shijiazhuang) Co. Ltd. |
April 1 2024 | Phase 1|Phase 2 |
| NCT06351020 | Not yet recruiting | Locally Advanced or Metastatic GC and GCJ Adenocarcinoma |
LaNova Medicines Zhejiang Co. Ltd.|LaNova Medicines Limited |
April 1 2024 | Phase 3 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































