research use only
Baricitinib (LY3009104) JAK inhibitor
Cat.No.S2851
Chemical Structure
Molecular Weight: 371.42
Quality Control
| Related Targets | EGFR STAT Pim |
|---|---|
| Other JAK Inhibitors | BMS-986165 (Deucravacitinib) AZD1480 WP1066 Momelotinib (CYT387) Filgotinib (GLPG0634) AT9283 Gandotinib (LY2784544) Pacritinib (SB1518) NVP-BSK805 2HCl TG101209 |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| human CD34+ cells | Function assay | 45 mins | Inhibition of JAK2 homodimer in human CD34+ cells spiked into human whole blood assessed as inhibition of EPO-induced STAT-5 phosphorylation preincubated for 45 mins followed by EPO addition measured after 15 mins by FACS analysis, IC50=0.0878μM | 24417533 | ||
| human UT7 cells | Function assay | Inhibition of JAK2 in human UT7 cells assessed as suppression of EPO-stimulated STAT5 phosphorylation by AlphaScreen assay | 26372653 | |||
| human TF1 cells | Function assay | Inhibition of JAK1 in human TF1 cells assessed as suppression of IL6-stimulated STAT3 phosphorylation by AlphaScreen assay | 26372653 | |||
| CD34+ | Function assay | 45 mins | Inhibition of JAK2 homodimer in human CD34+ cells spiked into human whole blood assessed as inhibition of EPO-induced STAT-5 phosphorylation preincubated for 45 mins followed by EPO addition measured after 15 mins by FACS analysis, IC50 = 0.0878 μM. | 24417533 | ||
| TF1 | Function assay | Inhibition of JAK1 in human TF1 cells assessed as suppression of IL6-stimulated STAT3 phosphorylation by AlphaScreen assay, INH = 0.017 μM. | 26372653 | |||
| UT7 | Function assay | Inhibition of JAK2 in human UT7 cells assessed as suppression of EPO-stimulated STAT5 phosphorylation by AlphaScreen assay, INH = 0.31 μM. | 26372653 | |||
| HeLa | Function assay | 5 uM | 2 hrs | Inhibition of IFNgamma-induced JAK2 phosphorylation in human HeLa cells at 5 uM incubated for 2 hrs by Western blot method | 27137359 | |
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-SH cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | |||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh41 cells | 29435139 | |||
| LAN-5 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for LAN-5 cells | 29435139 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 74 mg/mL
(199.23 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 371.42 | Formula | C16H17N7O2S |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1187594-09-7 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | INCB028050, LY3009104 | Smiles | CCS(=O)(=O)N1CC(C1)(CC#N)N2C=C(C=N2)C3=C4C=CNC4=NC=N3 | ||
Mechanism of Action
| Targets/IC50/Ki |
JAK2
(Cell-free assay) 5.7 nM
JAK1
(Cell-free assay) 5.9 nM
TYK2
(Cell-free assay) 53 nM
JAK3
(Cell-free assay) >400 nM
|
|---|---|
| In vitro |
Baricitinib (LY3009104) inhibits IL-6–stimulated phosphorylation of the canonical substrate STAT3 (pSTAT3) and subsequent production of the chemokine MCP-1 with IC50 values of 44 nM and 40 nM, respectively, in PBMCs. This compound also inhibits pSTAT3 stimulated by IL-23 with IC50 od 20 nM in isolated naive T-cells. |
| Kinase Assay |
Biochemical assays
|
|
Enzyme assays for Baricitinib (LY3009104) are performed using a homogeneous time-resolved fluorescence assay with recombinant epitope tagged kinase domains (JAK1, 837-1142; JAK2, 828-1132; JAK3, 718-1124; Tyk2, 873-1187) or full-length enzyme (cMET and Chk2) and peptide substrate. Each enzyme reaction is performed with or without this compound (11-point dilution), JAK, cMET, or Chk2 enzyme, 500 nM (100 nM for Chk2) peptide, ATP (at the Km specific for each kinase or 1 mM), and 2.0% DMSO in assay buffer. The calculated IC50 value is the concentration of it required for inhibition of 50% of the fluorescent signal.
|
|
| In vivo |
Baricitinib (LY3009104) inhibits IL-6–stimulated phosphorylation of STAT3 in whole blood with an IC50 of 128 nM. This compound (10 mg/kg p.o.) is expected to inhibit JAK1/2 signaling (by ≥50%) in rats for about 8 hours. It (10 mg/mL, p.o.) inhibits disease scores in dose-dependent manner in rats with established disease in the adjuvant arthritis model. This treatment, compared with vehicle, inhibits the increase in hind paw volumes during the 2 weeks of treatment by 50% at a dose of 1 mg/kg and >95% at doses of 3 mg/kg or 10 mg/kg. The compound, compared with vehicle, also inhibits composite score of immune infiltrate, edema, and periarticular tissue appearance by 27% at a dose of 1 mg/kg, 64% at doses of 3 mg/kg and 82% at doses of 10 mg/kg in rats with established disease in the adjuvant arthritis model. It reduces bone resorption by 15%, 61%, and 67% with increasing dose level (1, 3, and 10 mg/kg) in rats with established disease in the adjuvant arthritis model. This chemical (10 mg/kg, daily for 2 wk, p.o.) results in radiographic improvements with restoration of the normal architecture and appearance to the ankle and tarsals in rats with established disease in the adjuvant arthritis model. It reduces levels of pSTAT3 in a dose- and time-dependent manner in the peripheral blood of rAIA animals. This agent (10 mg/mL, p.o.) improves a composite score of joint damage by 47% in the murine CIA model. It (10 mg/kg) reduces pannus (74%) and bone damage (78%) and improves cartilage damage (43%) and signs of inflammation (33%), resulting in a 53% improvement in an aggregate score of disease in the collagen Ab-induced arthritis (CAIA) murine model. The compound (10 mg/kg) inhibits the delayed-type hypersensitivity response by 48% in both the CIA and CAIA models. It is efficacious in active rheumatoid arthritis patients refractory to disease modifying drugs and biologics. It preferentially inhibits JAK1 and JAK2, with 10-fold selectivity over Tyk2 and 100-fold over JAK3. The observed effects of GLPG-0634 on the ACR20, albeit in a smaller study, appear to be at least as good as that seen with tofacitinib and superior to that of baricitinib, since this chemical only moderately affect the ACR20 values in Phase IIa clinical studies. It has the dose-limiting side-effect of inducing anaemia which has been attributed to its effects on JAK2 but has clearly shown efficacy. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | phSTAT1 / phSTAT3 |
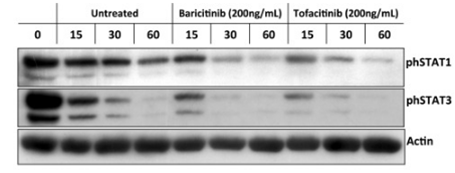
|
28369741 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05914584 | Not yet recruiting | Hospital-acquired Pneumonia |
Nantes University Hospital |
July 1 2023 | Phase 2|Phase 3 |
| NCT05189106 | Recruiting | Amyotrophic Lateral Sclerosis|Alzheimer Disease|Mild Cognitive Impairment |
Massachusetts General Hospital |
December 5 2022 | Phase 1|Phase 2 |
| NCT05074420 | Recruiting | Covid19|Corona Virus Infection |
Eli Lilly and Company |
December 21 2021 | Phase 3 |
| NCT04208464 | Completed | Idiopathic Inflammatory Myopathies |
University of Manchester|Eli Lilly and Company|Clinical Trials Unit Manchester|Karolinska Institutet |
October 7 2021 | Phase 2 |
| NCT04358614 | Completed | COVID|Pneumonia |
Fabrizio Cantini|Hospital of Prato |
March 16 2020 | Phase 2|Phase 3 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
Frequently Asked Questions
Question 1:
Do you know if it will dissolve directly into 0.5% methylcellulose (vehicle for oral gavage treatments) or is acid required to dissolve this compound?
Answer:
It can be dissolved directly into 0.5% methylcellulose, as cited from the reference. We also tested dissolving it into 30% PEG400/0.5% Tween80/5% propylene glycol, and the solubility is about 30 mg/mL.






































