research use only
Creatine JAK inhibitor
Cat.No.S5588
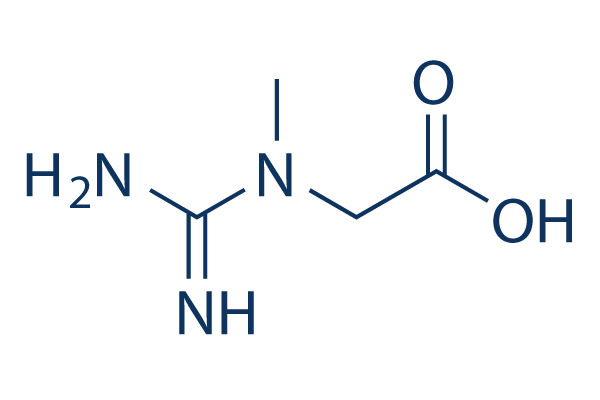
Chemical Structure
Molecular Weight: 131.13
Quality Control
| Related Targets | EGFR STAT Pim |
|---|---|
| Other JAK Inhibitors | BMS-986165 (Deucravacitinib) AZD1480 WP1066 Momelotinib (CYT387) Filgotinib (GLPG0634) AT9283 Gandotinib (LY2784544) Pacritinib (SB1518) TG101209 NVP-BSK805 2HCl |
Solubility
|
In vitro |
Water : 18 mg/mL (超声加热十分钟,60℃)
DMSO
: Insoluble
|
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 131.13 | Formula | C4H9N3O2 |
Storage (From the date of receipt) | 3 years -20°C powder |
|---|---|---|---|---|---|
| CAS No. | 57-00-1 | -- | Storage of Stock Solutions |
|
|
| Synonyms | Methylguanidoacetic acid | Smiles | CN(CC(=O)O)C(=N)N | ||
Mechanism of Action
| Targets/IC50/Ki |
JAK2
|
References |
|---|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT04218786 | Withdrawn | Myocardium; Injury|Myocardial Infarction|Myocardial Ischemia |
Shifa Tameer-e-Millat University|Rawalpindi Institute of Cardiology |
December 2025 | Phase 2 |
| NCT06225570 | Not yet recruiting | Acne Vulgaris |
Medical University of South Carolina |
January 2025 | Early Phase 1 |
| NCT05627479 | Not yet recruiting | Femoral Shaft Fracture|Tibial Shaft Fracture |
NYU Langone Health |
December 1 2024 | Phase 2 |
| NCT05647811 | Not yet recruiting | C3 Glomerulopathy |
NovelMed Therapeutics |
November 2024 | Phase 1|Phase 2 |
| NCT05946772 | Not yet recruiting | Takotsubo Cardiomyopathy |
University Hospital Heidelberg|German Centre of Cardiovascular Research (DZHK)|Coordinating Centre for Clinical Studies (KKS) Heidelberg |
October 2024 | Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































