research use only
Fasudil (HA-1077) Hydrochloride ROCK inhibitor
Cat.No.S1573
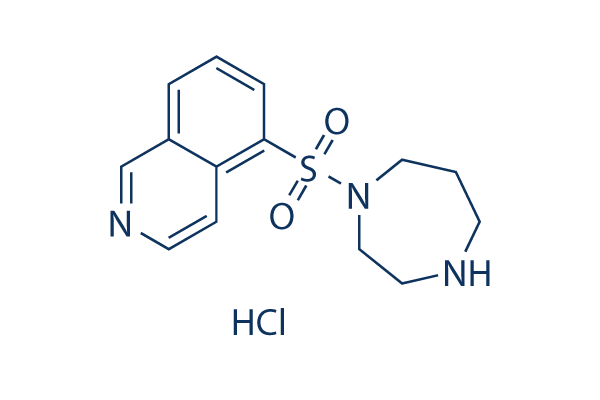
Chemical Structure
Molecular Weight: 327.83
Quality Control
Products Often Used Together with Fasudil (HA-1077) Hydrochloride
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| U937 cells | Function assay | Inhibition of MCP1-induced chemotaxis in U937 cells overexpressing CCR2, IC50=35 μM. | 17084087 | |||
| Sf9 cells | Function assay | Inhibition of His-tagged human MSK1 expressed in Sf9 cells, IC50=5 μM. | 10998351 | |||
| HEK293 cells | Function assay | Inhibition of His-tagged human PRK2 expressed in HEK293 cells, IC50=4 μM. | 10998351 | |||
| Sf9 cells | Function assay | Inhibition of rat ROCK2 expressed in Sf9 cells, IC50=1.9 μM. | 10998351 | |||
| insect cells | Function assay | 10 mins | Inhibition of human leukocytic ROCK1 expressed in insect cells using KKRNRTLSV as substrate after 10 mins by pyruvate kinase/lactate dehydrogenase coupled assay, Ki=0.53 μM. | 26039570 | ||
| Sf9 cells | Function assay | 90 mins | Inhibition of human recombinant N-terminal his-tagged ROCK1 (3-543) expressed in baculovirus infected Sf9 cells using Biotin-Ahx-AKRRLSSLRA-CONH2 substrate and [gamma-33P]ATP after 90 mins by scintillation proximity assay, IC50=0.3 μM. | 17018693 | ||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
Water : 65 mg/mL
DMSO
: 57 mg/mL
(173.87 mM)
Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 327.83 | Formula | C14H17N3O2S.HCl |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 105628-07-7 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | AT-877,HA-1077 HCl | Smiles | C1CNCCN(C1)S(=O)(=O)C2=CC=CC3=C2C=CN=C3.Cl | ||
Mechanism of Action
| Targets/IC50/Ki |
Rho
Calcium channel
ROCK2
(Cell-free assay) 330 nM(Ki)
PKA
(Cell-free assay) 1.6 μM(Ki)
PKG
(Cell-free assay) 1.6 μM(Ki)
PKC
(Cell-free assay) 3.3 μM(Ki)
MLCK
(Cell-free assay) 36 μM(Ki)
|
|---|---|
| In vitro |
Fasudil is a class of calcium antagonists. Fasudil produces a competitive inhibition of the Ca2+-induced contraction of the depolarized rabbit aorta. Fasudil is able to inhibit contractile responses to KCl, phenylephnne (PHE) and prostaglandin (PG) F2a. Fasudil also exhibits vasodilator actions by inhibition of 5-hydroxytryptamine, noradrenaline, histamine, angiotensin, and dopamine induced spiral strips contraction. Fasudil induces disorganization of actin stress fiber and cell migration inhibition. Fasudil inhibits hepatic stellate cells spreading, the formation of stress fibers, and expression of α-SMA with concomitant suppression of cell growth, but does not induce apoptosis. Fasudil suppresses the LPA-induced phosphorylation of ERK1/2, JNK and p38 MAPK. |
| Kinase Assay |
Cyclic AMP-dependent protein kinase activity assay
|
|
Cyclic AMP-dependent protein kinase activity is assayed in a reaction mixture containing, in a final volume of 0.2 mL, 50 mM Tris-HCl (pH 7.0), 10 mM magnesium acetate, 2 mM EGTA, 1 μM cyclic AMP or absence of cyclic AMP, 3.3 to 20 μM [r-32P] ATP (4×105 c.p.m.), 0.5 μg of the enzyme, 100 μg of histone H2B and compound. The mixture is incubated at 30 ℃ for 5 min. The reaction is terminated by adding 1mL of ice-cold 20% trichloroacetic acid after adding 500 μg of bovine serum albumin as a carrier protein. The sample is centrifuged at 3000 r.p.m. for 15min, the pellet is resuspended in ice-cold 10% trichloro-acetic acid solution and the centrifugation-resuspension cycle is repeated three times. The final pellet is dissolved in 1 mL of 1 N NaOH and radioactivity is measured with a liquid scintillation counter.
|
|
| In vivo |
Intra-coronary injection of Fasudil to dogs (30 μg i.a.) produces an approximate 50% increase in CBF. Fasudil (0.01, 0.03, 0.1 and 0.3 mg/kg, bolus, i.v.) dose-dependently decreases MBP and increases HR, VBF, CBF, RBF, and FBF. A total dose of 1.0 ng/mL Fasudil increases cardiac output. The infusion of Fasudil i.v. produces a significant fall in MBP, left ventricular systolic pressure and total peripheral resistance with an increase in HR and cardiac output, but without significant changes in right atrial pressure, dP/dt or left ventricular minute work in dogs. Fasudil administration displays protectable effects on cardiovascular disease and reduces the activation of JNK and attenuates mitochondrial-nuclear translocation of AIF under ischemic injury. The oral administration of Fasudil (a dosage of 100 mg/kg/day) significantly reduces incidence and mean maximum clinical score of EAE in SJL/J mice immunized with PLP p139-151. Treatment of mice with Fasudil suppresses the proliferative response of splenocytes to the antigen. Oral administration of Fasudil decreases inflammation, demyelination, axonal loss and APP positivein spinal cord of Fasudil-treated mice. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
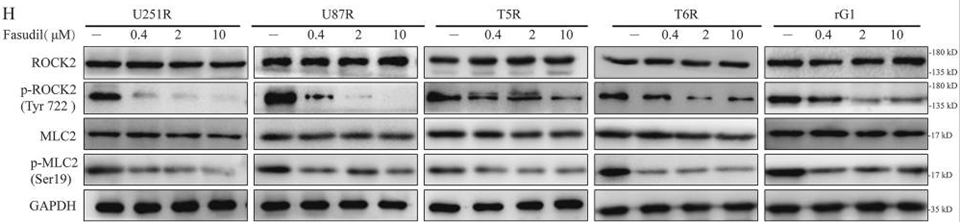
| Western blot | ROCK2 / p-ROCK2 / MLC2 / p-MLC2 ABCG2 Moesin / p-Moesin / β-catenin / p-β-catenin |
|
29416017 |
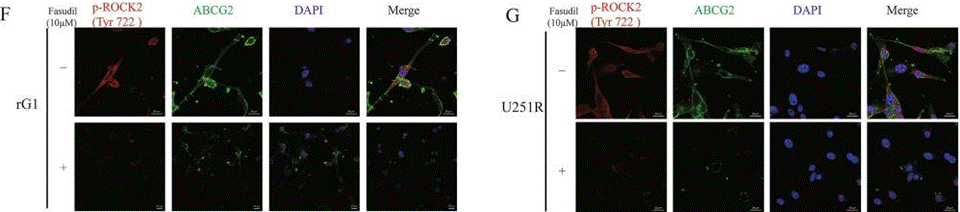
| Immunofluorescence | p-ROCK2 / ABCG2 |
|
29416017 |
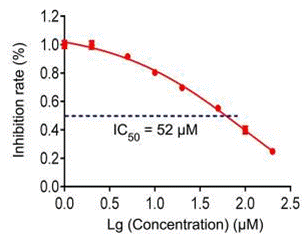
| Growth inhibition assay | Cell viability |
|
29262624 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































