research use only
Sodium ferulate 5-HT Receptor activator
Cat.No.S4740
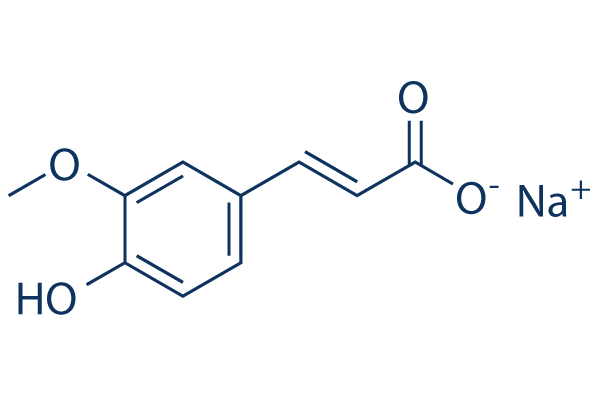
Chemical Structure
Molecular Weight: 216.17
Quality Control
Solubility
|
In vitro |
Water : 43 mg/mL
DMSO
: 2 mg/mL
(9.25 mM)
Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 216.17 | Formula | C10H9O4.Na |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 24276-84-4 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | Ferulic acid sodium salt | Smiles | COC1=C(C=CC(=C1)C=CC(=O)[O-])O.[Na+] | ||
Mechanism of Action
| In vitro |
SF is stable, water soluble. In vitro, SF (0.4 mg/mL) inhibits platelet aggregation induced by adenosine diphosphate (ADP) or collagen. At 1 mg/mL SF inhibits thrombin-induced platelet aggregation and release of [3H]5-HT from labelled platelets. The mechanism of SF action appears to be mediated by inhibition of cyclooxygenase and TXA2 synthase. In vitro SF inhibits lipid peroxide malondialdehyde (MDA) production from the platelets of rats, inhibits haemolysis induced by MDA and hydroxyl radical (OH·), and in erythrocyte membranes it inhibits lipid peroxidation induced by hydrogen peroxide (H2O2) and superoxide anions (O2-). SF is a direct scavenger of oxygen free radicals. SF is found to act as a novel non-peptide endothelin antagonist and to prevent the binding of ET-1 to its receptor.
|
|---|---|
| In vivo |
SF has antithrombotic, platelet aggregation inhibitory and antioxidant activities in animals and humans. In ethanol, carbon tetrachloride-, paracetamol-, or prednisolone-induced liver toxicity in mice, SF, 0.1 g/kg intragastrically, daily for 10 days inhibits the rise of liver lipid peroxides MDA content and alleviates liver lesions by stabilizing glutathione (GSH) and related enzymes levels. In glycerol-induced renal oxidative injury in mice, SF at 0.2 g/kg i.p., reverses the increase of renal MDA content and the decrease of GSH content, glutathione peroxidase (GSH-Px), glutathione S-transferase (GST), catalase (Cat), and SOD activities induced by glycerol injection, and improves renal histology. SF has a clear protective effect in experimental myocardial ischemia. In rabbits SF reduces the area of experimental myocardial infarction by 41% and decreases the oxygen consumption of guinea pig myocardial homogenates. SF has also a protective effect in myocardial ischemia reperfusion injury of rats. Furthermore, SF has an anti-atherogenetic effect in animal models. The antiarrhythmic effects of SF have been demonstrated in experimental animal models of arrhythmia, but not yet in clinical studies. The elimination half-life of SF is about 9.86 min. The acute oral LD50 of SF in mice is 3.2 g/kg.
|
References |
|
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































