research use only
SC-514 IκB/IKK inhibitor
Cat.No.S4907
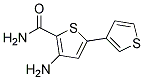
Chemical Structure
Molecular Weight: 224.3
Quality Control
| Related Targets | NF-κB HDAC Antioxidant ROS Nrf2 AP-1 MALT NOD |
|---|---|
| Other IκB/IKK Inhibitors | TBK1/IKKε-IN-5 Wedelolactone IKK-16 TPCA-1 BMS-345541 Bay 11-7085 IMD 0354 MRT67307 HCl PS-1145 WS6 |
Solubility
|
In vitro |
DMSO
: 45 mg/mL
(200.62 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 224.3 | Formula | C9H8N2OS2 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 354812-17-2 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | GK 01140 | Smiles | C1=CSC=C1C2=CC(=C(S2)C(=O)N)N | ||
Mechanism of Action
| Targets/IC50/Ki |
IKK2
3 μM-12 μM
CDK2/CyclinA
61 μM
AUR2
71 μM
PRAK
75 μM
MSK
123 μM
|
|---|---|
| In vitro |
SC-514 inhibits the native IKK complex or recombinant human IKK-1/IKK-2 heterodimer and IKK-2 homodimer similarly. This compound inhibits transcription of NF-κB-dependent IL-6, IL-8, and COX-2 genes in IL-1β-induced rheumatoid arthritis-derived synovial fibroblasts (RASFs) with IC50s of 20, 20 and 8 μM. 100 μM of this chemical blocks the phosphorylation and degradation of IκBα and also reduces the level of translocation of p65 into the nucleus in IL-1β-treated RASFs. It does not inhibit the phosphorylation and activation of the IKK complex. This inhibitor induces a delay but not a complete blockade in IκBα phosphorylation and degradation. Treatment with this compound shows a slightly slowed, decreased import of p65 into the nucleus and a faster export of p65 from the nucleus. It inhibits the phosphorylation of either IκBα or p65 similarly.
|
| In vivo |
SC-514 is efficacious in an acute model of inflammation, namely LPS-induced serum TNF-α production. This compound (50 mg/kg, i.p.) inhibits TNF-α production in vivo by ~70%.
|
References |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































