research use only
Azithromycin Dihydrate Anti-infection chemical
Cat.No.S4147
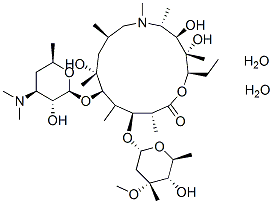
Chemical Structure
Molecular Weight: 785.02
Quality Control
Solubility
|
In vitro |
DMSO
: 100 mg/mL
(127.38 mM)
Ethanol : 100 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 785.02 | Formula | C38H72N2O12.2H2O |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 117772-70-0 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | CP-62993 dihydrate | Smiles | CCC1C(C(C(N(CC(CC(C(C(C(C(C(=O)O1)C)OC2CC(C(C(O2)C)O)(C)OC)C)OC3C(C(CC(O3)C)N(C)C)O)(C)O)C)C)C)O)(C)O.O.O | ||
Mechanism of Action
| In vitro |
Azithromycin displays a similar spectrum of antimicrobial activity. Azithromycin is marginally less active than erythromycin in vitro against Gram-positive organisms, although this is of doubtful clinical significance as susceptibility concentration fall within the range of achievable tissue Azithromycin concentrations. The mean MIC90 of Azithromycin against erythromycin susceptible/βL-ve strains, Streptococcus strains and NS (not selected for antimicrobial sesitivity)/erythromycin susceptible strains is 0.63, 0.35, and <0.27 mg/L, respectively. In contrast, Azithromycin appears to be more active than erythromycin against many Gram-negative pathogens and several other pathogens, notably Haemophilus influenza (MIC90, 1.34 mg/L), H. parainfluenzae(MIC90, 1 mg/L), Moraxella catarrhalis (MIC90, <0.1 mg/L), Neisseria gonorrhoeae (MIC90, 0.25 mg/L), Ureaplasma urealyticum, Bordetella spp (MIC90, 0.03-0.24 mg/L) and Borrelia burgdorferi. Azithromycin is also activity against clinical isolates of anaerobic bacteria Gram-positive cocci and propionibacterium acnes with MIC90 of 2.3, 0.03 mg/L. Like erythromycin and other macromycin, the activity of Azithromycin is unaffected by the production of β-lactamase. However, erythromycin-resistant organisms are also resistant to Azithromycin. |
References |
|---|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05972772 | Not yet recruiting | Infectious Disease|Therapeutics |
Lao-Oxford-Mahosot Hospital Wellcome Trust Research Unit|Mahidol Oxford Tropical Medicine Research Unit |
March 20 2024 | Phase 2|Phase 3 |
| NCT05796362 | Not yet recruiting | Infectious Disease|Drug Effect|Clinical Infection |
Brigham and Women''s Hospital|Bill and Melinda Gates Foundation |
April 2023 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































