research use only
Teicoplanin Bacterial chemical
Cat.No.S1399
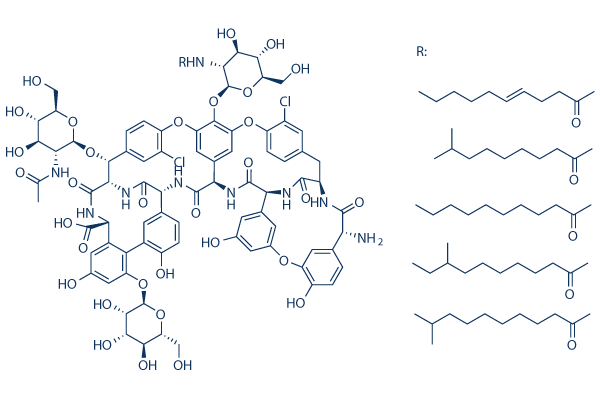
Chemical Structure
Molecular Weight: 1709.39
Quality Control
| Related Targets | Integrase Antibiotics Anti-infection Fungal Antiviral COVID-19 Parasite Reverse Transcriptase HIV HCV Protease |
|---|---|
| Other Bacterial Inhibitors | BTZ043 Racemate Pefloxacin Mesylate Ceftiofur Solithromycin 6-Methoxydihydrosanguinarine Proanthocyanidins Flagelin 22 Taurolidine Skatole Berberine Sulfate |
Solubility
|
In vitro |
Water : 100 mg/mL
DMSO
: Insoluble
Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 1709.39 | Formula | C77H77N9O31Cl2·R |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 61036-62-2 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | Teichomycin,Targocid,MDL-507 | Smiles | CCCCCCCCCC(=O)NC1C(C(C(OC1OC2=C3C=C4C=C2OC5=C(C=C(C=C5)C(C6C(=O)NC(C7=C(C(=CC(=C7)O)OC8C(C(C(C(O8)CO)O)O)O)C9=C(C=CC(=C9)C(C(=O)N6)NC(=O)C4NC(=O)C1C2=CC(=CC(=C2)OC2=C(C=CC(=C2)C(C(=O)NC(CC2=CC(=C(O3)C=C2)Cl)C(=O)N1)N)O)O)O)C(=O)O)OC1C(C(C(C(O1)CO)O)O)NC(=O)C)Cl)CO)O)O | ||
Mechanism of Action
| In vitro |
Teicoplanin is associated with small increases in the GI colonization by C. albicans. The inhibitory action of this compound on proteoglycan polymerization but not on nucleic acid and protein synthesis indicates that the effect in the wall is due to the accumulation at this level of proteoglycan soluble precursors continuously produced by the cell. It, at a concentration of 1 mg/mL, causes rapid lysis of exponential phase cells of Streptococcus faecalis. This compound inhibits peptidoglycan synthesis in intact cells and in cell-free systems. This chemical-induced lysis is dependent on the growth phase of B. pumilus at the time of addition of the antibiotic. It induces lysis after a longer lag phase with slowly growing Strep. jhecalis CCM 1875 and lysis is not very extensive. |
|---|---|
| In vivo |
Teicoplanin therapy increases the survival rate whereas G-CSF therapy does not in comparison to other groups in neutropenic mice. This compound and G-CSF combination therapy improves survival rate when compared with groups II, III, IV. |
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT03909698 | Completed | Kidney Failure Chronic|Antibiotics|Hemodialysis |
University Hospital Ghent|University Ghent |
September 15 2016 | -- |
| NCT03229135 | Completed | Pneumonia Gram-Positive Bacterial |
People''s Hospital of Zhengzhou University |
February 1 2015 | -- |
| NCT03177369 | Completed | Teicoplanin-based Antimicrobial Therapy|Staphylococcus Aureus|Bone and Joint Infection |
Hospices Civils de Lyon |
January 2015 | -- |
| NCT01324804 | Unknown status | Coronary Heart Disease|Surgical Wound Infection |
Medical University of Vienna |
November 2010 | -- |
| NCT00754273 | Completed | Methicillin-resistant Staphylococcal Aureus Pneumonia |
University of Kentucky|Pfizer |
September 2008 | -- |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































