research use only
Odanacatib Cysteine Protease inhibitor
Cat.No.S1115
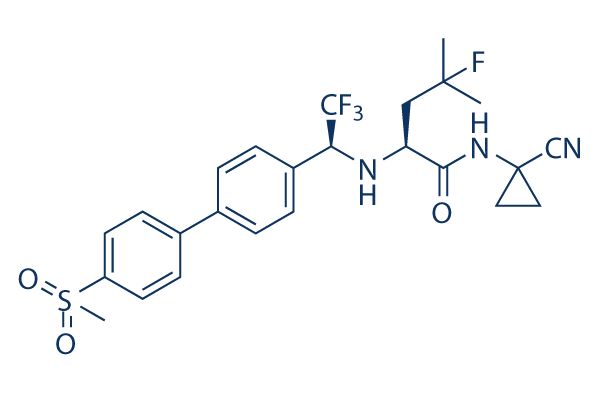
Chemical Structure
Molecular Weight: 525.56
Quality Control
| Related Targets | HDAC Caspase Proteasome Secretase MMP HCV Protease Tyrosinase HIV Protease DPP Serine Protease |
|---|---|
| Other Cysteine Protease Inhibitors | N-Ethylmaleimide (NEM) MG132 SSS Calpeptin Aloxistatin (E-64d) E-64 MG-101 (ALLN) Z-FA-FMK Cathepsin Inhibitor 1 PD 151746 Loxistatin Acid (E-64C) |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| ramos cells | Function assay | Inhibition of cathepsin S in human ramos cells, IC50=0.045μM | 18226527 | |||
| stem cells | Function assay | 7 days | Inhibition of osteoclastogenesis in human bone marrow-derived stem cells assessed as reduction of pit formation by measuring TRACP5b activity after 7 days by bone TRAP assay, IC50=0.1μM | 22984809 | ||
| HepG2 | Function assay | Inhibition of cathepsin B in human HepG2 cells, IC50=1.05μM | 18226527 | |||
| B cells | Function assay | Inhibition of antigen presenting mouse B cells, IC50=1.5μM | 18226527 | |||
| HepG2 | Function assay | Inhibition of cathepsin L in human HepG2 cells, IC50=4.843μM | 18226527 | |||
| SJ-GBM2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SJ-GBM2 cells | 29435139 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 100 mg/mL
(190.27 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 525.56 | Formula | C25H27F4N3O3S |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 603139-19-1 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | MK-0822 | Smiles | CC(C)(CC(C(=O)NC1(CC1)C#N)NC(C2=CC=C(C=C2)C3=CC=C(C=C3)S(=O)(=O)C)C(F)(F)F)F | ||
Mechanism of Action
| Features |
A potent, selective, and neutral cathepsin K inhibitor.
|
|---|---|
| Targets/IC50/Ki |
Cathepsin K (human)
0.2 nM
Cathepsin K (rabbit)
1 nM
|
| In vitro |
In vitro, Odanacatib shows the high inhibitory activity and selectivity on cathepsin K with IC50 values of 0.2 nM and 1 nM for human cathepsin K and rabbit cathepsin K, respectively. Furthermore, this compound also shows similar potencies in whole human cell enzyme occupancy assays with corrected IC50 of 5 nM. A recent study shows that this chemical results in reduction of Osteoclast (OC) resorption activity by interrupting intracellular vesicular trafficking.
|
| In vivo |
In preclinical rats, Odanacatib (10 mg/kg) exhibits excellent pharmacokinetics with clearance (Cl: 2 mL kg-1 min-1), low volume of distribution (Vdss: 1.1 L kg-1), half-life (T1/2: 6 hours) and oral bioavailability (F: 8%), respectively. Besides, this compound also exhibits excellent metabolic stability in rat hepatocytes with a 96% recovery of the parent identity. This chemical administrated by p.o. prevents bone loss in ovariectomized (OVX) rabbits in a dose-related manner. Moreover, it (9 µM/day) leads to a significant increase in proximal femur bone mineral density (BMD) (7.8%), femoral neck BMD (10.8%) and the greater trochanter BMD (6.5%). In the estrogen-deficient, skeletally mature rhesus monkeys, long-term treatment with this compound effectively inhibits bone turnover without reducing osteoclast number and maintains normal biomechanical properties of the spine of OVX nonhuman primates.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT01803607 | Terminated | Osteoporosis |
Merck Sharp & Dohme LLC |
March 14 2013 | Phase 3 |
| NCT01630616 | Terminated | Osteoporosis |
Merck Sharp & Dohme LLC |
March 12 2013 | Phase 1 |
| NCT01512693 | Completed | Hepatic Insufficiency |
Merck Sharp & Dohme LLC |
February 23 2012 | Phase 1 |
| NCT01512667 | Completed | Renal Insufficiency |
Merck Sharp & Dohme LLC |
January 17 2012 | Phase 1 |
| NCT01068262 | Completed | Osteoporosis |
Merck Sharp & Dohme LLC |
December 8 2009 | Phase 1 |
| NCT00691899 | Withdrawn | Prostate Cancer |
Merck Sharp & Dohme LLC |
September 2008 | Phase 3 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































