research use only
Doramapimod (BIRB 796) p38 MAPK inhibitor
Cat.No.S1574
Chemical Structure
Molecular Weight: 527.66
Quality Control
| Related Targets | ERK Raf JNK MEK Ras KRas S6 Kinase MAP4K TAK1 Mixed Lineage Kinase |
|---|---|
| Other p38 MAPK Inhibitors | SB202190 SB203580 (Adezmapimod) PH-797804 Ralimetinib (LY2228820) dimesylate VX-702 Losmapimod SB239063 Asiatic Acid Neflamapimod (VX-745) BMS-582949 |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| HOP-92 | Growth Inhibition Assay | IC50=24.3838 μM | SANGER | |||
| NCI-H2228 | Growth Inhibition Assay | IC50=23.6668 μM | SANGER | |||
| CAPAN-1 | Growth Inhibition Assay | IC50=22.1884 μM | SANGER | |||
| SK-MEL-1 | Growth Inhibition Assay | IC50=20.3683 μM | SANGER | |||
| DB | Growth Inhibition Assay | IC50=18.7923 μM | SANGER | |||
| AN3-CA | Growth Inhibition Assay | IC50=18.1 μM | SANGER | |||
| EW-13 | Growth Inhibition Assay | IC50=17.9516 μM | SANGER | |||
| LC-2-ad | Growth Inhibition Assay | IC50=17.4366 μM | SANGER | |||
| ES8 | Growth Inhibition Assay | IC50=17.167 μM | SANGER | |||
| NCI-H1581 | Growth Inhibition Assay | IC50=17.0447 μM | SANGER | |||
| HMV-II | Growth Inhibition Assay | IC50=14.2309 μM | SANGER | |||
| BEN | Growth Inhibition Assay | IC50=13.1264 μM | SANGER | |||
| NBsusSR | Growth Inhibition Assay | IC50=10.8235 μM | SANGER | |||
| MS-1 | Growth Inhibition Assay | IC50=10.8235 μM | SANGER | |||
| BFTC-905 | Growth Inhibition Assay | IC50=10.1233 μM | SANGER | |||
| NCI-SNU-1 | Growth Inhibition Assay | IC50=9.06739 μM | SANGER | |||
| MPP-89 | Growth Inhibition Assay | IC50=8.46251 μM | SANGER | |||
| T-24 | Growth Inhibition Assay | IC50=8.40673 μM | SANGER | |||
| KP-N-YN | Growth Inhibition Assay | IC50=8.2019 μM | SANGER | |||
| IST-MES1 | Growth Inhibition Assay | IC50=7.95637 μM | SANGER | |||
| NCI-H358 | Growth Inhibition Assay | IC50=7.538 μM | SANGER | |||
| GOTO | Growth Inhibition Assay | IC50=6.39161 μM | SANGER | |||
| DU-145 | Growth Inhibition Assay | IC50=3.93811 μM | SANGER | |||
| EoL-1-cell | Growth Inhibition Assay | IC50=0.34763 μM | SANGER | |||
| KYSE-270 | Growth Inhibition Assay | IC50=24.5573 μM | SANGER | |||
| HCC1806 | Growth Inhibition Assay | IC50=24.7799 μM | SANGER | |||
| HuO-3N1 | Growth Inhibition Assay | IC50=25.8185 μM | SANGER | |||
| HOS | Growth Inhibition Assay | IC50=25.9292 μM | SANGER | |||
| KYSE-510 | Growth Inhibition Assay | IC50=26.1612 μM | SANGER | |||
| COLO-741 | Growth Inhibition Assay | IC50=26.3329 μM | SANGER | |||
| H-EMC-SS | Growth Inhibition Assay | IC50=26.9245 μM | SANGER | |||
| HCC1937 | Growth Inhibition Assay | IC50=27.2238 μM | SANGER | |||
| NCI-H2126 | Growth Inhibition Assay | IC50=27.3975 μM | SANGER | |||
| NCI-H1703 | Growth Inhibition Assay | IC50=28.0413 μM | SANGER | |||
| U-2-OS | Growth Inhibition Assay | IC50=28.5515 μM | SANGER | |||
| DBTRG-05MG | Growth Inhibition Assay | IC50=28.5651 μM | SANGER | |||
| MHH-ES-1 | Growth Inhibition Assay | IC50=31.941 μM | SANGER | |||
| HCC1419 | Growth Inhibition Assay | IC50=32.1119 μM | SANGER | |||
| HOP-62 | Growth Inhibition Assay | IC50=32.2701 μM | SANGER | |||
| AM-38 | Growth Inhibition Assay | IC50=32.9931 μM | SANGER | |||
| NCI-H2009 | Growth Inhibition Assay | IC50=33.4007 μM | SANGER | |||
| EM-2 | Growth Inhibition Assay | IC50=33.5511 μM | SANGER | |||
| SW1116 | Growth Inhibition Assay | IC50=34.4838 μM | SANGER | |||
| SK-N-AS | Growth Inhibition Assay | IC50=35.0714 μM | SANGER | |||
| ChaGo-K-1 | Growth Inhibition Assay | IC50=35.6032 μM | SANGER | |||
| RT-112 | Growth Inhibition Assay | IC50=35.9879 μM | SANGER | |||
| HTC-C3 | Growth Inhibition Assay | IC50=36.2355 μM | SANGER | |||
| SK-NEP-1 | Growth Inhibition Assay | IC50=36.6106 μM | SANGER | |||
| LB831-BLC | Growth Inhibition Assay | IC50=37.6541 μM | SANGER | |||
| CTB-1 | Growth Inhibition Assay | IC50=38.4512 μM | SANGER | |||
| MOLT-4 | Growth Inhibition Assay | IC50=38.8391 μM | SANGER | |||
| SW756 | Growth Inhibition Assay | IC50=40.9385 μM | SANGER | |||
| CAL-72 | Growth Inhibition Assay | IC50=42.03 μM | SANGER | |||
| KNS-62 | Growth Inhibition Assay | IC50=42.6296 μM | SANGER | |||
| KARPAS-299 | Growth Inhibition Assay | IC50=43.3233 μM | SANGER | |||
| HEL | Growth Inhibition Assay | IC50=45.4646 μM | SANGER | |||
| KP-4 | Growth Inhibition Assay | IC50=46.7361 μM | SANGER | |||
| NEC8 | Growth Inhibition Assay | IC50=47.1661 μM | SANGER | |||
| G-402 | Growth Inhibition Assay | IC50=48.7012 μM | SANGER | |||
| Sf21 | Function assay | Binding affinity to wild type human biotin labelled p38 alpha (9 to 352 residues) expressed in sf21 insect cells SPR analysis, Kd = 0.000123 μM. | 28834431 | |||
| THP1 | Function assay | Inhibition of LPS-induced TNFalpha production in human THP1 cells, IC50 = 0.013 μM. | 18325768 | |||
| U937 | Antiinflammatory assay | 2 hrs | Antiinflammatory activity in differentiated human U937 cells assessed as inhibition of LPS-induced TNFalpha production preincubated for 2 hrs followed by LPS-stimulation for 4 hrs by sandwich ELISA relative to vehicle-treated control, IC50 = 0.015 μM. | 26800309 | ||
| THP1 | Function assay | Inhibition of LPS-induced TNFalpha production in human THP1 cells, IC50 = 0.018 μM. | 19356929 | |||
| THP1 | Function assay | Inhibition of LPS-induced tumor necrosis factor-alpha (TNF-alpha) production in THP-1 cells, EC50 = 0.018 μM. | 12086485 | |||
| THP | Function assay | Tested for inhibition of Tumor necrosis factor, alpha in THP cells, EC50 = 0.018 μM. | 14561087 | |||
| THP1 | Function assay | Inhibition of LPS-induced TNFalpha production in THP1 cells, IC50 = 0.018 μM. | 17560108 | |||
| THP1 | Function assay | Inhibition of LPS-stimulated TNFalpha production in human THP1 cells, IC50 = 0.018 μM. | 18462940 | |||
| HLF | Function assay | Inhibition of p38alpha phosphorylation in IL-1-alpha-stimulated HLF cells, IC50 = 0.047 μM. | 18602262 | |||
| HLF | Function assay | Inhibition of HSP27 phosphorylation in IL-1-alpha-stimulated HLF cells, IC50 = 0.058 μM. | 18602262 | |||
| HEK293F | Function assay | Inhibition of sodium arsenate activated N-terminal GST-tagged Brugia malayi MPK1 expressed in HEK293F cells using FAM-p38tide as substrate by IMAP assay, IC50 = 0.14 μM. | 29541362 | |||
| BL21(DE3) | Function assay | Inhibition of p38alpha active form expressed in Escherichia coli BL21(DE3) cells by HTRF assay, IC50 = 0.25 μM. | 19928858 | |||
| whole blood cells | Antiinflammatory assay | 4 hrs | Antiinflammatory activity in human whole blood cells assessed as inhibition of LPS-induced TNFalpha production after 4 hrs by time-resolved fluorescence assay, IC50 = 0.6 μM. | 22749282 | ||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | |||
| DAOY | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for DAOY cells | 29435139 | |||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for RD cells | 29435139 | |||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-SH cells | 29435139 | |||
| MG 63 (6-TG R) | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for MG 63 (6-TG R) cells | 29435139 | |||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | |||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 100 mg/mL
(189.51 mM)
Ethanol : 100 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 527.66 | Formula | C31H37N5O3 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 285983-48-4 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | CC1=CC=C(C=C1)N2C(=CC(=N2)C(C)(C)C)NC(=O)NC3=CC=C(C4=CC=CC=C43)OCCN5CCOCC5 | ||
Mechanism of Action
| Features |
The first p38 MAPK inhibitor to be tested in a phase III clinical trial.
|
|---|---|
| Targets/IC50/Ki |
JNK2
c-RAF
Fyn
p38α
(Cell-free assay) 0.1 nM(Kd)
p38α
38 nM
|
| In vitro |
Doramapimod (BIRB 796) shows no significant inhibition to ERK-1, SYK, IKK2β, ZAP-70, EGF receptor kinase, HER2, protein kinase A (PKA), PKC, PKC-α, PKC-β (I and II) and PKC-γ. This compound greatly improves binding affinity by forming a hydrogen bond between the morpholine oxygen and the ATP-binding domain of p38α. It represents one of the most potent and slowest dissociating inhibitors against human p38 MAP kinase now known. It potently inhibits c-Raf-1 and Jnk2α2 with IC50 of 1.4 and 0.1 nM, respectively. BIRB796 also inhibits the activity and the activation of SAPK3/p38γ at a higher concentration than it does in p38α. It blocks the stress-induced phosphorylation of the scaffold protein SAP97, which is a physiological substrate of SAPK3/p38γ. This compound blocks JNK1/2 activation and activity in HEK293 cells, while not inhibits the activation and activity of ERK1/ERK2 in Hela cells. Moreover, the binding of BIRB796 to the p38 MAPKs or JNK1/2 is impairing their phosphorylation by the upstream kinase MKK6 or MKK4 rather than enhancing their dephosphorylation. It blocks baseline and upregulation of p38 MAPK and Hsp27 phosphorylation, thereby enhancing cytotoxicity and caspase activation. This compound downregulates IL-6 and VEGF secretion in BMSCs triggered by TNF-α and TGF-β1. It has a pyrazole scaffold that places a lipophilic t-butyl group into the lower selectivity site and a tolyl ring into the upper selectivity site. This compound also inhibits B-Raf and Abl with IC50 of 83 nM and 14.6 μM, respectively. |
| Kinase Assay |
Procedures for the THP-1 cellular assay for inhibition of LPS-stimulated TNF-α production
|
|
THP-1 cells are preincubated in the presence and absence of Doramapimod (BIRB 796) for 30 min. The cell mixture is stimulated with LPS (1 μg/mL final) and incubation continued overnight (18−24 hours) as above. Supernatant is analyzed for human TNF-α by a commercially available ELISA. Data are combined and analyzed by nonlinear regression using a three parameter logistic model to obtain an EC50 value. This compound is analyzed in each experiment and the 95% confidence intervals for the EC50 are between 16 and 22 nM.
|
|
| In vivo |
Doramapimod (BIRB 796) (30 mg/kg) inhibits 84% of TNF-α in LPS-stimulated mice and demonstrates efficacy in a mouse model of established collagen-induced arthritis. This compound has good pharmacokinetic performance even after oral administration in mice. |
References |
|
Applications
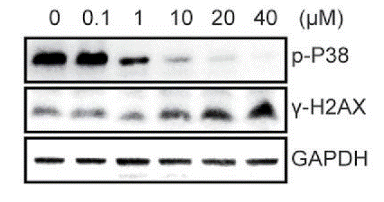
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | p-p38 / γ-H2AX mTOR / p-S6K / S6K / p-MK2 / MK2 / p-Hsp27 / Hsp27 p-p38 / p38 |
|
27082306 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT02211885 | Completed | Healthy |
Boehringer Ingelheim |
October 2002 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































