research use only
Sunitinib (SU-11248) Multi-targeted Receptor Tyrosine Kinase Inhibitor
Cat.No.S7781
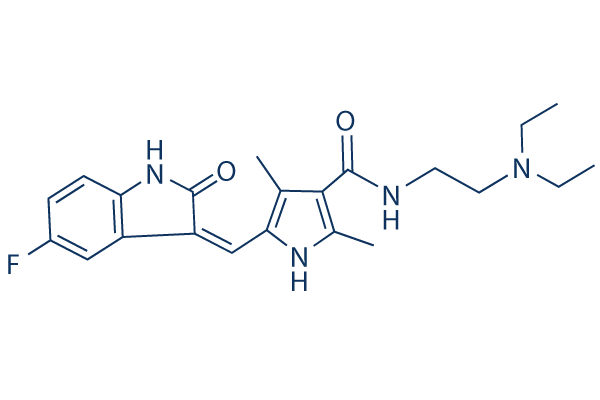
Chemical Structure
Molecular Weight: 398.47
Quality Control
| Related Targets | EGFR VEGFR FGFR c-Met Src MEK CSF-1R FLT3 HER2 c-Kit |
|---|---|
| Other PDGFR Inhibitors | CP-673451 Orantinib (SU6668) Tyrphostin AG 1296 Trapidil PP121 AZD2932 Sennoside B Tyrphostin AG1433 AG 1295 N-(p-Coumaroyl) Serotonin |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| HUVEC cell | Function assay | 24 h | Inhibition of VEGF-A induced HUVEC cell sprouting after 24 hrs by angiogenesis assay, IC50=0.12 μM | 21741249 | ||
| HEK293 | Function assay | Binding affinity to FLT3 catalytic domain expressed in HEK293 cells by competitive binding assay, Kd = 0.00047 μM. | 19754199 | |||
| MV4-11 | Antiproliferative assay | 72 hrs | Antiproliferative activity against FLT3-ITD dependent human MV4-11 cells assessed as reduction in cell viability after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 0.001 μM. | 27077705 | ||
| HL60 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HL60 cells assessed as reduction in cell viability after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 0.001 μM. | 27077705 | ||
| BAF3 | Function assay | 72 hrs | Inhibition of Tel-fused cKIT T670I mutant (unknown origin) transfected in mouse BAF3 cells assessed as decrease in cell proliferation after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 0.002 μM. | 27077705 | ||
| MV4-11 | Cytotoxicity assay | 72 hrs | Cytotoxicity against FLT3-dependent human MV4-11 cells after 72 hrs by MTT assay, IC50 = 0.003 μM. | 22452518 | ||
| MV411 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MV411 cells after 48 hrs by MTT assay, IC50 = 0.003 μM. | 24904961 | ||
| BA/F3 | Function assay | 72 hrs | Inhibition of Kit exon 11 deletion (557 to 558 residues) mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.004 μM. | 30204441 | ||
| MV4-11 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MV4-11 cells after 72 hrs by cell titer-blue cell viability assay, IC50 = 0.0043 μM. | 19754199 | ||
| MV4-11 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MV4-11 cells after 72 hrs by cell titer-blue assay, IC50 = 0.0043 μM. | 19654408 | ||
| Sf21 | Function assay | 60 mins | Inhibition of recombinant human N-terminal His6-tagged KDR (790 to end residues) expressed in baculovirus infected Sf21 insect cells after 60 mins by ELISA, IC50 = 0.0047 μM. | 30241010 | ||
| BA/F3 | Function assay | 72 hrs | Inhibition of Kit exon 11 deletion (557 to 558 residues) and D816H mutant and T670I mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.005 μM. | 30204441 | ||
| MOLM14 | Antiproliferative assay | 72 hrs | Antiproliferative activity against FLT3-ITD dependent human MOLM14 cells assessed as reduction in cell viability after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 0.005 μM. | 27077705 | ||
| BA/F3 | Function assay | 72 hrs | Inhibition of Kit exon 11 deletion (557 to 558 residues) and V654A mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.006 μM. | 30204441 | ||
| BAF3 | Function assay | 72 hrs | Inhibition of Tel-fused cKIT V559D/V654A double mutant (unknown origin) transfected in mouse BAF3 cells assessed as decrease in cell proliferation after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 0.006 μM. | 27077705 | ||
| MDA-MB-468 | Cytotoxicity assay | Cytotoxicity against human triple negative MDA-MB-468 cells, IC50 = 0.0061 μM. | 24890652 | |||
| 3T3 | Function assay | Inhibition of PDGF-induced BrdU incorporation in 3T3 cells without bovine serum albumin, IC50 = 0.007 μM. | 12646019 | |||
| 3T3 | Function assay | Inhibition of PDGF-induced BrdU incorporation in 3T3 cells with 0.1% bovine serum albumin, IC50 = 0.007 μM. | 12646019 | |||
| 3T3 | Function assay | Inhibition of PDGF-induced BrdU incorporation in 3T3 cells with 0.5% bovine serum albumin, IC50 = 0.007 μM. | 12646019 | |||
| BA/F3 | Function assay | 72 hrs | Inhibition of Kit exon 9 AY502 to 503 insertion mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.007 μM. | 30204441 | ||
| BA/F3 | Function assay | 72 hrs | Inhibition of Kit V560D/V654A mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.007 μM. | 30204441 | ||
| 3T3 | Function assay | Inhibition of PDGF-induced BrdU incorporation in 3T3 cells with 1% bovine serum albumin, IC50 = 0.008 μM. | 12646019 | |||
| 3T3 | Function assay | Inhibition of PDGF-induced BrdU incorporation in 3T3 cells, IC50 = 0.008 μM. | 12646019 | |||
| MV4-11 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MV4-11 cells after 72 hrs by MTT assay, IC50 = 0.009 μM. | 20570526 | ||
| MV4-11 | Cytotoxicity assay | Cytotoxicity against human MV4-11 cells by MTT assay, IC50 = 0.009 μM. | 20833039 | |||
| BAF3 | Function assay | 72 hrs | Inhibition of Tel-fused cKIT V559D mutant (unknown origin) transfected in mouse BAF3 cells assessed as decrease in cell proliferation after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 0.009 μM. | 27077705 | ||
| endothelial precursor cells | Function assay | Inhibition of endothelial cord area formation in endothelial precursor cells by CD31 cord area detection based phenotypic drug discovery based assay, IC50 = 0.0096 μM. | 22409666 | |||
| MDA-MB-435 | Cytotoxicity assay | Cytotoxicity against human MDA-MB-435 cells, IC50 = 0.0097 μM. | 24890652 | |||
| RS4-11 | Function assay | 2 hrs | Inhibition of FLT3 autophosphorylation in human RS4-11 cells after 2 hrs by electrochemiluminescence assay, IC50 = 0.0099 μM. | 19654408 | ||
| MV4-11 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MV4-11 cells after 72 hrs by Celltiter-Glo luminescent cell viability assay, IC50 = 0.01 μM. | 29935772 | ||
| BA/F3 | Function assay | 72 hrs | Inhibition of Kit exon 9 AY502 to 503 insertion and V654 mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.012 μM. | 30204441 | ||
| Kasumi-1 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human Kasumi-1 cells after 72 hrs by MTT assay, IC50 = 0.016 μM. | 20570526 | ||
| RS4-11 | Function assay | Inhibition of FLT3 autophosphorylation in human RS4-11 cells by Western blot analysis, IC50 = 0.016 μM. | 20833039 | |||
| 4T1 | Cytotoxicity assay | Cytotoxicity against mouse triple negative 4T1 cells, IC50 = 0.016 μM. | 24890652 | |||
| MOLM13 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MOLM13 cells assessed as cell viability after 48 hrs by MTT assay, IC50 = 0.0177 μM. | 25089810 | ||
| NIH3T3 | Function assay | 1 hr | Inhibitory concentration against human KDR kinase expressed in NIH3T3 cells with 4 uM Biotin-Ahx-AEEEYFFLFA-amide at ambient temperature for 1 hr, IC50 = 0.018 μM. | 16162008 | ||
| U251 | Function assay | Inhibition of VEGFR2 in human U251 cells by phosphotyrosine ELISA, IC50 = 0.0189 μM. | 24900865 | |||
| U251 | Function assay | Inhibition of VEGFR2 in human U251 cells by phosphotyrosine ELISA assay, IC50 = 0.0189 μM. | 24890652 | |||
| U251 | Function assay | 60 mins | Inhibition of VEGF-induced VEGFR2 activation in human U251 cells pretreated for 60 mins followed by VEGF addition and measured after 10 mins by ELISA, IC50 = 0.0189 μM. | 30098869 | ||
| U251 | Function assay | 60 mins | Inhibition of VEGFR-2 in human U251 cells assessed as reduction in VEGF induced phosphorylation preincubated for 60 mins followed by VEGF addition measured after 10 mins by ELISA method, IC50 = 0.0189 μM. | 27894589 | ||
| U251 | Function assay | 60 min | Inhibition of VEGFR2 in human U251 cells compound pretreated for 60 min before VEGF stimulation for 10 mins by phosphotyrosine ELISA cytoblot method, IC50 = 0.0189 μM. | 25882519 | ||
| BAF3 | Function assay | 72 hrs | Inhibition of Tel-fused cKIT V654A mutant (unknown origin) transfected in mouse BAF3 cells assessed as decrease in cell proliferation after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 0.02 μM. | 27077705 | ||
| MDA-MB-231 | Cytotoxicity assay | Cytotoxicity against human triple negative MDA-MB-231 cells, IC50 = 0.0223 μM. | 24890652 | |||
| BAF3 | Function assay | 72 hrs | Inhibition of Tel-fused cKIT L567P mutant (unknown origin) transfected in mouse BAF3 cells assessed as decrease in cell proliferation after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 0.023 μM. | 27077705 | ||
| BA/F3 | Function assay | 72 hrs | Inhibition of Kit exon 11 deletion (560 to 578 residues) mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.024 μM. | 30204441 | ||
| MV4-11 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MV4-11 cells assessed as cell viability after 48 hrs by MTT assay, IC50 = 0.0243 μM. | 25089810 | ||
| BA/F3 | Function assay | 72 hrs | Inhibition of Kit V560D mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.027 μM. | 30204441 | ||
| MCF7 | Cytotoxicity assay | Cytotoxicity against human ER-positive MCF7 cells, IC50 = 0.0271 μM. | 24890652 | |||
| MCF7 | Cytotoxicity assay | Cytotoxicity against human taxoid-resistant MCF7 cells, IC50 = 0.0293 μM. | 24890652 | |||
| primary leukemia cells | Cytotoxicity assay | 72 hrs | Cytotoxicity against human primary leukemia cells isolated from acute myeloid leukemia patient expressing FLT3-ITD mutation assessed as cell viability after 72 hrs by luciferase assay, IC50 = 0.03 μM. | 22221201 | ||
| primary leukemia cells | Cytotoxicity assay | 72 hrs | Cytotoxicity against human primary leukemia cells isolated from acute myeloid leukemia patient expressing wild type FLT3 assessed as cell viability after 72 hrs by luciferase assay, IC50 = 0.03 μM. | 22221201 | ||
| BA/F3 | Function assay | 72 hrs | Inhibition of TEL-fused PDGFRalpha (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.03 μM. | 30204441 | ||
| BA/F3 | Function assay | 72 hrs | Inhibition of KDR (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.033 μM. | 30204441 | ||
| RS4-11 | Function assay | 2 hrs | Inhibition of FLT3 ITD mutant autophosphorylation in human RS4-11 cells after 2 hrs by electrochemiluminescence assay, IC50 = 0.034 μM. | 19654408 | ||
| HL60 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60 cells assessed as cell viability after 48 hrs by MTT assay, IC50 = 0.0368 μM. | 25089810 | ||
| MV4-11 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MV4-11 cells harboring FLT3 ITD mutant after 72 hrs by MTS assay, IC50 = 0.0385 μM. | 28038328 | ||
| TT | Antiproliferative assay | 72 hrs | Antiproliferative activity against human TT cells pretreated for 72 hrs followed by compound-washout measured after 72 hrs by MTT assay, IC50 = 0.04 μM. | 24904961 | ||
| GISTT1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human GISTT1 cells assessed as cell growth inhibition after 72 hrs by CellTiterGlo assay, GI50 = 0.04 μM. | 28991465 | ||
| GISTT1 | Antiproliferative assay | 72 hrs | Antiproliferative activity against cKIT dependent human GISTT1 cells assessed as reduction in cell viability after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 0.041 μM. | 27077705 | ||
| THP1 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human THP1 cells assessed as cell viability after 48 hrs by MTT assay, IC50 = 0.0457 μM. | 25089810 | ||
| 3T3 | Function assay | Inhibition of Vascular endothelial growth factor receptor in 3T3 cells, IC50 = 0.05 μM. | 12646019 | |||
| BAF3 | Function assay | 72 hrs | Inhibition of Tel-fused cKIT T670I/V559D double mutant (unknown origin) transfected in mouse BAF3 cells assessed as decrease in cell proliferation after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 0.053 μM. | 27077705 | ||
| BAF3 | Function assay | 72 hrs | Inhibition of Tel-fused cKIT (unknown origin) transfected in mouse BAF3 cells assessed as decrease in cell proliferation after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 0.078 μM. | 27077705 | ||
| 3T3 | Function assay | Inhibition of PDGF-induced BrdU incorporation in 3T3 cells with 5% bovine serum albumin, IC50 = 0.083 μM. | 12646019 | |||
| SH-SY5Y | Function assay | Inhibition of PDGFRbeta in human SH-SY5Y cells by phosphotyrosine ELISA assay, IC50 = 0.0831 μM. | 24890652 | |||
| SH-SY5Y | Function assay | 60 mins | Inhibition of PDGF-BB-induced PDGFRbeta activation in human SH-SY5Y cells pretreated for 60 mins followed by PDGF-BB addition and measured after 10 mins by ELISA, IC50 = 0.0831 μM. | 30098869 | ||
| SF539 | Function assay | 60 mins | Inhibition of PDGFR-beta in human SF539 cells assessed as reduction in PDGF-BB induced phosphorylation preincubated for 60 mins followed by PDGF-BB addition measured after 10 mins by ELISA method, IC50 = 0.0831 μM. | 27894589 | ||
| U251 | Function assay | 60 min | Inhibition of PDGFR-beta in human U251 cells compound pretreated for 60 min before PDGF-BB stimulation for 10 mins by phosphotyrosine ELISA cytoblot method, IC50 = 0.0831 μM. | 25882519 | ||
| GISTT1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human GISTT1 cells harboring KIT T670I mutant assessed as cell growth inhibition after 72 hrs by CellTiterGlo assay, GI50 = 0.09 μM. | 28991465 | ||
| GIST430 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human GIST430 cells harboring KIT V654A mutant assessed as cell growth inhibition after 72 hrs by CellTiterGlo assay, GI50 = 0.098 μM. | 28991465 | ||
| HAEC | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HAEC cells expressing VEGFR after 72 hrs by MTT assay, IC50 = 0.1 μM. | 22444679 | ||
| GIST882 | Antiproliferative assay | 72 hrs | Antiproliferative activity against cKIT dependent human GIST882 cells assessed as reduction in cell viability after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 0.11 μM. | 27077705 | ||
| BA/F3 | Function assay | 72 hrs | Inhibition of TEL-fused PDGFRbeta (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.114 μM. | 30204441 | ||
| FDC-P1 | Function assay | 48 hrs | Inhibition of human FMS expressed in growth factor dependent mouse FDC-P1 cells assessed as inhibition of FMS-mediated cell proliferation in presence human CSF1 after 48 hrs by resazurin dye reduction assay, IC50 = 0.155 μM. | 20156689 | ||
| A431 | Function assay | Inhibition of EGFR in human A431 cells by phosphotyrosine ELISA assay, IC50 = 0.1721 μM. | 24890652 | |||
| A431 | Function assay | 60 mins | Inhibition of EGF-induced EGFR activation in human A431 cells pretreated for 60 mins followed by EGF addition and measured after 10 mins by ELISA, IC50 = 0.1721 μM. | 30098869 | ||
| A431 | Function assay | 60 mins | Inhibition of EGFR in human A431 cells compound pretreated for 60 min before EGF stimulation for 10 mins by phosphotyrosine ELISA cytoblot method, IC50 = 0.1721 μM. | 25882519 | ||
| Sf9 | Function assay | Inhibition of GST-tagged VEGFR expressed in Sf9 cells, IC50 = 0.185 μM. | 19854051 | |||
| BA/F3 | Function assay | 72 hrs | Inhibition of Kit exon 11 deletion (557 to 558 residues) and D820A mutant and D820A mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.214 μM. | 30204441 | ||
| BaPTC2 | Growth inhibition assay | 72 hrs | Growth inhibition of mouse BaPTC2 cells assessed as incorporation of [3H]thymidine incorporation after 72 hrs by liquid scintillation counting, IC50 = 0.22 μM. | 20117004 | ||
| BA/F3 | Function assay | 72 hrs | Inhibition of Kit exon 11 deletion (557 to 558 residues) and N822K mutant and Y823D mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.271 μM. | 30204441 | ||
| HT-29 | Cytotoxicity assay | 72 hrs | Cytotoxicity against VEGFR expressing human HT-29 cells after 72 hrs by MTT assay, IC50 = 0.33 μM. | 23131541 | ||
| HT-29 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HT-29 cells expressing VEGFR after 72 hrs by MTT assay, IC50 = 0.33 μM. | 22444679 | ||
| BA/F3 | Function assay | 72 hrs | Inhibition of Kit exon 11 deletion (557 to 558 residues) and D816H mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.398 μM. | 30204441 | ||
| BA/F3 | Function assay | 72 hrs | Inhibition of Kit exon 11 deletion (557 to 558 residues) and A829P mutant and Y823D mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.43 μM. | 30204441 | ||
| THP1 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human THP1 cells after 72 hrs by MTT assay, IC50 = 0.5 μM. | 20570526 | ||
| CHO | Function assay | Inhibition of human ERG expressed in CHO cells by Qpatch assay, IC50 = 0.5 μM. | 28038328 | |||
| BAF3 | Function assay | 72 hrs | Inhibition of Tel-fused cKIT N822K mutant (unknown origin) transfected in mouse BAF3 cells assessed as decrease in cell proliferation after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 0.51 μM. | 27077705 | ||
| BA/F3 | Function assay | 72 hrs | Inhibition of Kit exon 9 AY502 to 503 insertion and D816 mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.584 μM. | 30204441 | ||
| CAKI-1 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human CAKI-1 cells after 48 hrs by SRB assay, GI50 = 0.63 μM. | 22560627 | ||
| BA/F3 | Function assay | 72 hrs | Inhibition of PDGFRalpha V561D/D842V mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.631 μM. | 30204441 | ||
| BA/F3 | Function assay | 72 hrs | Inhibition of Kit D816V mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.638 μM. | 30204441 | ||
| BA/F3 | Function assay | 72 hrs | Inhibition of Kit V560D/D816H mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.696 μM. | 30204441 | ||
| BA/F3 | Function assay | 72 hrs | Inhibition of Kit exon 11 deletion (557 to 558 residues) and Y823D mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.782 μM. | 30204441 | ||
| NCI-H3122 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human NCI-H3122 cells after 72 hrs by MTT assay, IC50 = 0.83 μM. | 24904961 | ||
| Rec1 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human Rec1 cells assessed as reduction in cell viability after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 0.87 μM. | 27077705 | ||
| RS4:11 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human RS4:11 cells after 72 hrs by MTT assay, IC50 = 1 μM. | 20570526 | ||
| K562 | Antiproliferative assay | 72 hrs | Antiproliferative activity against BCR-ABL dependent human K562 cells assessed as reduction in cell viability after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 1 μM. | 27077705 | ||
| NCI-H526 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human NCI-H526 cells after 72 hrs by MTT assay, IC50 = 1.01 μM. | 24904961 | ||
| primary leukemia cells | Cytotoxicity assay | 72 hrs | Cytotoxicity against human primary leukemia cells isolated from acute myeloid leukemia patient expressing FLT3-D835Y mutation assessed as cell viability after 72 hrs by luciferase assay, IC50 = 1.1 μM. | 22221201 | ||
| KU812 | Antiproliferative assay | 72 hrs | Antiproliferative activity against BCR-ABL dependent human KU812 cells assessed as reduction in cell viability after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 1.1 μM. | 27077705 | ||
| BA/F3 | Growth inhibition assay | 72 hrs | Growth inhibition of mouse BA/F3 cells assessed as incorporation of [3H]thymidine incorporation after 72 hrs by liquid scintillation counting, IC50 = 1.2 μM. | 20117004 | ||
| MEG01 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human BCR-ABL dependent MEG01 cells assessed as reduction in cell viability after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 1.2 μM. | 27077705 | ||
| Sf9 | Function assay | Inhibition of human recombinant His-tagged RET expressed in Sf9 insect cells, IC50 = 1.3 μM. | 20117004 | |||
| HT-29 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HT-29 cells assessed as cell growth inhibition after 72 hrs by MTT assay, IC50 = 1.3 μM. | 21621880 | ||
| SW620 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human SW620 cells after 48 hrs by SRB assay, GI50 = 1.3 μM. | 22560627 | ||
| IM9 | Antitumor assay | Antitumor activity against human IM9 cells, IC50 = 1.35 μM. | 21450463 | |||
| BAF3 | Function assay | 72 hrs | Inhibition of Tel-fused cKIT D816V mutant (unknown origin) transfected in mouse BAF3 cells assessed as decrease in cell proliferation after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 1.38 μM. | 27077705 | ||
| HT-29 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HT-29 cells after 72 hrs by MTT assay, IC50 = 1.47 μM. | 23999040 | ||
| HT-29 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HT-29 cells after 48 hrs by SRB assay, GI50 = 1.6 μM. | 22560627 | ||
| HT-29 | Cytotoxicity assay | Cytotoxicity against human HT-29 cells by MTT assay, IC50 = 1.7 μM. | 26920800 | |||
| U937 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human U937 cells assessed as reduction in cell viability after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 1.7 μM. | 27077705 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells after 48 hrs by SRB assay, GI50 = 2 μM. | 22560627 | ||
| MDA-MB-435 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MDA-MB-435 cells after 48 hrs by SRB assay, GI50 = 2 μM. | 22560627 | ||
| GIST48B | Antiproliferative assay | 72 hrs | Antiproliferative activity against human GIST48B cells assessed as reduction in cell viability after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 2.01 μM. | 27077705 | ||
| CHO | Antiproliferative assay | 72 hrs | Antiproliferative activity against CHO cells assessed as reduction in cell viability after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 2.03 μM. | 27077705 | ||
| KB-3-1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human P-gp-negative KB-3-1 cells after 72 hrs by MTT assay, IC50 = 2.3 μM. | 19397322 | ||
| K562 | Antitumor assay | Antitumor activity against human K562 cells, IC50 = 2.41 μM. | 21450463 | |||
| A549 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A549 cells assessed as growth inhibition after 72 hrs by MTT assay, IC50 = 2.44 μM. | 23602441 | ||
| T24 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human T24 cells after 72 hrs by MTT assay, IC50 = 2.44 μM. | 23999040 | ||
| CHL | Antiproliferative assay | 72 hrs | Antiproliferative activity against CHL cells assessed as reduction in cell viability after 72 hrs by celltiter-glo/CCK8 assay, GI50 = 2.48 μM. | 27077705 | ||
| ACHN | Cytotoxicity assay | 6 days | Cytotoxicity against human ACHN cells assessed as inhibition of cell viability after 6 days by MTT assay, IC50 = 2.5 μM. | 23360104 | ||
| Bel7402 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human Bel7402 cells after 72 hrs by MTT assay, IC50 = 2.67 μM. | 24904961 | ||
| H460 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human H460 cells assessed as cell growth inhibition after 72 hrs by MTT assay, IC50 = 2.7 μM. | 21621880 | ||
| BAF3 | Antiproliferative assay | Antiproliferative activity against mouse BAF3 cells, GI50 = 2.78 μM. | 27077705 | |||
| HuH7 | Antiproliferative assay | Antiproliferative activity against human HuH7 cells by MTT assay, IC50 = 3.03 μM. | 30059803 | |||
| HuH7 | Growth inhibition assay | 48 hrs | Growth inhibition of human HuH7 cells after 48 hrs by MTT assay, IC50 = 3.03 μM. | 27956344 | ||
| OVCAR3 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human OVCAR3 cells after 48 hrs by SRB assay, GI50 = 3.2 μM. | 22560627 | ||
| HL60 | Antitumor assay | Antitumor activity against human HL60 cells, IC50 = 3.53 μM. | 21450463 | |||
| BxPC3 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human BxPC3 cells after 72 hrs by MTT assay, IC50 = 3.63 μM. | 23999040 | ||
| MDA-MB-231 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MDA-MB-231 cells assessed as cell growth inhibition after 72 hrs by MTT assay, IC50 = 3.7 μM. | 21621880 | ||
| HepG2 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HepG2 cells after 72 hrs by MTT assay, IC50 = 3.81 μM. | 20570526 | ||
| UACC257 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human UACC257 cells after 48 hrs by SRB assay, GI50 = 4 μM. | 22560627 | ||
| RCC4 | Anticancer assay | 48 hrs | Anticancer activity against human RCC4 cells after 48 hrs by XTT assay, IC50 = 4 μM. | 28325600 | ||
| KBV1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human P-glycoprotein-expressing KBV1 cells after 72 hrs by MTT assay, IC50 = 4.1 μM. | 19397322 | ||
| A498 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human A498 cells after 72 hrs by MTT assay, IC50 = 4.3 μM. | 23489626 | ||
| NCI-H460 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human NCI-H460 cells after 72 hrs by MTT assay, IC50 = 4.31 μM. | 24904961 | ||
| MDA-MB-231 | Antitumor assay | Antitumor activity against human MDA-MB-231 cells, IC50 = 4.51 μM. | 21450463 | |||
| L02 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human L02 cells after 72 hrs by CCK-8 assay, IC50 = 4.62 μM. | 27643639 | ||
| HT-29 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HT-29 cells assessed as inhibition of cell viability after 48 hrs by SRB assay, IC50 = 4.7 μM. | 26009164 | ||
| HuH7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HuH7 cells after 48 hrs by MTT assay, IC50 = 4.707 μM. | 29057042 | ||
| HCT116 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HCT116 cells assessed as growth inhibition after 72 hrs by MTT assay, IC50 = 4.71 μM. | 23602441 | ||
| BGC | Antiproliferative assay | 72 hrs | Antiproliferative activity against human BGC cells after 72 hrs by MTT assay, IC50 = 4.78 μM. | 23999040 | ||
| BA/F3 | Cytotoxicity assay | 72 hrs | Cytotoxicity in mouse parental BA/F3 cells incubated for 72 hrs by MTS assay, GI50 = 4.789 μM. | 30204441 | ||
| HL60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60 cells after 72 hrs by CCK-8 assay, IC50 = 4.95 μM. | 27643639 | ||
| A375 | Toxicity against | 72 hrs | Toxicity against human A375 cells after 72 hrs by cell titer-blue assay, IC50 = 5.4 μM. | 19654408 | ||
| HepG2 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HepG2 cells after 72 hrs by MTT assay, IC50 = 5.61 μM. | 23999040 | ||
| HCT116 | Antiproliferative assay | Antiproliferative activity against human HCT116 cells by MTT assay, IC50 = 5.62 μM. | 30059803 | |||
| HCT116 | Growth inhibition assay | 48 hrs | Growth inhibition of human HCT116 cells after 48 hrs by MTT assay, IC50 = 5.62 μM. | 27956344 | ||
| ACHN | Cytotoxicity assay | 72 hrs | Cytotoxicity against human ACHN cells after 72 hrs by MTT assay, IC50 = 5.7 μM. | 23489626 | ||
| HK2 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HK2 cells after 72 hrs by MTT assay, IC50 = 5.85 μM. | 24904961 | ||
| GISTT1 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human GISTT1 cells harboring KIT D816E mutant assessed as cell growth inhibition after 72 hrs by CellTiterGlo assay, GI50 = 5.88 μM. | 28991465 | ||
| MDA-MB-231 | Anticancer assay | 48 hrs | Anticancer activity against human MDA-MB-231 cells after 48 hrs by XTT assay, IC50 = 6 μM. | 28325600 | ||
| HCT116 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HCT116 cells after 48 hrs by MTT assay, IC50 = 6.1 μM. | 22019188 | ||
| HEK293 | Function assay | 2 mins | Inhibition of human OCT1 expressed in HEK293 cells assessed as decrease in uptake of ASP+ after 2 mins by fluorescence assay, IC50 = 6.1 μM. | 28230985 | ||
| SW620 | Antiproliferative assay | Antiproliferative activity against human SW620 cells by MTT assay, IC50 = 6.25 μM. | 30059803 | |||
| SW620 | Growth inhibition assay | 48 hrs | Growth inhibition of human SW620 cells after 48 hrs by MTT assay, IC50 = 6.25 μM. | 27956344 | ||
| MCF7 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MCF7 cells assessed as growth inhibition after 72 hrs by MTT assay, IC50 = 6.29 μM. | 23602441 | ||
| TK10 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human TK10 cells after 48 hrs by SRB assay, GI50 = 6.3 μM. | 22560627 | ||
| SMMC7721 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SMMC7721 cells assessed as cell growth inhibition after 72 hrs by MTT assay, IC50 = 6.47 μM. | 21621880 | ||
| A549 | Antitumor assay | Antitumor activity against human A549 cells, IC50 = 6.61 μM. | 21450463 | |||
| MDA-MB-231 | Cytotoxicity assay | 24 hrs | Cytotoxicity in human MDA-MB-231 cells assessed as reduction in cell viability incubated for 24 hrs by MTT assay, IC50 = 6.98 μM. | 28760313 | ||
| MDA-MB-231 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MDA-MB-231 cells after 48 hrs by MTT assay, IC50 = 7.4 μM. | 27128173 | ||
| MDA-MB-231 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MDA-MB-231 cells assessed as cell growth inhibition after 48 hrs by MTT assay, IC50 = 7.4 μM. | 27639369 | ||
| MDA-MB-231 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MDA-MB-231 cells assessed as reduction in cell viability after 48 hrs by MTT assay, IC50 = 7.44 μM. | 27448916 | ||
| HepG2 | Antiproliferative assay | Antiproliferative activity against human HepG2 cells by MTT assay, IC50 = 7.78 μM. | 30059803 | |||
| EKVX | Antiproliferative assay | 48 hrs | Antiproliferative activity against human EKVX cells after 48 hrs by SRB assay, GI50 = 7.9 μM. | 22560627 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells assessed as inhibition of cell viability after 48 hrs by SRB assay, IC50 = 7.93 μM. | 26009164 | ||
| CHO | Function assay | Inhibition of rapid delayed inward rectifying potassium current (IKr) in Chinese hamster ovary (CHO) cells stable expressing hERG measured using IonWorks Barracuda automated patch clamp platform, IC50 = 7.94328 μM. | 25087753 | |||
| PA1 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human PA1 cells assessed as decrease in cell viability after 48 hrs by MTT assay, IC50 = 7.97 μM. | 27210438 | ||
| A498 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human A498 cells after 48 hrs by MTT assay, IC50 = 7.981 μM. | 29057042 | ||
| COLO205 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human COLO205 cells after 24 hrs by MTT assay, IC50 = 8 μM. | 28756024 | ||
| SVEC4-10 | Cytotoxicity assay | 24 hrs | Cytotoxicity against mouse SVEC4-10 cells assessed as cell viability after 24 hrs by MTS assay, IC50 = 8 μM. | 26912111 | ||
| WI38 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human WI38 cells assessed as cell growth inhibition after 72 hrs by MTT assay, IC50 = 8.56 μM. | 21621880 | ||
| A549 | Cytotoxicity assay | Cytotoxicity against human A549 cells by MTT assay, IC50 = 9.1 μM. | 26920800 | |||
| SKOV3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human SKOV3 cells assessed as inhibition of cell viability after 48 hrs by SRB assay, IC50 = 9.21 μM. | 26009164 | ||
| MDA-MB-435 | Cytotoxicity assay | Cytotoxicity against human MDA-MB-435 cells by MTT assay, IC50 = 9.3 μM. | 26920800 | |||
| SNB19 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human SNB19 cells after 48 hrs by SRB assay, GI50 = 10 μM. | 22560627 | ||
| CHO K1 | Function assay | Inhibition of rapid delayed inward rectifying potassium current (IKr) in Chinese hamster ovary (CHO) K1 cells stably expressing hERG measured using IonWorks Quattro automated patch clamp platform, IC50 = 10 μM. | 25087753 | |||
| NCI-H727 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human NCI-H727 cells after 48 hrs by MTT assay, IC50 = 10.12 μM. | 29057042 | ||
| HeLa | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HeLa cells assessed as cell growth inhibition after 48 hrs by MTT assay, IC50 = 10.4 μM. | 27639369 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 10.5 μM. | 25064347 | ||
| PC9 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human PC9 cells after 48 hrs by MTT assay, IC50 = 10.97 μM. | 22483608 | ||
| Bel7402 | Photocytotoxicity assay | 5 hrs | Photocytotoxicity against human Bel7402 cells assessed as cell viability preincubated for 5 hrs with cells followed by UVA-1 irradiation at 3.0 mW/cm'2 for 20 mins measured after 48 hrs by CCK8 assay, EC50 = 11 μM. | 26584085 | ||
| A375 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A375 cells after 48 hrs by MTT assay, IC50 = 11.58 μM. | 22483608 | ||
| SF539 | Function assay | 10 mins | Inhibition of PDGFRbeta phosphorylation in human SF539 cells after 10 mins by FLISA, IC50 = 12.2 μM. | 20403700 | ||
| A431 | Function assay | Inhibition of PDGFRbeta expressed in human A431 cells, IC50 = 12.2 μM. | 20558072 | |||
| SF-539 | Function assay | 60 mins | Inhibition of PDGFRbeta tyrosine kinase activity in PDGF-BB-stimulated human SF-539 cells after 60 mins by ELISA, IC50 = 12.2 μM. | 22204741 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by MTT assay, IC50 = 12.35 μM. | 22483608 | ||
| PC3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human PC3 cells assessed as cell growth inhibition after 48 hrs by MTT assay, IC50 = 12.6 μM. | 27639369 | ||
| LNCAP | Cytotoxicity assay | 48 hrs | Cytotoxicity against human LNCAP cells assessed as decrease in cell viability after 48 hrs by MTT assay, IC50 = 12.61 μM. | 27210438 | ||
| PANC1 | Photocytotoxicity assay | 5 hrs | Photocytotoxicity against human PANC1 cells assessed as cell viability preincubated for 5 hrs with cells followed by UVA-1 irradiation at 3.0 mW/cm'2 for 20 mins measured after 48 hrs by CCK8 assay, EC50 = 13 μM. | 26584085 | ||
| HepG2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HepG2 cells after 48 hrs by MTT assay, IC50 = 13.24 μM. | 22483608 | ||
| endothelial precursor cells | Function assay | Inhibition of cell migration in endothelial precursor cells by Oris cell migration kit based phenotypic drug discovery based assay, IC50 = 13.6 μM. | 22409666 | |||
| HepG2 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HepG2 cells after 48 hrs by MTT assay, IC50 = 13.6 μM. | 25064347 | ||
| K562 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human K562 cells after 24 hrs by MTT assay, IC50 = 14 μM. | 28756024 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells assessed as reduction in cell viability after 48 hrs by MTT assay, IC50 = 14.4 μM. | 27448916 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells assessed as cell growth inhibition after 48 hrs by MTT assay, IC50 = 14.4 μM. | 27639369 | ||
| PANC1 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human PANC1 cells assessed as inhibition of cell viability after 48 hrs by SRB assay, IC50 = 14.94 μM. | 26009164 | ||
| HepG2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HepG2 cells after 48 hrs by MTT assay, IC50 = 15.4 μM. | 22019188 | ||
| HL60 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HL60 cells after 48 hrs by MTT assay, IC50 = 15.5 μM. | 22019188 | ||
| BT549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human BT549 cells after 48 hrs by MTT assay, IC50 = 15.5 μM. | 27128173 | ||
| BT549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human BT549 cells assessed as cell growth inhibition after 48 hrs by MTT assay, IC50 = 15.5 μM. | 27639369 | ||
| BT549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human BT549 cells assessed as reduction in cell viability after 48 hrs by MTT assay, IC50 = 15.54 μM. | 27448916 | ||
| CHO K1 | Function assay | Inhibition of fast sodium current (INa) in Chinese Hamster Ovary (CHO) K1 cells transfected with human Nav1.5 measured using IonWorks Quattro automated patch clamp platform, IC50 = 15.8489 μM. | 25087753 | |||
| HepG2 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HepG2 cells after 48 hrs by MTT assay, IC50 = 16.06 μM. | 28057423 | ||
| HepG2 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HepG2 cells assessed as inhibition of cell viability after 48 hrs by SRB assay, IC50 = 16.06 μM. | 26009164 | ||
| DU145 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human DU145 cells assessed as cell growth inhibition after 48 hrs by MTT assay, IC50 = 16.3 μM. | 27639369 | ||
| DU145 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human DU145 cells assessed as reduction in cell viability after 48 hrs by MTT assay, IC50 = 16.38 μM. | 27448916 | ||
| DU145 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human DU145 cells after 48 hrs by MTT assay, IC50 = 16.4 μM. | 27128173 | ||
| HCT116 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human HCT116 cells after 48 hrs by MTT assay, IC50 = 18.35 μM. | 22483608 | ||
| A549 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human A549 cells after 48 hrs by MTT assay, IC50 = 18.5 μM. | 22019188 | ||
| U251 | Function assay | 10 mins | Inhibition of VEGFR2 phosphorylation in human U251 cells after 10 mins by FLISA, IC50 = 18.9 μM. | 20403700 | ||
| A431 | Function assay | Inhibition of VEGFR2 expressed in human A431 cells, IC50 = 18.9 μM. | 20558072 | |||
| U251 | Function assay | 60 mins | Inhibition of VEGFR2 tyrosine kinase activity in VEGF-stimulated human U251 cells after 60 mins by ELISA, IC50 = 18.9 μM. | 22204741 | ||
| U251 | Function assay | 60 mins | Inhibition of VEGF-induced VEGFR2 phosphorylation in human U251 cells overexpressing Flk-1 pretreated for 60 mins prior to VEGF addition measured after 10 mins by phosphotyrosine ELISA cytoblot analysis, IC50 = 18.9 μM. | 23375090 | ||
| U251 | Function assay | 60 mins | Inhibition of VEGFR2 in human U251 cells assessed as inhibition of VEGF-induced tyrosine phosphorylation incubated for 60 mins prior to VEGF-activation measured 10 mins by ELISA, IC50 = 18.9 μM. | 23434139 | ||
| U251 | Function assay | 60 mins | Inhibition of VEGFR2 in human U251 cells pretreated for 60 mins measured after 1 hr by ELISA, IC50 = 18.9 μM. | 22739090 | ||
| PC3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human PC3 cells after 48 hrs by MTT assay, IC50 = 19.6 μM. | 27128173 | ||
| PC3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human PC3 cells assessed as reduction in cell viability after 48 hrs by MTT assay, IC50 = 19.6 μM. | 27448916 | ||
| LO2 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human LO2 cells after 72 hrs by MTT assay, IC50 = 19.93 μM. | 24904961 | ||
| K562 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human K562 cells after 48 hrs by MTT assay, IC50 = 21.9 μM. | 22019188 | ||
| RWPE1 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human RWPE1 cells assessed as decrease in cell viability after 48 hrs by MTT assay, IC50 = 23 μM. | 27210438 | ||
| RWPE1 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human RWPE1 cells after 48 hrs by MTT assay, IC50 = 23 μM. | 27128173 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells assessed as decrease in cell viability after 48 hrs by MTT assay, IC50 = 23.76 μM. | 27210438 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells assessed as reduction in cell viability after 48 hrs by MTT assay, IC50 = 23.8 μM. | 27448916 | ||
| PC3 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human PC3 cells after 48 hrs by MTT assay, IC50 = 25.1 μM. | 22019188 | ||
| RWPE1 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human RWPE1 cells assessed as cell growth inhibition after 48 hrs by MTT assay, IC50 = 25.2 μM. | 27639369 | ||
| MCF7 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MCF7 cells after 48 hrs by MTT assay, IC50 = 25.41 μM. | 28057423 | ||
| MCF7 | Cytotoxicity assay | 48 hrs | Cytotoxicity against human MCF7 cells assessed as inhibition of cell viability after 48 hrs by SRB assay, IC50 = 25.41 μM. | 26009164 | ||
| MDA-MB-231 | Cytotoxicity assay | 24 hrs | Cytotoxicity against human MDA-MB-231 cells after 24 hrs by MTT assay, IC50 = 32 μM. | 28756024 | ||
| CHO | Function assay | Inhibition of Cav1.2 current measured using QPatch automatic path clamp system in CHO cells expressing Cav1.2, beta-2 and alpha-2/delta-1 subunits, IC50 = 33.4 μM. | 23812503 | |||
| HEK293 | Function assay | Inhibition of fast sodium current (INa) in HEK293 cells transfected with human Nav1.5 measured using IonWorks Quattro automated patch clamp platform, IC50 = 39.8107 μM. | 25087753 | |||
| H4 | Toxicity assay | 10 uM | Toxicity in human H4 cells at 10 uM, Activity = 40 μM. | 20350806 | ||
| MV4-11 | Antitumor assay | 30 mg/kg | 28 days | Antitumor activity against human MV4-11 cells xenografted in athymic nude mouse assessed as suppression of tumor volume at 30 mg/kg, po QD for 28 days measured after dosing period | 19754199 | |
| MV4-11 | Antitumor assay | 30 mg/kg | 28 days | Antitumor activity against human MV4-11 cells xenografted in athymic nude mouse assessed as tumor growth regression at 30 mg/kg, po QD for 28 days measured after dosing period | 19754199 | |
| MV4-11 | Antitumor assay | 30 mg/kg | 28 days | Antitumor activity against human MV4-11 cells xenografted in athymic nude mouse assessed as inhibition of tumor growth at 30 mg/kg, po QD for 28 days measured during first 14 days of dosing | 19754199 | |
| SK-RC-52 | Antitumor assay | 20 mg/kg | Antitumor activity against human SK-RC-52 cells xenografted BALB/c nu/nu mouse assessed as inhibition of tumor growth at 20 mg/kg, ip | 19615903 | ||
| MV411 | Antitumor assay | 10 mg/kg | 28 days | Antitumor activity against human MV411 cells in xenografted athymic nude mouse model assessed as decrease in tumor size at 10 mg/kg, po qd for 28 days measured after 29 to 36 days | 19646870 | |
| MV411 | Antitumor assay | 30 mg/kg | 28 days | Antitumor activity against human MV411 cells in xenografted athymic nude mouse model assessed as decrease in tumor size at 30 mg/kg, po qd for 28 days | 19646870 | |
| MV4-11 | Antitumor assay | 10 mg/kg | Antitumor activity against human MV4-11 cells xenografted mouse model assessed as inhibition of tumor growth at at 10 mg/kg, po | 19654408 | ||
| HEK293 | Function assay | 100 nM | 1 hr | Inhibition of VEGF-induced autophosphorylation of tyrosine 1175 residue on VEGFR2 expressed in HEK293 cells at 100 nM after 1 hr by ELISA | 21885287 | |
| Calu6 | Antitumor assay | 80 mg/kg | 11 days | Antitumor activity against human Calu6 cells xenografted in BALB/c mouse assessed as tumor growth inhibition at 80 mg/kg, po qd for 11 days | 22269278 | |
| BA/F3 | Antitumor assay | 0.05 to 5 mg/kg | 100 days | Antitumor activity against mouse BA/F3 cells expressing FLT3-ITD mutant xenografted in BALB/c mouse myeloproliferative model assessed as disease related death at 0.05 to 5 mg/kg, po measured after 100 days | 22221201 | |
| MDA-MB-435 | Antiangiogenic assay | 20 mg/kg | 2 days | Antiangiogenic activity in human MDA-MB-435 cells xenografted in chicken embryo chorioallantoic membrane assessed as inhibition of blood vessel formation at 20 mg/kg measured after 2 days | 23664876 | |
| MDA-MB-435 | Antitumor assay | 20 mg/kg | 2 days | Antitumor activity against human MDA-MB-435 cells xenografted in chicken embryo chorioallantoic membrane assessed as inhibition of tumor volume at 20 mg/kg measured after 2 days | 23664876 | |
| U251 | Antitumor assay | 30 mg/kg | up to 34 days | Antitumor activity against human U251 cells xenografted into athymic NCr mouse assessed as decrease in tumor growth at 30 mg/kg twice weekly measured up to 34 days | 24900865 | |
| MDA-MB-435 | Antitumor assay | 30 mg/kg | up to 34 days | Antitumor activity against human MDA-MB-435 cells xenografted into flank of athymic BALBc/J mouse assessed as decrease in tumor growth at 30 mg/kg twice weekly measured up to 34 days | 24900865 | |
| MDA-MB-435 | Antitumor assay | 30 mg/kg | Antitumor activity against human MDA-MB-435 cells xenografted into flank of athymic BALBc/J mouse assessed as decrease in tumor vascular density at 30 mg/kg twice weekly by CD31/PECAM-1 immunohistochemical staining | 24900865 | ||
| NCI-H460 | Antitumor assay | 40 mg/kg | up to 21 days | Antitumor activity against human NCI-H460 cells xenografted in BALB/c nude mouse assessed as tumor growth inhibition at 40 mg/kg/day administered qd measured up to day 21 relative to control | 24904961 | |
| HT-29 | Antitumor assay | 40 mg/kg | up to 21 days | Antitumor activity against human HT-29 cells xenografted in BALB/c nude mouse assessed as tumor growth inhibition at 80 mg/kg/day administered qd measured up to day 21 relative to control | 24904961 | |
| MCF7 | Function assay | 4 uM | 24 hrs | Inhibition of VEGFR2 phosphorylation in human MCF7 cells at 4 uM incubated for 24 hrs by Western blot method | 28942113 | |
| HUVEC | Antiangiogenic assay | 10 uM | 5 hrs | Antiangiogenic activity in human HUVEC cells assessed as angiogenesis rate at 10 uM after 5 hrs by endothelial tube formation assay relative to control | 26912111 | |
| MCF7 | Function assay | 1 uM | 24 hrs | Inhibition of VEGFR2 phosphorylation in human MCF7 cells assessed as reduction in expression of phosphorylated ERK1/2 at 1 uM incubated for 24 hrs by Western blot analysis | 27176944 | |
| MCF7 | Function assay | 1 uM | 24 hrs | Inhibition of VEGFR2 phosphorylation in human MCF7 cells assessed as reduction in expression of phosphorylated Raf1 at 1 uM incubated for 24 hrs by Western blot analysis | 27176944 | |
| HCT116 | Antimigratory assay | 24 hrs | Antimigratory activity against human HCT116 cells assessed as inhibition of cell migration after 24 hrs by wound healing assay | 23153200 | ||
| U87MG | Function assay | 10 mg/kg | 3 days | In-vivo inhibition of angiogenesis in human U87MG cells xenografted in athymic nude Harlan mouse at 10 mg/kg oral gavage dosed twice daily for 3 days measured 3 days post dose by fluorescence microscopy | 22409666 | |
| EoL-1-cell | Growth inhibition assay | Inhibition of human EoL-1-cell cell growth in a cell viability assay, IC50 = 0.0000016 μM. | SANGER | |||
| MV-4-11 | Growth inhibition assay | Inhibition of human MV-4-11 cell growth in a cell viability assay, IC50 = 0.0002722 μM. | SANGER | |||
| NOS-1 | Growth inhibition assay | Inhibition of human NOS-1 cell growth in a cell viability assay, IC50 = 0.0153 μM. | SANGER | |||
| CGTH-W-1 | Growth inhibition assay | Inhibition of human CGTH-W-1 cell growth in a cell viability assay, IC50 = 0.03094 μM. | SANGER | |||
| MONO-MAC-6 | Growth inhibition assay | Inhibition of human MONO-MAC-6 cell growth in a cell viability assay, IC50 = 0.0338 μM. | SANGER | |||
| ALL-PO | Growth inhibition assay | Inhibition of human ALL-PO cell growth in a cell viability assay, IC50 = 0.07989 μM. | SANGER | |||
| NKM-1 | Growth inhibition assay | Inhibition of human NKM-1 cell growth in a cell viability assay, IC50 = 0.09852 μM. | SANGER | |||
| KM12 | Growth inhibition assay | Inhibition of human KM12 cell growth in a cell viability assay, IC50 = 0.35014 μM. | SANGER | |||
| TE-15 | Growth inhibition assay | Inhibition of human TE-15 cell growth in a cell viability assay, IC50 = 0.50761 μM. | SANGER | |||
| 697 | Growth inhibition assay | Inhibition of human 697 cell growth in a cell viability assay, IC50 = 0.61425 μM. | SANGER | |||
| MOLT-16 | Growth inhibition assay | Inhibition of human MOLT-16 cell growth in a cell viability assay, IC50 = 0.63132 μM. | SANGER | |||
| GB-1 | Growth inhibition assay | Inhibition of human GB-1 cell growth in a cell viability assay, IC50 = 0.71023 μM. | SANGER | |||
| TE-12 | Growth inhibition assay | Inhibition of human TE-12 cell growth in a cell viability assay, IC50 = 0.80455 μM. | SANGER | |||
| ES6 | Growth inhibition assay | Inhibition of human ES6 cell growth in a cell viability assay, IC50 = 0.98106 μM. | SANGER | |||
| LC-2-ad | Growth inhibition assay | Inhibition of human LC-2-ad cell growth in a cell viability assay, IC50 = 1.11407 μM. | SANGER | |||
| BL-70 | Growth inhibition assay | Inhibition of human BL-70 cell growth in a cell viability assay, IC50 = 1.11846 μM. | SANGER | |||
| ETK-1 | Growth inhibition assay | Inhibition of human ETK-1 cell growth in a cell viability assay, IC50 = 1.2858 μM. | SANGER | |||
| A4-Fuk | Growth inhibition assay | Inhibition of human A4-Fuk cell growth in a cell viability assay, IC50 = 1.34141 μM. | SANGER | |||
| OCI-AML2 | Growth inhibition assay | Inhibition of human OCI-AML2 cell growth in a cell viability assay, IC50 = 1.36851 μM. | SANGER | |||
| SIG-M5 | Growth inhibition assay | Inhibition of human SIG-M5 cell growth in a cell viability assay, IC50 = 1.37009 μM. | SANGER | |||
| NCI-SNU-16 | Growth inhibition assay | Inhibition of human NCI-SNU-16 cell growth in a cell viability assay, IC50 = 1.46486 μM. | SANGER | |||
| PSN1 | Growth inhibition assay | Inhibition of human PSN1 cell growth in a cell viability assay, IC50 = 1.50676 μM. | SANGER | |||
| SR | Growth inhibition assay | Inhibition of human SR cell growth in a cell viability assay, IC50 = 1.54572 μM. | SANGER | |||
| A3-KAW | Growth inhibition assay | Inhibition of human A3-KAW cell growth in a cell viability assay, IC50 = 1.62546 μM. | SANGER | |||
| KS-1 | Growth inhibition assay | Inhibition of human KS-1 cell growth in a cell viability assay, IC50 = 1.69247 μM. | SANGER | |||
| CTV-1 | Growth inhibition assay | Inhibition of human CTV-1 cell growth in a cell viability assay, IC50 = 1.72751 μM. | SANGER | |||
| LB1047-RCC | Growth inhibition assay | Inhibition of human LB1047-RCC cell growth in a cell viability assay, IC50 = 1.81624 μM. | SANGER | |||
| MEG-01 | Growth inhibition assay | Inhibition of human MEG-01 cell growth in a cell viability assay, IC50 = 1.83563 μM. | SANGER | |||
| TE-11 | Growth inhibition assay | Inhibition of human TE-11 cell growth in a cell viability assay, IC50 = 1.83985 μM. | SANGER | |||
| CMK | Growth inhibition assay | Inhibition of human CMK cell growth in a cell viability assay, IC50 = 1.95517 μM. | SANGER | |||
| NB1 | Growth inhibition assay | Inhibition of human NB1 cell growth in a cell viability assay, IC50 = 1.96117 μM. | SANGER | |||
| HAL-01 | Growth inhibition assay | Inhibition of human HAL-01 cell growth in a cell viability assay, IC50 = 2.05946 μM. | SANGER | |||
| DEL | Growth inhibition assay | Inhibition of human DEL cell growth in a cell viability assay, IC50 = 2.08482 μM. | SANGER | |||
| RL95-2 | Growth inhibition assay | Inhibition of human RL95-2 cell growth in a cell viability assay, IC50 = 2.11137 μM. | SANGER | |||
| KARPAS-299 | Growth inhibition assay | Inhibition of human KARPAS-299 cell growth in a cell viability assay, IC50 = 2.11313 μM. | SANGER | |||
| EW-16 | Growth inhibition assay | Inhibition of human EW-16 cell growth in a cell viability assay, IC50 = 2.13508 μM. | SANGER | |||
| RS4-11 | Growth inhibition assay | Inhibition of human RS4-11 cell growth in a cell viability assay, IC50 = 2.17924 μM. | SANGER | |||
| BB30-HNC | Growth inhibition assay | Inhibition of human BB30-HNC cell growth in a cell viability assay, IC50 = 2.22375 μM. | SANGER | |||
| DOHH-2 | Growth inhibition assay | Inhibition of human DOHH-2 cell growth in a cell viability assay, IC50 = 2.33431 μM. | SANGER | |||
| RPMI-8402 | Growth inhibition assay | Inhibition of human RPMI-8402 cell growth in a cell viability assay, IC50 = 2.33618 μM. | SANGER | |||
| BV-173 | Growth inhibition assay | Inhibition of human BV-173 cell growth in a cell viability assay, IC50 = 2.3366 μM. | SANGER | |||
| TE-10 | Growth inhibition assay | Inhibition of human TE-10 cell growth in a cell viability assay, IC50 = 2.34384 μM. | SANGER | |||
| TE-8 | Growth inhibition assay | Inhibition of human TE-8 cell growth in a cell viability assay, IC50 = 2.37038 μM. | SANGER | |||
| K052 | Growth inhibition assay | Inhibition of human K052 cell growth in a cell viability assay, IC50 = 2.40202 μM. | SANGER | |||
| KARPAS-45 | Growth inhibition assay | Inhibition of human KARPAS-45 cell growth in a cell viability assay, IC50 = 2.49446 μM. | SANGER | |||
| SK-NEP-1 | Growth inhibition assay | Inhibition of human SK-NEP-1 cell growth in a cell viability assay, IC50 = 2.60237 μM. | SANGER | |||
| KGN | Growth inhibition assay | Inhibition of human KGN cell growth in a cell viability assay, IC50 = 2.60339 μM. | SANGER | |||
| ML-2 | Growth inhibition assay | Inhibition of human ML-2 cell growth in a cell viability assay, IC50 = 2.63512 μM. | SANGER | |||
| LAMA-84 | Growth inhibition assay | Inhibition of human LAMA-84 cell growth in a cell viability assay, IC50 = 2.69545 μM. | SANGER | |||
| LXF-289 | Growth inhibition assay | Inhibition of human LXF-289 cell growth in a cell viability assay, IC50 = 2.72989 μM. | SANGER | |||
| A101D | Growth inhibition assay | Inhibition of human A101D cell growth in a cell viability assay, IC50 = 2.78304 μM. | SANGER | |||
| KY821 | Growth inhibition assay | Inhibition of human KY821 cell growth in a cell viability assay, IC50 = 2.79758 μM. | SANGER | |||
| ES4 | Growth inhibition assay | Inhibition of human ES4 cell growth in a cell viability assay, IC50 = 2.80628 μM. | SANGER | |||
| SCC-3 | Growth inhibition assay | Inhibition of human SCC-3 cell growth in a cell viability assay, IC50 = 2.82891 μM. | SANGER | |||
| NALM-6 | Growth inhibition assay | Inhibition of human NALM-6 cell growth in a cell viability assay, IC50 = 2.90473 μM. | SANGER | |||
| BL-41 | Growth inhibition assay | Inhibition of human BL-41 cell growth in a cell viability assay, IC50 = 2.91222 μM. | SANGER | |||
| OPM-2 | Growth inhibition assay | Inhibition of human OPM-2 cell growth in a cell viability assay, IC50 = 3.05954 μM. | SANGER | |||
| SF126 | Growth inhibition assay | Inhibition of human SF126 cell growth in a cell viability assay, IC50 = 3.06198 μM. | SANGER | |||
| BE-13 | Growth inhibition assay | Inhibition of human BE-13 cell growth in a cell viability assay, IC50 = 3.23111 μM. | SANGER | |||
| SF268 | Growth inhibition assay | Inhibition of human SF268 cell growth in a cell viability assay, IC50 = 3.32869 μM. | SANGER | |||
| MOLT-4 | Growth inhibition assay | Inhibition of human MOLT-4 cell growth in a cell viability assay, IC50 = 3.39838 μM. | SANGER | |||
| PF-382 | Growth inhibition assay | Inhibition of human PF-382 cell growth in a cell viability assay, IC50 = 3.4457 μM. | SANGER | |||
| HEL | Growth inhibition assay | Inhibition of human HEL cell growth in a cell viability assay, IC50 = 3.48005 μM. | SANGER | |||
| RPMI-6666 | Growth inhibition assay | Inhibition of human RPMI-6666 cell growth in a cell viability assay, IC50 = 3.54171 μM. | SANGER | |||
| QIMR-WIL | Growth inhibition assay | Inhibition of human QIMR-WIL cell growth in a cell viability assay, IC50 = 3.63391 μM. | SANGER | |||
| ATN-1 | Growth inhibition assay | Inhibition of human ATN-1 cell growth in a cell viability assay, IC50 = 3.67114 μM. | SANGER | |||
| BB49-HNC | Growth inhibition assay | Inhibition of human BB49-HNC cell growth in a cell viability assay, IC50 = 3.70508 μM. | SANGER | |||
| HCE-4 | Growth inhibition assay | Inhibition of human HCE-4 cell growth in a cell viability assay, IC50 = 3.78642 μM. | SANGER | |||
| SK-LMS-1 | Growth inhibition assay | Inhibition of human SK-LMS-1 cell growth in a cell viability assay, IC50 = 3.83338 μM. | SANGER | |||
| MS-1 | Growth inhibition assay | Inhibition of human MS-1 cell growth in a cell viability assay, IC50 = 3.83778 μM. | SANGER | |||
| JAR | Growth inhibition assay | Inhibition of human JAR cell growth in a cell viability assay, IC50 = 3.98832 μM. | SANGER | |||
| KE-37 | Growth inhibition assay | Inhibition of human KE-37 cell growth in a cell viability assay, IC50 = 4.06158 μM. | SANGER | |||
| LB996-RCC | Growth inhibition assay | Inhibition of human LB996-RCC cell growth in a cell viability assay, IC50 = 4.12168 μM. | SANGER | |||
| HH | Growth inhibition assay | Inhibition of human HH cell growth in a cell viability assay, IC50 = 4.20914 μM. | SANGER | |||
| HL-60 | Growth inhibition assay | Inhibition of human HL-60 cell growth in a cell viability assay, IC50 = 4.21065 μM. | SANGER | |||
| HOP-62 | Growth inhibition assay | Inhibition of human HOP-62 cell growth in a cell viability assay, IC50 = 4.3352 μM. | SANGER | |||
| NOMO-1 | Growth inhibition assay | Inhibition of human NOMO-1 cell growth in a cell viability assay, IC50 = 4.33699 μM. | SANGER | |||
| DU-4475 | Growth inhibition assay | Inhibition of human DU-4475 cell growth in a cell viability assay, IC50 = 4.36729 μM. | SANGER | |||
| LC4-1 | Growth inhibition assay | Inhibition of human LC4-1 cell growth in a cell viability assay, IC50 = 4.3807 μM. | SANGER | |||
| MC116 | Growth inhibition assay | Inhibition of human MC116 cell growth in a cell viability assay, IC50 = 4.43081 μM. | SANGER | |||
| SW982 | Growth inhibition assay | Inhibition of human SW982 cell growth in a cell viability assay, IC50 = 4.55307 μM. | SANGER | |||
| SK-N-DZ | Growth inhibition assay | Inhibition of human SK-N-DZ cell growth in a cell viability assay, IC50 = 4.67099 μM. | SANGER | |||
| EW-1 | Growth inhibition assay | Inhibition of human EW-1 cell growth in a cell viability assay, IC50 = 4.67911 μM. | SANGER | |||
| SU-DHL-1 | Growth inhibition assay | Inhibition of human SU-DHL-1 cell growth in a cell viability assay, IC50 = 4.80174 μM. | SANGER | |||
| L-363 | Growth inhibition assay | Inhibition of human L-363 cell growth in a cell viability assay, IC50 = 4.80621 μM. | SANGER | |||
| OS-RC-2 | Growth inhibition assay | Inhibition of human OS-RC-2 cell growth in a cell viability assay, IC50 = 4.8731 μM. | SANGER | |||
| HD-MY-Z | Growth inhibition assay | Inhibition of human HD-MY-Z cell growth in a cell viability assay, IC50 = 5.17301 μM. | SANGER | |||
| MHH-PREB-1 | Growth inhibition assay | Inhibition of human MHH-PREB-1 cell growth in a cell viability assay, IC50 = 5.32312 μM. | SANGER | |||
| HC-1 | Growth inhibition assay | Inhibition of human HC-1 cell growth in a cell viability assay, IC50 = 5.43291 μM. | SANGER | |||
| SK-MM-2 | Growth inhibition assay | Inhibition of human SK-MM-2 cell growth in a cell viability assay, IC50 = 5.4782 μM. | SANGER | |||
| SH-4 | Growth inhibition assay | Inhibition of human SH-4 cell growth in a cell viability assay, IC50 = 5.4837 μM. | SANGER | |||
| MHH-CALL-2 | Growth inhibition assay | Inhibition of human MHH-CALL-2 cell growth in a cell viability assay, IC50 = 5.76719 μM. | SANGER | |||
| KG-1 | Growth inhibition assay | Inhibition of human KG-1 cell growth in a cell viability assay, IC50 = 6.02878 μM. | SANGER | |||
| J-RT3-T3-5 | Growth inhibition assay | Inhibition of human J-RT3-T3-5 cell growth in a cell viability assay, IC50 = 6.07909 μM. | SANGER | |||
| MMAC-SF | Growth inhibition assay | Inhibition of human MMAC-SF cell growth in a cell viability assay, IC50 = 6.10949 μM. | SANGER | |||
| IST-SL2 | Growth inhibition assay | Inhibition of human IST-SL2 cell growth in a cell viability assay, IC50 = 6.12519 μM. | SANGER | |||
| SW954 | Growth inhibition assay | Inhibition of human SW954 cell growth in a cell viability assay, IC50 = 6.27517 μM. | SANGER | |||
| HDLM-2 | Growth inhibition assay | Inhibition of human HDLM-2 cell growth in a cell viability assay, IC50 = 6.32109 μM. | SANGER | |||
| ST486 | Growth inhibition assay | Inhibition of human ST486 cell growth in a cell viability assay, IC50 = 6.3471 μM. | SANGER | |||
| DG-75 | Growth inhibition assay | Inhibition of human DG-75 cell growth in a cell viability assay, IC50 = 6.43069 μM. | SANGER | |||
| EW-3 | Growth inhibition assay | Inhibition of human EW-3 cell growth in a cell viability assay, IC50 = 6.43207 μM. | SANGER | |||
| 8-MG-BA | Growth inhibition assay | Inhibition of human 8-MG-BA cell growth in a cell viability assay, IC50 = 6.51507 μM. | SANGER | |||
| GT3TKB | Growth inhibition assay | Inhibition of human GT3TKB cell growth in a cell viability assay, IC50 = 6.60763 μM. | SANGER | |||
| KU812 | Growth inhibition assay | Inhibition of human KU812 cell growth in a cell viability assay, IC50 = 6.6942 μM. | SANGER | |||
| CESS | Growth inhibition assay | Inhibition of human CESS cell growth in a cell viability assay, IC50 = 7.10438 μM. | SANGER | |||
| BC-1 | Growth inhibition assay | Inhibition of human BC-1 cell growth in a cell viability assay, IC50 = 7.24812 μM. | SANGER | |||
| MZ1-PC | Growth inhibition assay | Inhibition of human MZ1-PC cell growth in a cell viability assay, IC50 = 7.30389 μM. | SANGER | |||
| NCI-H82 | Growth inhibition assay | Inhibition of human NCI-H82 cell growth in a cell viability assay, IC50 = 7.3785 μM. | SANGER | |||
| NCI-H1355 | Growth inhibition assay | Inhibition of human NCI-H1355 cell growth in a cell viability assay, IC50 = 7.45804 μM. | SANGER | |||
| RPMI-8226 | Growth inhibition assay | Inhibition of human RPMI-8226 cell growth in a cell viability assay, IC50 = 7.50739 μM. | SANGER | |||
| ARH-77 | Growth inhibition assay | Inhibition of human ARH-77 cell growth in a cell viability assay, IC50 = 7.53597 μM. | SANGER | |||
| MN-60 | Growth inhibition assay | Inhibition of human MN-60 cell growth in a cell viability assay, IC50 = 7.54088 μM. | SANGER | |||
| IMR-5 | Growth inhibition assay | Inhibition of human IMR-5 cell growth in a cell viability assay, IC50 = 7.54876 μM. | SANGER | |||
| KARPAS-422 | Growth inhibition assay | Inhibition of human KARPAS-422 cell growth in a cell viability assay, IC50 = 7.57206 μM. | SANGER | |||
| CA46 | Growth inhibition assay | Inhibition of human CA46 cell growth in a cell viability assay, IC50 = 7.58012 μM. | SANGER | |||
| SJSA-1 | Growth inhibition assay | Inhibition of human SJSA-1 cell growth in a cell viability assay, IC50 = 7.66061 μM. | SANGER | |||
| no-11 | Growth inhibition assay | Inhibition of human no-11 cell growth in a cell viability assay, IC50 = 7.77557 μM. | SANGER | |||
| IST-SL1 | Growth inhibition assay | Inhibition of human IST-SL1 cell growth in a cell viability assay, IC50 = 8.02417 μM. | SANGER | |||
| NCI-H209 | Growth inhibition assay | Inhibition of human NCI-H209 cell growth in a cell viability assay, IC50 = 8.11652 μM. | SANGER | |||
| TALL-1 | Growth inhibition assay | Inhibition of human TALL-1 cell growth in a cell viability assay, IC50 = 8.18384 μM. | SANGER | |||
| KMOE-2 | Growth inhibition assay | Inhibition of human KMOE-2 cell growth in a cell viability assay, IC50 = 8.1942 μM. | SANGER | |||
| HCC1599 | Growth inhibition assay | Inhibition of human HCC1599 cell growth in a cell viability assay, IC50 = 8.19987 μM. | SANGER | |||
| GI-1 | Growth inhibition assay | Inhibition of human GI-1 cell growth in a cell viability assay, IC50 = 8.20411 μM. | SANGER | |||
| NCI-H1304 | Growth inhibition assay | Inhibition of human NCI-H1304 cell growth in a cell viability assay, IC50 = 8.20661 μM. | SANGER | |||
| Daudi | Growth inhibition assay | Inhibition of human Daudi cell growth in a cell viability assay, IC50 = 8.23546 μM. | SANGER | |||
| CPC-N | Growth inhibition assay | Inhibition of human CPC-N cell growth in a cell viability assay, IC50 = 8.29199 μM. | SANGER | |||
| MC-CAR | Growth inhibition assay | Inhibition of human MC-CAR cell growth in a cell viability assay, IC50 = 8.3358 μM. | SANGER | |||
| SW872 | Growth inhibition assay | Inhibition of human SW872 cell growth in a cell viability assay, IC50 = 8.34758 μM. | SANGER | |||
| OVCAR-4 | Growth inhibition assay | Inhibition of human OVCAR-4 cell growth in a cell viability assay, IC50 = 8.50351 μM. | SANGER | |||
| OCUB-M | Growth inhibition assay | Inhibition of human OCUB-M cell growth in a cell viability assay, IC50 = 8.56508 μM. | SANGER | |||
| SK-PN-DW | Growth inhibition assay | Inhibition of human SK-PN-DW cell growth in a cell viability assay, IC50 = 8.60985 μM. | SANGER | |||
| NCCIT | Growth inhibition assay | Inhibition of human NCCIT cell growth in a cell viability assay, IC50 = 8.71745 μM. | SANGER | |||
| NCI-H1648 | Growth inhibition assay | Inhibition of human NCI-H1648 cell growth in a cell viability assay, IC50 = 9.13834 μM. | SANGER | |||
| COR-L279 | Growth inhibition assay | Inhibition of human COR-L279 cell growth in a cell viability assay, IC50 = 9.3909 μM. | SANGER | |||
| LS-123 | Growth inhibition assay | Inhibition of human LS-123 cell growth in a cell viability assay, IC50 = 9.68161 μM. | SANGER | |||
| LP-1 | Growth inhibition assay | Inhibition of human LP-1 cell growth in a cell viability assay, IC50 = 9.78083 μM. | SANGER | |||
| NB13 | Growth inhibition assay | Inhibition of human NB13 cell growth in a cell viability assay, IC50 = 9.79994 μM. | SANGER | |||
| ONS-76 | Growth inhibition assay | Inhibition of human ONS-76 cell growth in a cell viability assay, IC50 = 9.81016 μM. | SANGER | |||
| VA-ES-BJ | Growth inhibition assay | Inhibition of human VA-ES-BJ cell growth in a cell viability assay, IC50 = 9.99933 μM. | SANGER | |||
| GR-ST | Growth inhibition assay | Inhibition of human GR-ST cell growth in a cell viability assay, IC50 = 10.2207 μM. | SANGER | |||
| ES1 | Growth inhibition assay | Inhibition of human ES1 cell growth in a cell viability assay, IC50 = 10.2984 μM. | SANGER | |||
| NB14 | Growth inhibition assay | Inhibition of human NB14 cell growth in a cell viability assay, IC50 = 10.9276 μM. | SANGER | |||
| Ramos-2G6-4C10 | Growth inhibition assay | Inhibition of human Ramos-2G6-4C10 cell growth in a cell viability assay, IC50 = 11.265 μM. | SANGER | |||
| RXF393 | Growth inhibition assay | Inhibition of human RXF393 cell growth in a cell viability assay, IC50 = 11.4834 μM. | SANGER | |||
| NCI-H2107 | Growth inhibition assay | Inhibition of human NCI-H2107 cell growth in a cell viability assay, IC50 = 11.5984 μM. | SANGER | |||
| K-562 | Growth inhibition assay | Inhibition of human K-562 cell growth in a cell viability assay, IC50 = 11.8702 μM. | SANGER | |||
| LOUCY | Growth inhibition assay | Inhibition of human LOUCY cell growth in a cell viability assay, IC50 = 11.9875 μM. | SANGER | |||
| TGBC1TKB | Growth inhibition assay | Inhibition of human TGBC1TKB cell growth in a cell viability assay, IC50 = 12.002 μM. | SANGER | |||
| COLO-320-HSR | Growth inhibition assay | Inhibition of human COLO-320-HSR cell growth in a cell viability assay, IC50 = 12.1565 μM. | SANGER | |||
| K5 | Growth inhibition assay | Inhibition of human K5 cell growth in a cell viability assay, IC50 = 12.2985 μM. | SANGER | |||
| BC-3 | Growth inhibition assay | Inhibition of human BC-3 cell growth in a cell viability assay, IC50 = 12.4651 μM. | SANGER | |||
| REH | Growth inhibition assay | Inhibition of human REH cell growth in a cell viability assay, IC50 = 12.6498 μM. | SANGER | |||
| NEC8 | Growth inhibition assay | Inhibition of human NEC8 cell growth in a cell viability assay, IC50 = 12.6887 μM. | SANGER | |||
| IST-MEL1 | Growth inhibition assay | Inhibition of human IST-MEL1 cell growth in a cell viability assay, IC50 = 12.8695 μM. | SANGER | |||
| NCI-H128 | Growth inhibition assay | Inhibition of human NCI-H128 cell growth in a cell viability assay, IC50 = 13.0723 μM. | SANGER | |||
| NCI-H1694 | Growth inhibition assay | Inhibition of human NCI-H1694 cell growth in a cell viability assay, IC50 = 13.1144 μM. | SANGER | |||
| TGW | Growth inhibition assay | Inhibition of human TGW cell growth in a cell viability assay, IC50 = 13.3276 μM. | SANGER | |||
| NCI-SNU-1 | Growth inhibition assay | Inhibition of human NCI-SNU-1 cell growth in a cell viability assay, IC50 = 13.345 μM. | SANGER | |||
| IST-MES1 | Growth inhibition assay | Inhibition of human IST-MES1 cell growth in a cell viability assay, IC50 = 13.6729 μM. | SANGER | |||
| CTB-1 | Growth inhibition assay | Inhibition of human CTB-1 cell growth in a cell viability assay, IC50 = 13.7294 μM. | SANGER | |||
| HUTU-80 | Growth inhibition assay | Inhibition of human HUTU-80 cell growth in a cell viability assay, IC50 = 13.7573 μM. | SANGER | |||
| LAN-6 | Growth inhibition assay | Inhibition of human LAN-6 cell growth in a cell viability assay, IC50 = 14.2457 μM. | SANGER | |||
| KP-N-YS | Growth inhibition assay | Inhibition of human KP-N-YS cell growth in a cell viability assay, IC50 = 14.2894 μM. | SANGER | |||
| CCRF-CEM | Growth inhibition assay | Inhibition of human CCRF-CEM cell growth in a cell viability assay, IC50 = 14.4169 μM. | SANGER | |||
| NCI-H1770 | Growth inhibition assay | Inhibition of human NCI-H1770 cell growth in a cell viability assay, IC50 = 14.5369 μM. | SANGER | |||
| MZ2-MEL | Growth inhibition assay | Inhibition of human MZ2-MEL cell growth in a cell viability assay, IC50 = 15.2251 μM. | SANGER | |||
| COR-L88 | Growth inhibition assay | Inhibition of human COR-L88 cell growth in a cell viability assay, IC50 = 16.0012 μM. | SANGER | |||
| LOXIMVI | Growth inhibition assay | Inhibition of human LOXIMVI cell growth in a cell viability assay, IC50 = 16.256 μM. | SANGER | |||
| KALS-1 | Growth inhibition assay | Inhibition of human KALS-1 cell growth in a cell viability assay, IC50 = 16.5931 μM. | SANGER | |||
| D-283MED | Growth inhibition assay | Inhibition of human D-283MED cell growth in a cell viability assay, IC50 = 16.8489 μM. | SANGER | |||
| NCI-H719 | Growth inhibition assay | Inhibition of human NCI-H719 cell growth in a cell viability assay, IC50 = 16.9161 μM. | SANGER | |||
| MLMA | Growth inhibition assay | Inhibition of human MLMA cell growth in a cell viability assay, IC50 = 16.9907 μM. | SANGER | |||
| EVSA-T | Growth inhibition assay | Inhibition of human EVSA-T cell growth in a cell viability assay, IC50 = 17.0485 μM. | SANGER | |||
| SK-N-FI | Growth inhibition assay | Inhibition of human SK-N-FI cell growth in a cell viability assay, IC50 = 17.6913 μM. | SANGER | |||
| NTERA-S-cl-D1 | Growth inhibition assay | Inhibition of human NTERA-S-cl-D1 cell growth in a cell viability assay, IC50 = 17.8572 μM. | SANGER | |||
| NCI-H1882 | Growth inhibition assay | Inhibition of human NCI-H1882 cell growth in a cell viability assay, IC50 = 17.9834 μM. | SANGER | |||
| A704 | Growth inhibition assay | Inhibition of human A704 cell growth in a cell viability assay, IC50 = 17.9904 μM. | SANGER | |||
| L-428 | Growth inhibition assay | Inhibition of human L-428 cell growth in a cell viability assay, IC50 = 18.0151 μM. | SANGER | |||
| HCC1187 | Growth inhibition assay | Inhibition of human HCC1187 cell growth in a cell viability assay, IC50 = 18.0188 μM. | SANGER | |||
| NCI-H1581 | Growth inhibition assay | Inhibition of human NCI-H1581 cell growth in a cell viability assay, IC50 = 18.0866 μM. | SANGER | |||
| BB65-RCC | Growth inhibition assay | Inhibition of human BB65-RCC cell growth in a cell viability assay, IC50 = 18.4162 μM. | SANGER | |||
| EM-2 | Growth inhibition assay | Inhibition of human EM-2 cell growth in a cell viability assay, IC50 = 18.5672 μM. | SANGER | |||
| Raji | Growth inhibition assay | Inhibition of human Raji cell growth in a cell viability assay, IC50 = 19.9565 μM. | SANGER | |||
| TE-1 | Growth inhibition assay | Inhibition of human TE-1 cell growth in a cell viability assay, IC50 = 20.4104 μM. | SANGER | |||
| SW962 | Growth inhibition assay | Inhibition of human SW962 cell growth in a cell viability assay, IC50 = 20.4293 μM. | SANGER | |||
| MHH-NB-11 | Growth inhibition assay | Inhibition of human MHH-NB-11 cell growth in a cell viability assay, IC50 = 20.5521 μM. | SANGER | |||
| no-10 | Growth inhibition assay | Inhibition of human no-10 cell growth in a cell viability assay, IC50 = 21.0265 μM. | SANGER | |||
| GDM-1 | Growth inhibition assay | Inhibition of human GDM-1 cell growth in a cell viability assay, IC50 = 21.9414 μM. | SANGER | |||
| KMS-12-PE | Growth inhibition assay | Inhibition of human KMS-12-PE cell growth in a cell viability assay, IC50 = 22.2739 μM. | SANGER | |||
| NCI-H510A | Growth inhibition assay | Inhibition of human NCI-H510A cell growth in a cell viability assay, IC50 = 24.1278 μM. | SANGER | |||
| ES5 | Growth inhibition assay | Inhibition of human ES5 cell growth in a cell viability assay, IC50 = 24.7349 μM. | SANGER | |||
| JiyoyeP-2003 | Growth inhibition assay | Inhibition of human JiyoyeP-2003 cell growth in a cell viability assay, IC50 = 26.2742 μM. | SANGER | |||
| NMC-G1 | Growth inhibition assay | Inhibition of human NMC-G1 cell growth in a cell viability assay, IC50 = 27.1822 μM. | SANGER | |||
| NCI-H446 | Growth inhibition assay | Inhibition of human NCI-H446 cell growth in a cell viability assay, IC50 = 27.4946 μM. | SANGER | |||
| NB7 | Growth inhibition assay | Inhibition of human NB7 cell growth in a cell viability assay, IC50 = 27.9229 μM. | SANGER | |||
| A388 | Growth inhibition assay | Inhibition of human A388 cell growth in a cell viability assay, IC50 = 28.0073 μM. | SANGER | |||
| JVM-2 | Growth inhibition assay | Inhibition of human JVM-2 cell growth in a cell viability assay, IC50 = 28.2898 μM. | SANGER | |||
| HT-144 | Growth inhibition assay | Inhibition of human HT-144 cell growth in a cell viability assay, IC50 = 28.6899 μM. | SANGER | |||
| NCI-H747 | Growth inhibition assay | Inhibition of human NCI-H747 cell growth in a cell viability assay, IC50 = 28.9195 μM. | SANGER | |||
| NCI-H1650 | Growth inhibition assay | Inhibition of human NCI-H1650 cell growth in a cell viability assay, IC50 = 29.0176 μM. | SANGER | |||
| EB-3 | Growth inhibition assay | Inhibition of human EB-3 cell growth in a cell viability assay, IC50 = 29.5309 μM. | SANGER | |||
| KLE | Growth inhibition assay | Inhibition of human KLE cell growth in a cell viability assay, IC50 = 29.619 μM. | SANGER | |||
| TK10 | Growth inhibition assay | Inhibition of human TK10 cell growth in a cell viability assay, IC50 = 30.126 μM. | SANGER | |||
| COLO-668 | Growth inhibition assay | Inhibition of human COLO-668 cell growth in a cell viability assay, IC50 = 30.792 μM. | SANGER | |||
| NCI-H23 | Growth inhibition assay | Inhibition of human NCI-H23 cell growth in a cell viability assay, IC50 = 31.1063 μM. | SANGER | |||
| GOTO | Growth inhibition assay | Inhibition of human GOTO cell growth in a cell viability assay, IC50 = 31.6085 μM. | SANGER | |||
| MSTO-211H | Growth inhibition assay | Inhibition of human MSTO-211H cell growth in a cell viability assay, IC50 = 31.8678 μM. | SANGER | |||
| LB831-BLC | Growth inhibition assay | Inhibition of human LB831-BLC cell growth in a cell viability assay, IC50 = 32.3843 μM. | SANGER | |||
| SCH | Growth inhibition assay | Inhibition of human SCH cell growth in a cell viability assay, IC50 = 32.8485 μM. | SANGER | |||
| EHEB | Growth inhibition assay | Inhibition of human EHEB cell growth in a cell viability assay, IC50 = 34.1193 μM. | SANGER | |||
| U-266 | Growth inhibition assay | Inhibition of human U-266 cell growth in a cell viability assay, IC50 = 34.2782 μM. | SANGER | |||
| EW-11 | Growth inhibition assay | Inhibition of human EW-11 cell growth in a cell viability assay, IC50 = 34.4725 μM. | SANGER | |||
| TE-9 | Growth inhibition assay | Inhibition of human TE-9 cell growth in a cell viability assay, IC50 = 37.0401 μM. | SANGER | |||
| ES3 | Growth inhibition assay | Inhibition of human ES3 cell growth in a cell viability assay, IC50 = 37.5004 μM. | SANGER | |||
| NCI-H2141 | Growth inhibition assay | Inhibition of human NCI-H2141 cell growth in a cell viability assay, IC50 = 38.0843 μM. | SANGER | |||
| MPP-89 | Growth inhibition assay | Inhibition of human MPP-89 cell growth in a cell viability assay, IC50 = 42.0586 μM. | SANGER | |||
| SK-MEL-2 | Growth inhibition assay | Inhibition of human SK-MEL-2 cell growth in a cell viability assay, IC50 = 42.6405 μM. | SANGER | |||
| LC-1F | Growth inhibition assay | Inhibition of human LC-1F cell growth in a cell viability assay, IC50 = 43.3682 μM. | SANGER | |||
| NH-12 | Growth inhibition assay | Inhibition of human NH-12 cell growth in a cell viability assay, IC50 = 43.9359 μM. | SANGER | |||
| RKO | Growth inhibition assay | Inhibition of human RKO cell growth in a cell viability assay, IC50 = 44.1252 μM. | SANGER | |||
| KM-H2 | Growth inhibition assay | Inhibition of human KM-H2 cell growth in a cell viability assay, IC50 = 44.9577 μM. | SANGER | |||
| SK-UT-1 | Growth inhibition assay | Inhibition of human SK-UT-1 cell growth in a cell viability assay, IC50 = 49.8825 μM. | SANGER | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 25 mg/mL
(62.73 mM)
Ethanol : 8 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 398.47 | Formula | C22H27FN4O2 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 557795-19-4 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | SU11248 | Smiles | CCN(CC)CCNC(=O)C1=C(NC(=C1C)C=C2C3=C(C=CC(=C3)F)NC2=O)C | ||
Mechanism of Action
| Targets/IC50/Ki |
FLT3
(Cell-free assay) c-Kit
(Cell-free assay) PDGFRβ
(Cell-free assay) 2 nM
VEGFR2
(Cell-free assay) 80 nM
|
|---|---|
| In vitro |
Sunitinib also potently inhibits Kit and FLT-3. This compound is a potent ATP-competitive inhibitor of VEGFR2 (Flk1) and PDGFRβ with Ki of 9 nM and 8 nM, respectively, displaying >10-fold higher selectivity for VEGFR2 and PDGFR than FGFR-1, EGFR, Cdk2, Met, IGFR-1, Abl, and src. In serum-starved NIH-3T3 cells expressing VEGFR2 or PDGFRβ, it inhibits VEGF-dependent VEGFR2 phosphorylation and PDGF-dependent PDGFRβ phosphorylation with IC50 of 10 nM and 10 nM, respectively. This chemical inhibits VEGF-induced proliferation of serum-starved HUVECs with IC50 of 40 nM, and inhibits PDGF-induced proliferation of NIH-3T3 cells overexpressing PDGFRβ or PDGFRα with IC50 of 39 nM and 69 nM, respectively. It inhibits phosphorylation of wild-type FLT3, FLT3-ITD, and FLT3-Asp835 with IC50 of 250 nM, 50 nM, and 30 nM, respectively. This inhibitor inhibits the proliferation of MV4;11 and OC1-AML5 cells with IC50 of 8 nM and 14 nM, respectively, and induces apoptosis in a dose-dependent manner.
|
| Kinase Assay |
Biochemical Tyrosine Kinase Assays
|
|
IC50 values for Sunitinib against VEGFR2 (Flk-1) and PDGFRβ are determined using glutathione S-transferase fusion proteins containing the complete cytoplasmic domain of the RTK. Biochemical tyrosine kinase assays to quantitate the trans-phosphorylation activity of VEGFR2 (Flk-1) and PDGFRβ are performed in 96-well microtiter plates precoated (20 μg/well in PBS; incubated overnight at 4 °C) with the peptide substrate poly-Glu,Tyr (4:1). Excess protein binding sites are blocked with the addition of 1-5% (w/v) BSA in PBS. Purified GST-fusion proteins are produced in baculovirus-infected insect cells. GST-VEGFR2 and GST-PDGFRβ are then added to the microtiter wells in 2 × concentration kinase dilution buffer consisting of 100 mM HEPES, 50 mM NaCl, 40 μM NaVO4, and 0.02% (w/v) BSA. The final enzyme concentration for GST-VEGFR2 or GST-PDGFRβ is 50 ng/mL. Twenty-five μL of diluted this compound are subsequently added to each reaction well to produce a range of inhibitor concentrations appropriate for each enzyme. The kinase reaction is initiated by the addition of different concentrations of ATP in a solution of MnCl2 so that the final ATP concentrations spanned the Km for the enzyme, and the final concentration of MnCl2 is 10 mM. The plates are incubated for 5-15 minutes at room temperature before stopping the reaction with the addition of EDTA. The plates are then washed three times with TBST. Rabbit polyclonal antiphosphotyrosine antisera are added to the wells at a 1:10,000 dilution in TBST containing 0.5% (w/v) BSA, 0.025% (w/v) nonfat dry milk, and 100 μM NaVO4 and incubated for 1 hour at 37 °C. The plates are then washed three times with TBST, followed by the addition of goat antirabbit antisera conjugated with horseradish peroxidase (1:10,000 dilution in TBST). The plates are incubated for 1 hour at 37 °C and then washed three times with TBST. The amount of phosphotyrosine in each well is quantitated after the addition of 2,2′-azino-di-[3-ethylbenzthiazoline sulfonate] as substrate.
|
|
| In vivo |
Consistent with the substantial and selective inhibition of VEGFR2 or PDGFR phosphorylation and signaling in vivo, Sunitinib (20-80 mg/kg/day) exhibits broad and potent dose-dependent anti-tumor activity against a variety of tumor xenograft models including HT-29, A431, Colo205, H-460, SF763T, C6, A375, or MDA-MB-435. This compound dosing at 80 mg/kg/day for 21 days leads to complete tumor regression in six of eight mice, without tumor re-growing during a 110-day observation period after the end of treatment. Second round of treatment with this compound remains efficacious against tumors that are not fully regressed during the first round of treatment. This compound treatment results in significant decrease in tumor MVD, with ~40% reduction in SF763T glioma tumors. SU11248 treatment results in a complete inhibition of additional tumor growth of luciferase-expressing PC-3M xenografts, despite no reduction in tumor size. This compound treatment (20 mg/kg/day) dramatically suppresses the growth subcutaneous MV4;11 (FLT3-ITD) xenografts and prolongs survival in the FLT3-ITD bone marrow engraftment model.
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | p-AKT / AKT / p-ERK / ERK p-GSK3β / GSK3β / MYCN p-STAT3 / STAT3 / p-Src / Src / p-AKT / AKT / p-ERK / ERK |
|
24759734 |
| Growth inhibition assay | Cell viability |
|
24369536 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06222593 | Not yet recruiting | Carcinoma Renal Cell |
State University of New York at Buffalo |
June 1 2024 | Phase 1|Phase 2 |
| NCT06208748 | Not yet recruiting | Gastrointestinal Stromal Tumors|GIST |
Sarcoma Alliance for Research through Collaboration|Cogent Biosciences Inc.|Dana-Farber Cancer Institute|The Life Raft Group |
March 2024 | Phase 2 |
| NCT05745142 | Completed | Carcinoma Renal Cell|Clear-cell Metastatic Renal Cell Carcinoma |
Pfizer |
February 23 2023 | -- |
| NCT05043090 | Recruiting | Papillary Renal Cell Carcinoma |
AstraZeneca |
October 28 2021 | Phase 3 |
| NCT04669366 | Completed | Kidney Neoplasms |
Pfizer |
January 20 2021 | -- |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































