research use only
Dronedarone HCl Potassium Channel inhibitor
Cat.No.S2114
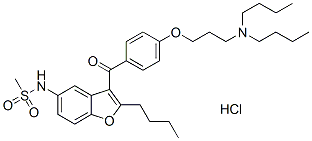
Chemical Structure
Molecular Weight: 593.22
Quality Control
| Related Targets | CFTR CRM1 CD markers AChR Calcium Channel Sodium Channel GABA Receptor TRP Channel ATPase GluR |
|---|---|
| Other Potassium Channel Inhibitors | Nigericin TRAM-34 Nicorandil ML133 HCl Hydralazine HCl Sophocarpine Senicapoc (ICA-17043) Gliquidone PAP-1 E-4031 dihydrochloride |
Solubility
|
In vitro |
DMSO
: 80 mg/mL
(134.85 mM)
Ethanol : 40 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 593.22 | Formula | C31H44N2O5S.HCl |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 141625-93-6 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | SR33589 | Smiles | CCCCC1=C(C2=C(O1)C=CC(=C2)NS(=O)(=O)C)C(=O)C3=CC=C(C=C3)OCCCN(CCCC)CCCC.Cl | ||
Mechanism of Action
| Targets/IC50/Ki |
Potassium channel
Sodium channel
Calcium channel
|
|---|---|
| In vitro |
Dronedarone reduces the incidence of early and delayed after depolarizations evoked by 1 mM Dofetilide and 0.2 mM Strophantidine in Purkinje fibres. Dronedarone (10 mM) markedly reduces the L-type calcium current (76.5%) and the rapid component of the delayed rectifier potassium current (97%) in ventricular myocytes. Dronedarone inhibits the activity of I(K(ACh)) channels recorded from cell-attached patches by reducing the channel open probability (from 0.56 to 0.11) without modification of the single-channel conductance in single cells isolated from sinoatrial node (SAN) tissue of rabbit hearts. Dronedarone exhibits a state-dependent inhibition of the fast Na(+) channel current with an IC50 of 0.7 μM in guinea pig ventricular myocytes, when the holding potential (V (hold)) is -80 mV. Dronedarone blocks Ca(2+) currents elicited by rectangular pulses at V (hold) = -40 mV with IC50 value of 0.4 μM, whereas at V (hold) = -80 mV, Dronedarone (10 μM) blocks only 20 % of the current.
|
| In vivo |
Dronedarone increases action potential duration in normal hearts of rats. Dronedarone reduces the late sustained K(+) current, I(K) (or Isus) by 69%. Dronedarone induces only a tonic block of I(K). Dronedarone induces a weak increase in the fast transient outward current, I(to), in rats after myocardial infarction.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT01430806 | Unknown status | ICD|ICD SHOCKS|DRONEDARONE |
Rabin Medical Center |
October 2011 | Not Applicable |
| NCT01266681 | Unknown status | Atrial Fibrillation |
Eastbourne General Hospital |
March 2011 | Not Applicable |
| NCT01199081 | Completed | Atrial Fibrillation |
Sanofi |
October 2010 | Phase 4 |
| NCT01213368 | Completed | Atrial Fibrillation |
Sanofi |
September 2010 | Phase 2 |
| NCT01140581 | Completed | Atrial Fibrillation |
Sanofi |
September 2010 | Phase 4 |
| NCT01070667 | Unknown status | Paroxysmal Atrial Fibrillation |
Eastbourne General Hospital |
March 2010 | Phase 4 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































