research use only
Bazedoxifene (WAY-140424) HCl Estrogen/progestogen Receptor modulator
Cat.No.S2128
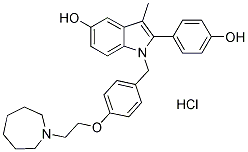
Chemical Structure
Molecular Weight: 507.06
Quality Control
| Related Targets | Adrenergic Receptor GPR Androgen Receptor Glucocorticoid Receptor ACE RAAS Progesterone Receptor Opioid Receptor PGES THR |
|---|---|
| Other Estrogen/progestogen Receptor Inhibitors | Elacestrant (RAD1901) Dihydrochloride Vepdegestrant (ARV-471) MPP dihydrochloride Kaempferol Cholesterol PHTPP G15 Licochalcone A Endoxifen HCl Chrysin |
Solubility
|
In vitro |
DMSO
: 90 mg/mL
(177.49 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 507.06 | Formula | C30H34N2O3.HCl |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 198480-56-7 | -- | Storage of Stock Solutions |
|
|
| Synonyms | TSE-424 | Smiles | CC1=C(N(C2=C1C=C(C=C2)O)CC3=CC=C(C=C3)OCCN4CCCCCC4)C5=CC=C(C=C5)O.Cl | ||
Mechanism of Action
| Targets/IC50/Ki |
ERα
(radioligand binding assay) 23 nM
ERβ
(radioligand binding assay) 89 nM
|
|---|---|
| In vitro |
Bazedoxifene is a third generation selective estrogen receptor modulator (SERM). Bazedoxifene does not stimulate ERα mediated transcriptional activity and acts as an antagonist to estradiol in cultured breast cancer (bMCF-7) cells. Similar results are seen in other cell lines including CHO (ovarian), HepG2 (hepatic) or GTI-7 (neuronal) with bazedoxifene having no ERα agonist activity and acting as an antagonist to estradiol action. Bazedoxifene does not stimulate proliferation of MCF-7 cells but did inhibit 17β-estradiol-induced proliferation with IC50 of 0.19 nM.
|
| Kinase Assay |
Ligand binding competition experiments
|
|
Test compounds are initially solubilized in DMSO and the final concentration of DMSO in the binding assay is ≤ 1%. Eight dilutions of each test compound are used as an unlabelled competitor for [3H]17β-estradiol. Typically, a set of compound dilutions would be tested simultaneously on human, rat and mouse ER-α and ER-β. The results are plotted as measured DPM vs. concentration of test compound. For dose-response curve fitting, a four parameter logistic model on the transformed, weighted data are fit and the IC50 is defined as the concentration of compound decreasing maximum [3H]estradiol binding by 50%. For active compounds, the IC50 is determined at least three times. It should be noted that IC50 values are not direct measures of a ligand’s affinity for the receptor. Rather, they can only be compared as relative values, in this case to 17β-estradiol.
|
|
| In vivo |
In an immature rat model, bazedoxifene increases uterine wet weight 35% at a dose of 0.5 mg/kg compared to an 85% increase with raloxifene at the same dose and a 300% increase in uterine weight with ethinyl estradiol at a dose of 10 μg/kg. Ovarectomized rats treated with 0.3 mg/d bazedoxifene displayed maintenance of bone mass and bone strength similar to effects seen with 2 μg/d ethinyl estradiol, 3 mg/d raloxifene, or sham operated animals.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT03475719 | Unknown status | Osteoporosis Postmenopausal |
Huons Co. Ltd. |
January 11 2018 | Phase 1 |
| NCT03005340 | Unknown status | Healthy |
Alvogen Korea |
December 2016 | Phase 1 |
| NCT02729701 | Completed | Breast Cancer |
University of Kansas Medical Center|Pfizer |
May 2016 | Phase 2 |
| NCT01973738 | Unknown status | Selective Estrogen Receptor Modulator |
Toshihiko Kono|Tomidahama Hospital |
January 2012 | -- |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































