research use only
Phenformin HCl AMPK activator
Cat.No.S2542
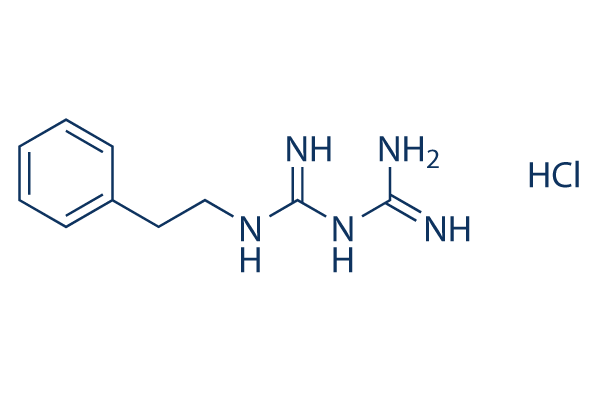
Chemical Structure
Molecular Weight: 241.72
Quality Control
| Related Targets | PI3K Akt mTOR GSK-3 ATM/ATR DNA-PK PDPK1 PTEN PP2A PDK |
|---|---|
| Other AMPK Inhibitors | Dorsomorphin Dihydrochloride Dorsomorphin (Compound C) AICAR (Acadesine) A-769662 GSK621 WZ4003 Ex229 (Compound 991) BAY-3827 HTH-01-015 MK-8722 |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| CHO | Function assay | TP_TRANSPORTER: uptake in OCT1-expressing CHO cells, Km=15.6μM | 12130709 | |||
| DAOY | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for DAOY cells | 29435139 | |||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | |||
| BT-12 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-12 cells | 29435139 | |||
| OHS-50 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for OHS-50 cells | 29435139 | |||
| HCT116 | Function assay | 0.5 mM | 24 hrs | Effect on mitochondrial respiration in human HCT116 cells expressing carbonic anhydrase 9 assessed as reduction in oxygen consumption rate at 0.5 mM after 24 hrs under hypoxic condition by Western blot analysis | 27823879 | |
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
Water : 48 mg/mL
DMSO
: Insoluble
Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 241.72 | Formula | C10H15N5.HCl |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 834-28-6 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | NSC-756501, Phenethylbiguanide | Smiles | C1=CC=C(C=C1)CCN=C(N)N=C(N)N.Cl | ||
Mechanism of Action
| Targets/IC50/Ki |
AMPK
(Cell-free assay) |
|---|---|
| In vitro |
Phenformin stimulates the phosphorylation and activation of AMPKalpha1 and AMPKalpha2 without altering LKB1 activity. Phenformin increases AMPK activity and phosphorylation in the isolated heart, the increase in AMPK activity is always preceded by and correlated with increased cytosolic [AMP]. Phenformin is a 50-fold more potent inhibitor of mitochondrial complex I than 1,1-Dimethylbiguanide. Phenformin robustly induces apoptosis in LKB1 deficient NSCLC cell lines. Phenformin at 2 mM similarly induces AMPK signaling as shown by increased P-AMPK and P-Raptor levels. Phenformin induces higher levels of cellular stress, triggering induction of P-Ser51 eIF2α and its downstream target CHOP, and markers of apoptosis at later times. Phenformin induces a significant increase in survival and therapeutic response in KLluc mice following long-term treatment. Phenformin and AICAR increases AMPK activity in H441 cells in a dose-dependent fashion, stimulating the kinase maximally at 5-10 mm and 2 mm, respectively. Phenformin significantly decreases basal ion transport (measured as short circuit current) across H441 monolayers by approximately 50% compared with that of controls. Phenformin and AICAR significantly reduce MK-870-sensitive transepithelial Na+ transport compared with controls. Phenformin and AICAR suppress MK-870-sensitive Na+ transport across H441 cells via a pathway that includes activation of AMPK and inhibition of both apical Na+ entry through ENaC and basolateral Na+ extrusion via the Na+,K+-ATPase. Phenformin-treated rats reveals a tendency towards a decrease in blood insulin level (radioimmunoassay). |
| In vivo |
Phenformin also increases levels of P-eIF2α and its target BiP/Grp78 in normal lung as well as in lung tumors of mice. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | E-cadherin / Vimentin / Snail / Slug / Twist1 p-ErbB2 / ErbB2 / p-AKT / AKT / p-mTOR / mTOR / p-ERK / ERK p-AMPKα / AMPK IRβ / p-IRS1 / IRS1 / p-IGF1Rβ / IGF1R |
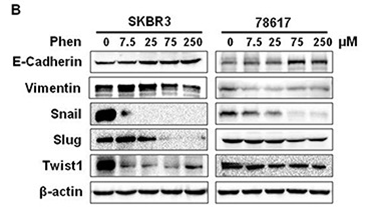
|
28947975 |
| Immunofluorescence | E-cadherin / Vimentin |
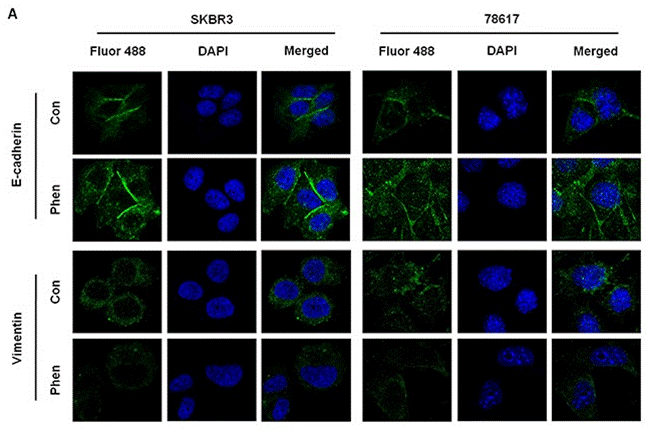
|
28947975 |
| Growth inhibition assay | Cell viability |
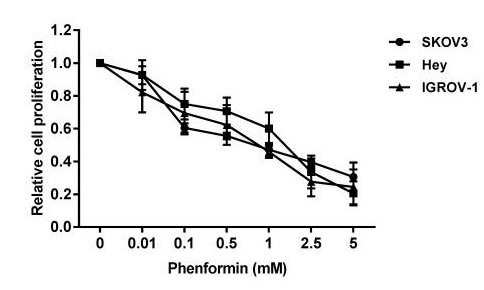
|
29245964 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































