research use only
Darifenacin HBr AChR antagonist
Cat.No.S3144
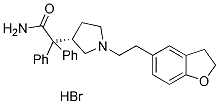
Chemical Structure
Molecular Weight: 507.46
Quality Control
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| HEK cells | Function assay | Agonist activity at human beta2-adrenoceptor expressed in HEK cells assessed as increase of cAMP level after 10 mins by radioimmunoassay, EC50=0.00082 μM | ||||
| H292 cells | Function assay | Agonist activity at human adrenergic beta2 receptor expressed in H292 cells assessed as stimulation of cAMP accumulation after 60 mins, EC50=0.01585 μM | ||||
| HEK293 cells | Function assay | Agonist activity at human beta2 adrenergic receptor expressed in HEK293 cells assessed as cAMP accumulation using [125I]cAMP by scintillation counting, EC50=0.0007943 μM | ||||
| CHO-K1 cells | Function assay | Inhibition of fast sodium current (INa) in Chinese Hamster Ovary (CHO) K1 cells transfected with human Nav1.5 measured using IonWorks Quattro automated patch clamp platform, IC50=1.58489 μM | ||||
| PC3 | Proliferation assay | 10 μM and 20 μM | blockade of CHRM3 by darifenacin could effectively reduce cell proliferation | |||
| 22Rv1 | Proliferation assay | 10 μM and 20 μM | blockade of CHRM3 by darifenacin could effectively reduce cell proliferation | |||
| H1299 | Proliferation assay | 0.3–100 µM | 72 h | significantly inhibited H1299 cell proliferation in a concentration-dependent manner | ||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 117 mg/mL
(230.56 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 507.46 | Formula | C28H30N2O2.HBr |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 133099-07-7 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | UK-88525 | Smiles | C1CN(CC1C(C2=CC=CC=C2)(C3=CC=CC=C3)C(=O)N)CCC4=CC5=C(C=C4)OCC5.Br | ||
Mechanism of Action
| Targets/IC50/Ki |
M3 mAChR
8.9(pKi)
|
|---|---|
| In vitro |
Darifenacin exerts non-parallel rightward displacement of the agonist curve and also significant depression of the maximum response (+)-cis-Dioxolane produced concentration-dependent contraction of the isolated bladder of rat. Darifenacin produces a concentration dependent increase in R123 (P-gp probe) accumulation in MDCK cells. Darifenacin stimulates ATPase activity in P-gp membrane in a clear concentration dependent response manner with an estimated ED50 value of 1.6 µM. Darifenacin (100 nM) shows a significantly greater permeability for darifenacin in the basolateral to apical direction resulting in an efflux ratio in BBMEC monolayers of approximately 2.6.
|
| In vivo |
Darifenacin produces dose-dependent inhibition of amplitude of volume-induced bladder contractions(VIBCAMP), producing 35% inhibition at dose of 283.3 nmol/kg and maximal inhibition of approximately 50–55%. Darifenacin (0.1 mg/kg i.v.) reduces bladder afferent activity in both Aδ and C fibers in female Sprague-Dawley rats, the decrease in afferent spikes in C fibers may be more pronounced than that in Aδ fibers. Darifenacin (7.5 mg and 15 mg, daily) reduces the number of incontinence episodes per week from baseline by 67.7% and 72.8% respectively compared with 55.9% with placebo in patients with overactive bladder (OAB). Darifenacin (7.5 mg and 15 mg, daily) also shows significantly superior to placebo for improvements in micturition frequency, bladder capacity, frequency of urgency, severity of urgency and number of incontinence episodes leading to a change in clothing or pads in patients with overactive bladder (OAB).
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT00703703 | Completed | Healthy |
Novartis|Procter and Gamble |
May 2008 | Phase 1 |
| NCT00921245 | Completed | Overactive Bladder |
Bayer |
June 2007 | -- |
| NCT00413790 | Completed | Healthy |
Novartis|Procter and Gamble |
November 2006 | Phase 4 |
| NCT00413426 | Completed | Healthy |
Novartis|Procter and Gamble |
June 2006 | Phase 1 |
| NCT00366002 | Completed | Overactive Bladder (OAB) |
Novartis|Procter and Gamble |
June 2006 | Phase 4 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































