research use only
Apremilast PDE inhibitor
Cat.No.S8034
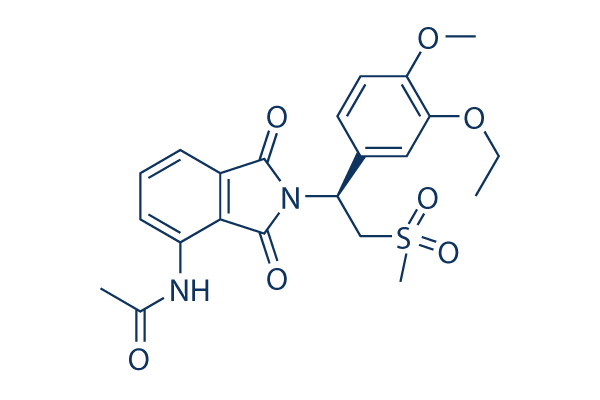
Chemical Structure
Molecular Weight: 460.5
Quality Control
Solubility
|
In vitro |
DMSO
: 92 mg/mL
(199.78 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 460.5 | Formula | C22H24N2O7S |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 608141-41-9 | -- | Storage of Stock Solutions |
|
|
| Synonyms | CC-10004 | Smiles | CCOC1=C(C=CC(=C1)C(CS(=O)(=O)C)N2C(=O)C3=C(C2=O)C(=CC=C3)NC(=O)C)OC | ||
Mechanism of Action
| Targets/IC50/Ki |
PDE4
74 nM
TNF-α
77 nM
|
|---|---|
| In vitro |
Apremilast is more potent for inhibition of PDE4 compared with cAMP or cGMP hydrolysing enzymes from other PDE families. This compound displays a broad pattern of anti-inflammatory activity in a variety of cell types, inhibits TNF-α, IL-12 and IL-23 production, as well as NK and keratinocyte responses. It is found to inhibit the zymosan-induced PMN production of IL-8 with IC50 of 94 nM. This chemical inhibits fMLF-induced PMN CD18 and CD11b expression with IC50 of 390 nM and 74 nM, respectively, and inhibits fMLF-induced adhesion of PMN to HUVECs with IC50 of 150 nM. It inhibits keratinocyte TNF-αproduction, with no effect on keratinocyte cell viability as measured by intracellular ATP levels. .
|
| Kinase Assay |
PDE4 inhibitory activity
|
|
Cells (1×109) are washed in PBS and lysed in cold homogenization buffer (20 mM Tris-HCl, pH 7.1, 3 mM 2-mercaptoethanol, 1 mM MgCl2, 0.1 mM EGTA, 1 μM PMSF, 1 μg/mL leupeptin). Following homogenization in a Dounce homogenizer the supematant is collected by centrifugation and loaded onto a Sephacryl S-200 column equilibrated in homogenization buffer. PDE is eluted in homogenization buffer and the rolipram sensitive fractions pooled and stored in aliquots. Enzyme activity is assayed in 50 mM Tris- HCl, pH 7.5, 5 mM MgCl2 and 1 μM cAMP (of which 1% is3H cAMP) in the presence of varying concentrations of inhibitors. The amount of extract used is pre-determined to ensure that reactions are within the linear range and consumed lessthan15%of the total substrate. Reactions are performed at 30°C for 30 min and terminated by boiling for 2 min. The samples are then chilled and treated with snake 'venom (1mg/mL) at 30 °C for 15 min. Unused substrate is removed by incubation with 200 μL AG1-X8 resin for 15 min. Samples are then spun at 3000 rpm for 5 min and 50 μL of the aqueous phase taken for counting. Eachdata point is carried out in duplicate with activity expressed as percentage of control. IC50 is determined from dose response curves derived from three independent experiments.
|
|
| In vivo |
Apremilast is stable in the presence of human microsomes (t1/2 > 60 min). It is 90% protein bound in human plasma. Oral and intravenous administration of this compound in female rats showed that it have good pharmacokinetics with low clearance, a moderate volume of distribution, and a 64% oral bioavailability. In a LPS-induced TNF-αinhibition model in rats, examined the TNF-α inhibitory ability of this compound in vivo, and the ED50 is determined to be 0.03 mg/kg. In another LPS-induced neutrophilia model in rats, this compound exhibited an ED50 range from 0.3 mg/kg to 0.9 mg/kg.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06382987 | Recruiting | Plaque Psoriasis |
Bristol-Myers Squibb |
January 22 2024 | -- |
| NCT06122649 | Recruiting | Plaque Psoriasis |
Amgen |
November 27 2023 | Phase 3 |
| NCT06108544 | Recruiting | Plaque Psoriasis |
Takeda |
November 6 2023 | Phase 3 |
| NCT06088043 | Recruiting | Plaque Psoriasis |
Takeda |
November 6 2023 | Phase 3 |
| NCT05926882 | Completed | Alopecia Areata |
Jinnah Postgraduate Medical Centre |
August 1 2022 | Phase 4 |
| NCT04804553 | Recruiting | Active Juvenile Psoriatic Arthritis |
Amgen |
March 17 2022 | Phase 3 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































