research use only
Nebivolol hydrochloride (R-65824) Adrenergic Receptor antagonist
Cat.No.S1549
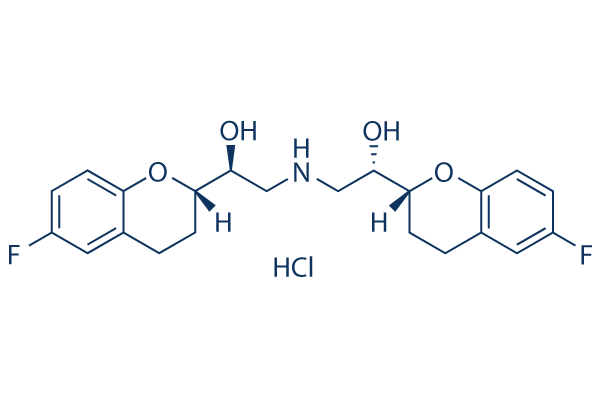
Chemical Structure
Molecular Weight: 441.9
Quality Control
| Related Targets | AChR 5-HT Receptor COX Calcium Channel Histamine Receptor Dopamine Receptor GABA Receptor TRP Channel Cholinesterase (ChE) GluR |
|---|---|
| Other Adrenergic Receptor Inhibitors | ICI 118551 Hydrochloride (Zenidolol) L755507 Yohimbine HCl Atipamezole Higenamine hydrochloride Fenoterol hydrobromide Medetomidine HCl Isoprenaline L-Adrenaline Detomidine HCl |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| (CHO) K1 cells | Function assay | Inhibition of rapid delayed inward rectifying potassium current (IKr) in Chinese hamster ovary (CHO) K1 cells stably expressing hERG measured using IonWorks Quattro automated patch clamp platform, IC50=6.30957 μM | ||||
| HEK293 cells | Function assay | Inhibition of fast sodium current (INa) in HEK293 cells transfected with human Nav1.5 measured using IonWorks Quattro automated patch clamp platform, IC50=7.94328 μM | ||||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 88 mg/mL
(199.14 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 441.9 | Formula | C22H25F2NO4.HCl |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 152520-56-4 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | R-65824 hydrochloride | Smiles | C1CC2=C(C=CC(=C2)F)OC1C(CNCC(C3CCC4=C(O3)C=CC(=C4)F)O)O.Cl | ||
Mechanism of Action
| Features |
A highly cardioselective compound under certain conditions.
|
|---|---|
| Targets/IC50/Ki |
β1-adrenoceptor
0.8 nM
|
| In vitro |
Nebivolol shows high affinity and selectivity for beta 1-adrenergic receptor sites in a rabbit lung membrane preparation (Ki value = 0.9 nM and beta 2/beta 1 ratio = 50). Nebivolol displays β1-adrenoceptor selectivity with the Ki(β2)/Ki(β1) value of 40.7 judged by competition experiments to 3H-CGP 12.1777 in the presence of CGP 207.12 A (300 nM, Kiβ2) or ICI 118.551 (50 nM, Kiβ1). Nebivolol reduces cell proliferation of human coronary smooth muscle cells (haCSMCs) and endothelial cells (haECs) in a concentration- and time-dependent maner. Nebivolol treatment for 7 days causes significant reduction in cell growth of haCSMCs with IC50 of 6.1 μM, and inhibits accelerated haCSMC proliferation stimulated by growth factors PDGF-BB, bFGF, and TGFβ with IC50 values of 6.8 μM, 6.4 μM and 7.7 μM, repectively. Nebivolol treatment (10-5 M) of haCSMCs for 48 hours induces a moderate apoptosis of 23% and a decrease from 16% to 5% in the number of cells in S-phase. During Nebivolol incubation, NO formation of HaCEs increases, while endothelin-1 transcription and secretion are suppressed. |
| In vivo |
Administratiion of Nebivolol (initially by iv within 10 minutes of reperfusion and then orally) to rats with myocardial infarction (MI) reduces myocardial apoptosis, which is mediated by regulation of NO . Nebivolol, significantly, prevents left ventricular (LV) pressure changes, reduces total and regional apoptotic cardiomyocytes. Nebivolol treatment lowers mean blood pressure (MBP) in rats with MI slightly, but not significantly. |
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06004453 | Recruiting | Heart Failure |
Novartis Pharmaceuticals|Novartis |
April 4 2023 | -- |
| NCT04888728 | Completed | Idiopathic Pulmonary Fibrosis |
Daewoong Pharmaceutical Co. LTD. |
June 30 2021 | Phase 1 |
| NCT03847350 | Completed | Essential Hypertension |
A.Menarini Asia-Pacific Holdings Pte Ltd |
July 1 2015 | -- |
| NCT01885988 | Unknown status | Erectile Dysfunction |
Martin M. Miner MD|Forest Laboratories|The Miriam Hospital |
March 2013 | Phase 4 |
| NCT01057251 | Completed | Hypertension |
Forest Laboratories |
March 2010 | Phase 4 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































