research use only
LCL161 IAP inhibitor
Cat.No.S7009
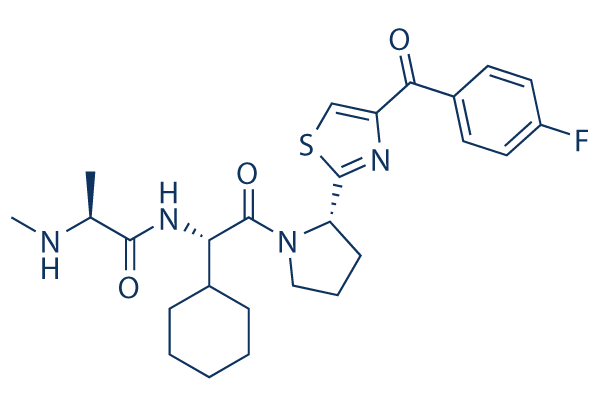
Chemical Structure
Molecular Weight: 500.63
Quality Control
| Related Targets | Bcl-2 Caspase PD-1/PD-L1 Ferroptosis p53 Apoptosis related Synthetic Lethality STAT TNF-alpha Ras |
|---|---|
| Other IAP Inhibitors | BV6 SM-164 Birinapant (TL32711) Xevinapant (AT406) GDC-0152 AZD5582 Tolinapant (ASTX660) |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| LOX | Function assay | 100 mg/kg | 8 hrs | Potentiation of conatumumab-induced cIAP1 degradation in human LOX cells xenografted in mouse at 100 mg/kg, po after 8 hrs by Western blotting analysis | 24083782 | |
| LOX | Function assay | 8 hrs | Potentiation of conatumumab-induced caspase 3/7 activation in human LOX cells xenografted in mouse after 8 hrs by fluorescence assay | 24083782 | ||
| MDA-MB-231 | Function assay | 2.5 to 10 uM | 19 hrs | Binding affinity to cIAP1 BIR3 domain in human MDA-MB-231 cells assessed as increase in TNFalpha level at 2.5 to 10 uM after 19 hrs by ELISA | 24083782 | |
| BxPC3 | Function assay | 3.3 uM | 5 days | Potentiation of conatumumab-induced cytotoxicity against human BxPC3 cells at 3.3 uM after 5 days by MTS assay | 24083782 | |
| HCT15 | Function assay | 3.3 uM | 5 days | Potentiation of conatumumab-induced cytotoxicity against human HCT15 cells at 3.3 uM after 5 days by MTS assay | 24083782 | |
| SW620 | Function assay | 1.1 uM | 5 days | Potentiation of conatumumab-induced cytotoxicity against human SW620 cells at 1.1 uM after 5 days by MTS assay | 24083782 | |
| SW620 | Function assay | 3.3 uM | 5 days | Potentiation of conatumumab-induced cytotoxicity against human SW620 cells at 3.3 uM after 5 days by MTS assay | 24083782 | |
| LOX | Function assay | 3.3 uM | 3 days | Potentiation of conatumumab-induced cytotoxicity against human LOX cells at 3.3 uM after 3 days by MTS assay | 24083782 | |
| LS180 | Function assay | 3.3 uM | 5 days | Potentiation of conatumumab-induced cytotoxicity against human LS180 cells at 3.3 uM after 5 days by MTS assay | 24083782 | |
| H460 | Function assay | 3.3 uM | 5 days | Potentiation of conatumumab-induced cytotoxicity against human H460 cells at 3.3 uM after 5 days by MTS assay | 24083782 | |
| LOX | Function assay | 100 mg/kg | 8 hrs | Plasma concentration in nude mouse xenografted with human LOX cells at 100 mg/kg, po measured at 8 hrs, Cp = 3.3 μM. | 24093940 | |
| SW620 | Function assay | 5 days | Induction of sensitization of human SW620 cells to conatumumab-induced apoptosis assessed as cell viability after 5 days by MTS assay, EC90 = 6.66 μM. | 24093940 | ||
| LOX | Function assay | 100 mg/kg | 8 hrs | Drug uptake in tumor of nude mouse xenografted with human LOX cells at 100 mg/kg, po measured at 8 hrs, Drug uptake = 18.4 μM. | 24093940 | |
| SW620 | Function assay | 2.5 uM | 5 days | Induction of sensitization of human SW620 cells to conatumumab-induced apoptosis assessed as cell viability at 2.5 uM after 5 days by MTS assay | 24093940 | |
| LOX | Function assay | 100 mg/kg | 8 hrs | In vivo inhibition of XIAP BIR2 domain in human LOX cells xenografted in nude mouse assessed as potentiation of conatumumab-induced caspase 3/7 activity at 100 mg/kg, po after 8 hrs by Western blot analysis | 24093940 | |
| LOX | Function assay | 100 mg/kg | 8 hrs | In vivo inhibition of XIAP BIR2 domain in human LOX cells xenografted in nude mouse assessed as increase in caspase 3/7 activity at 100 mg/kg, po after 8 hrs by Western blot analysis | 24093940 | |
| MDA-MB-231 | Function assay | 0.37 to 3.3 uM | 19 hrs | Inhibition of cIAP1/2 in human MDA-MB-231 cells assessed as induction of TNFalpha level at 0.37 to 3.3 uM after 19 hrs by ELISA | 24093940 | |
| CHL1 | Function assay | 0.4 to 10 uM | 28 hrs | Inhibition of cIAP1 in human CHL1 cells at 0.4 to 10 uM after 28 hrs by Western blot analysis | 24093940 | |
| SKMES1 | Function assay | 2.5 uM | 5 days | Potentiation of conatumumab-induced apoptosis in human SKMES1 cells at 2.5 uM after 5 days by MTS assay | 24093940 | |
| Capan1 | Function assay | 2.5 uM | 5 days | Potentiation of conatumumab-induced apoptosis in human Capan1 cells at 2.5 uM after 5 days by MTS assay | 24093940 | |
| AGS | Function assay | 2.5 uM | 5 days | Induction of sensitization of human AGS cells to conatumumab-induced apoptosis assessed as cell viability at 2.5 uM after 5 days by MTS assay | 24093940 | |
| U118MG | Function assay | 2.5 uM | 5 days | Potentiation of conatumumab-induced apoptosis in human U118MG cells at 2.5 uM after 5 days by MTS assay | 24093940 | |
| PC3 | Function assay | 2.5 uM | 5 days | Induction of sensitization of human PC3 cells to conatumumab-induced apoptosis assessed as cell viability at 2.5 uM after 5 days by MTS assay | 24093940 | |
| CCRF-CEM | Antiproliferative assay | Antiproliferative activity against human CCRF-CEM cells, GI50 = 0.25 μM. | 28435526 | |||
| MDA-MB-231 | Function assay | 2 hrs | Induction of cIAP1 degradation in human MDA-MB-231 cells after 2 hrs, EC50 = 0.0004 μM. | 28492317 | ||
| MDA-MB-231 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human MDA-MB-231 cells assessed as decrease in cell proliferation after 72 hrs by alamar blue assay, EC50 = 0.0078 μM. | 28492317 | ||
| HEK293 | Function assay | 2 hrs | Inhibition of full length FLAG-tagged XIAP (unknown origin) interaction with full length untagged caspase-9 expressed in HEK293 cells after 2 hrs by immunoprecipitation assay, EC50 = 0.035 μM. | 28492317 | ||
| MDA-MB-231 | Antitumor assay | 30 mg/kg | 24 days | Antitumor activity against human MDA-MB-231 cells xenografted in Balb/c SCID mouse assessed as tumor growth inhibition at 30 mg/kg administered via oral gavage for 24 days | 28492317 | |
| MDA-MB-231 | Function assay | 2 hrs | Induction of intracellular cIAP1 degradation in human MDA-MB-231 cells after 2 hrs, IC50 = 0.0004 μM. | 30091600 | ||
| MDA-MB-231 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MDA-MB-231 cells after 72 hrs by Alamar blue assay, IC50 = 0.0078 μM. | 30091600 | ||
| HEK293 | Function assay | 2 hrs | Inhibition of full length FLAG-tagged XIAP (unknown origin) interaction with full length untagged caspase-9 expressed in HEK293 cells after 2 hrs by immunoprecipitation assay, IC50 = 0.035 μM. | 30091600 | ||
| SKOV3 | Apoptosis assay | 48 hrs | Induction of apoptosis in human SKOV3 cells assessed caspase-3 activation after 48 hrs by IncuCyte S3 live-cell analysis, EC50 = 0.001 μM. | 31095386 | ||
| SKOV3 | Apoptosis assay | 24 hrs | Induction of apoptosis in human SKOV3 cells assessed caspase-3 activation after 24 hrs by IncuCyte S3 live-cell analysis, EC50 = 0.003 μM. | 31095386 | ||
| BL21(DE3) | Function assay | 2 hrs | Displacement of biotinylated AVPF from N-terminal His tagged recombinant human XIAP-BIR3 domain (253 to 347 residues) expressed in Escherichia coli BL21(DE3) cells incubated for 2 hrs by DELFIA, IC50 = 0.048 μM. | 31095386 | ||
| BL21(DE3) | Function assay | 8 hrs | Displacement of biotinylated AVPF from N-terminal His tagged recombinant human XIAP-BIR3 domain (253 to 347 residues) expressed in Escherichia coli BL21(DE3) cells incubated for 8 hrs by DELFIA, IC50 = 0.053 μM. | 31095386 | ||
| A549 | Function assay | 1 uM | 3 hrs | Induction of cIAP2 degradation in human A549 cells assessed as reduction in cIAP2 protein level at 1 uM incubated for 3 hrs by Western blot analysis | 31550155 | |
| SK-MEL-28 | Function assay | 1 uM | 3 hrs | Induction of cIAP1 degradation in human SK-MEL-28 cells assessed as reduction in cIAP1 protein level at 1 uM incubated for 3 hrs by Western blot analysis | 31550155 | |
| SK-MEL-28 | Function assay | 1 uM | 3 hrs | Induction of cIAP2 degradation in human SK-MEL-28 cells assessed as reduction in cIAP2 protein level at 1 uM incubated for 3 hrs by Western blot analysis | 31550155 | |
| HEK293T | Function assay | 10 uM | 6 hrs | Covalent binding affinity to HA-BIR3 domain of XIAP (unknown origin) expressed in HEK293T cells assessed as increase in band intensity at 10 uM incubated for 6 hrs by Western blot analysis | 31550155 | |
| A549 | Function assay | 1 uM | 3 hrs | Induction of cIAP1 degradation in human A549 cells assessed as reduction in cIAP1 protein level at 1 uM incubated for 3 hrs by Western blot analysis | 31550155 | |
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 100 mg/mL
(199.74 mM)
Ethanol : 100 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 500.63 | Formula | C26H33FN4O3S |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1005342-46-0 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | CC(C(=O)NC(C1CCCCC1)C(=O)N2CCCC2C3=NC(=CS3)C(=O)C4=CC=C(C=C4)F)NC | ||
Mechanism of Action
| Targets/IC50/Ki |
cIAP
XIAP
|
|---|---|
| In vitro |
LCL161 binds to inhibitors of apoptosis proteins (IAPs) with high affinity and initiates the destruction of cIAP1 and cIAP2, which further induces apoptosis via caspase activation. This compound modestly inhibits the growth of FLT3-ITD-expressing cells when administered alone, with an IC50 ranging from ~0.5 μM (Ba/F3-FLT3-ITD cells) to ~4 μM (MOLM13-luc+ cells). The potency of this agent against the D835Y mutant is observed to be considerably higher, with an IC50 of ~50 nM when tested against Ba/F3-D835Y cells. Treatment of MOLM13-luc+ cells with a combination of this compound and PKC412 leads to significantly more killing of cells than either agent alone, with Calcusyn combination indices suggestive of synergy. PKC412 and this chemical induces apoptosis of MOLM13-luc+ cells. The combination of PKC412 and this agent leads to a higher induction of apoptosis than either agent alone. It is able to override stromal-mediated rescue of mutant FLT3-expressing cells through positive combination with PKC412. This compound inhibits the growth of Ba/F3.p210 cells with an IC50 of ~100 nM. It also has demonstrated activity against drug-resistant cells expressing point mutations in the target proteins. This agent at 1000 nM is able to mostly or completely kill Ba/F3-derived cell lines conferring resistance to PKC412, which express FLT3-ITD harboring point mutations in the ATP-binding pocket of FLT3. It also shows activity at concentrations ranging from 100 to 1000 nM against Ba/F3 cells expressing various. This compound is evaluated against the 23 cell lines in the Pediatric Preclinical Testing Program (PPTP) in vitro panel using 96 hr. It achieves 50% growth inhibition against only 3 of the 23 tested PPTP cell lines under concentration of 10 μM. The three cell lines includes two T-cell ALL cell lines (COG-LL-317 and CCRF-CEM) and an anaplastic large cell lymphoma cell line (Karpas-299), with CCRF-CEM and Karpas-299 showing the lowest relative IC50 values (0.25 and 1.6 μM, respectively). It shows immunomodulatory properties on human immune subsets. T lymphocytes treated with this compound demonstrates significantly enhanced cytokine secretion upon activation, with little effect on CD4 and CD8 T-cell survival or proliferation. This agent treatment of peripheral blood mononuclear cells significantly enhances priming of naïve T cells with synthetic peptides in vitro. Myeloid dendritic cells undergoes phenotypic maturation upon this chemical and demonstrates a reduced capacity to cross-present a tumor antigen-based vaccine. These effects are potentially mediated through an observed activation of the canonical and non-canonical NF-κB pathways, following this compound with a resulting upregulation of anti-apoptotic molecules. |
| In vivo |
LCL161 significantly enhances the ability of PKC412 to inhibit the growth of Ba/F3-FLT3-ITD-luc+ cells in vivo. This compound is also shown to positively combine with the standard chemotherapeutic agents, Ara-c and doxorubicin, against FLT3-ITD-expressing cells and against D835Y-expressing cells. There is an additive effect achieved by combining both this chemical in suppressing leukemia growth. This agent (100 mg/kg) enhances in vivo effects of high-moderate doses on leukemia burden in mice. This compound is tested against the Pediatric Preclinical Testing Program (PPTP) in vivo panels (30 or 75 mg/kg [solid tumors] or 100 mg/kg [ALL]) administered orally twice in a week. It induces significant differences in EFS distribution in approximately one-third of solid tumor xenografts (osteosarcoma and glioblastoma), but not in ALL xenografts. No objective tumor responses are observed. In vivo this chemical demonstrates limited single agent activity against the pediatric preclinical models studied. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | cIAP1 / cIAP2 / XIAP / surivivin |
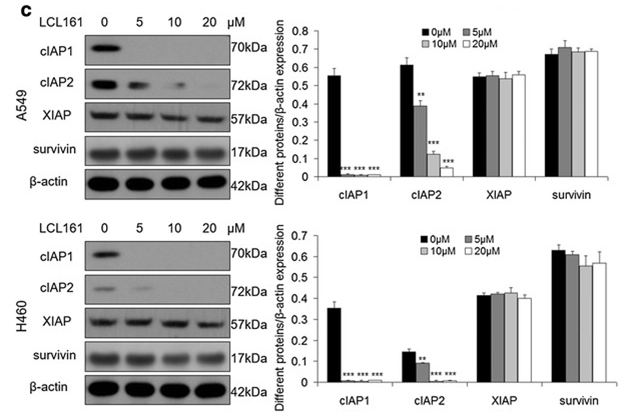
|
27737687 |
| Growth inhibition assay | Cell viability |
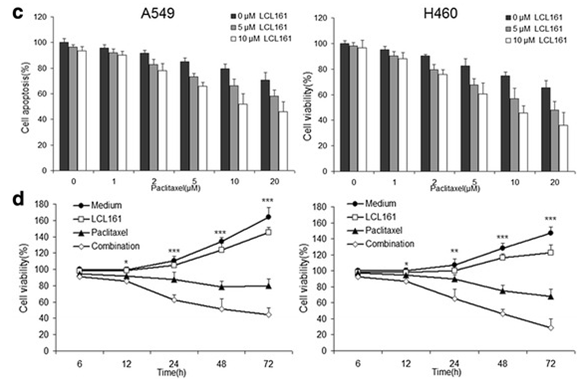
|
27737687 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT03111992 | Completed | Multiple Myeloma |
Novartis Pharmaceuticals|Novartis |
December 18 2017 | Phase 1 |
| NCT01934634 | Unknown status | Metastatic Pancreatic Cancer |
US Oncology Research|Novartis Pharmaceuticals|Delta Clinical Research LLC |
March 2014 | Phase 1 |
| NCT01968915 | Completed | Neoplasms |
Novartis Pharmaceuticals|Novartis |
November 2013 | Phase 1 |
| NCT01617668 | Completed | Breast Cancer |
Novartis Pharmaceuticals|Novartis |
August 2012 | Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































