research use only
Riluzole Sodium Channel inhibitor
Cat.No.S1614
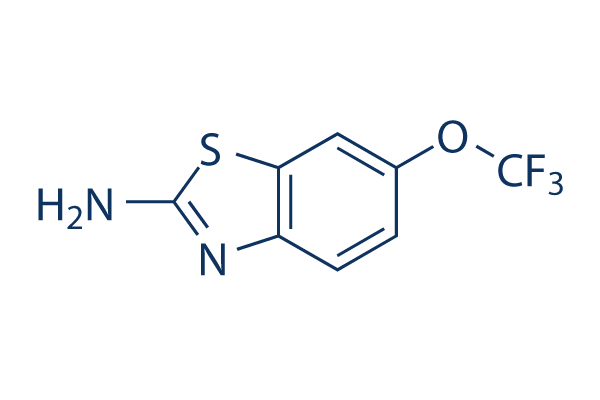
Chemical Structure
Molecular Weight: 234.2
Quality Control
| Related Targets | CFTR CRM1 CD markers AChR Calcium Channel Potassium Channel GABA Receptor TRP Channel ATPase GluR |
|---|---|
| Other Sodium Channel Inhibitors | Camostat Mesilate A-803467 cariporide Veratramine Tolperisone HCl Bulleyaconi cine A Vinpocetine Tenapanor PF-06869206 Sparteine |
Solubility
|
In vitro |
DMSO
: 47 mg/mL
(200.68 mM)
Ethanol : 47 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 234.2 | Formula | C8H5F3N2OS |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1744-22-5 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | RP-54274, PK 26124 | Smiles | C1=CC2=C(C=C1OC(F)(F)F)SC(=N2)N | ||
Mechanism of Action
| Targets/IC50/Ki |
Sodium channel
NMDA receptor
Glutamate release
|
|---|---|
| In vitro |
Riluzole inhibits the release of glutamic acid from cultured neurons, and from brain slices. These effects may be partly due to inactivation of voltage-dependent sodium channels on glutamatergic nerve terminals, as well as activation of a G-protein-dependent signal transduction process. Electrophysiologic experiments performed on isolated excitatory amino acid receptors expressed in the Xenopus oocyte have revealed that this compound inhibits currents evoked by N-methyl-D-aspartate (NMDA) (IC50 = 18 μM) and kainic acid (IC50 = 167 μM). It has been shown to stabilize inactivated sodium channels in frog sciatic nerve, in rat cerebellar granule cells, and on recombinant rat sodium channels expressed in Xenopus oocytes (Ki = 0.2 μM). This chemical also blocks some of the postsynaptic effects of glutamic acid by noncompetitive blockade of NMDA receptors. It protects cultured neurons from anoxic damage, from the toxic effects of glutamic-acid-uptake inhibitors, and from the toxic factor in the CSF of patients with amyotrophic lateral sclerosis.
|
| In vivo |
Riluzole can easily cross the blood-brain barrier. This compound has neuroprotective, anticonvulsant, and sedative properties in vivo. In a rodent model of transient global cerebral ischemia, a complete suppression of the ischemia-evoked surge in glutamic acid release has been observed by this chemical treatment (8 mg/kg i.p.).
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
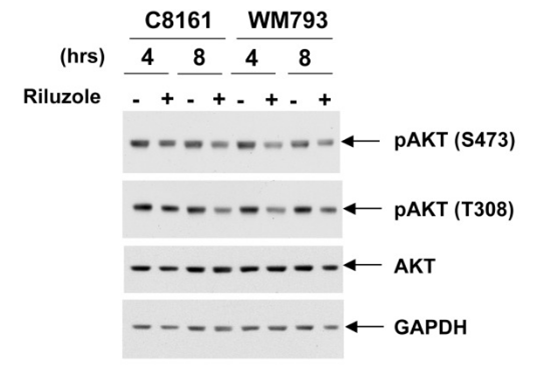
| Western blot | p-AKT / AKT p-Smad2 / Smad2 / p-Smad3 / Smad3 |
|
23077590 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06270108 | Recruiting | Treatment-resistant Schizophrenia|Treatment-responsive Schizophrenia|Healthy Controls |
King''s College London|South London and Maudsley NHS Foundation Trust |
November 14 2022 | Early Phase 1 |
| NCT05292209 | Recruiting | Atrial Fibrillation Paroxysmal |
University of Utah|Ohio State University |
June 15 2022 | Phase 2 |
| NCT04819438 | Completed | Bioequivalence |
Cross Research S.A.|Zambon SpA |
January 15 2021 | Phase 1 |
| NCT02859792 | Recruiting | Spinal Cord Injury |
Assistance Publique Hopitaux De Marseille |
May 27 2019 | Phase 2 |
| NCT04630444 | Completed | Posttraumatic Stress Disorder |
Mclean Hospital|Brain & Behavior Research Foundation |
March 16 2017 | Early Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































