research use only
Vitamin A Acetate Vitamin chemical
Cat.No.S4083
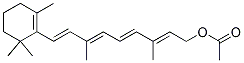
Chemical Structure
Molecular Weight: 328.49
Quality Control
Solubility
|
In vitro |
DMSO
: 65 mg/mL
(197.87 mM)
Ethanol : 65 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 328.49 | Formula | C22H32O2 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 127-47-9 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | Retinyl (Retinol) Acetate | Smiles | CC1=C(C(CCC1)(C)C)C=CC(=CC=CC(=CCOC(=O)C)C)C | ||
Mechanism of Action
| In vitro |
Vitamin A deficiency impairs innate immunity by impeding normal regeneration of mucosal barriers damaged by infection, and by diminishing the function of neutrophils, macrophages, and natural killer cells. Vitamin A is also required for adaptive immunity and plays a role in the development of T both-helper (Th) cells and B-cells. Vitamin A deficiency diminishes antibody-mediated responses directed by Th2 cells, although some aspects of Th1-mediated immunity are also diminished.
|
|---|---|
| In vivo |
Vitamin A acetate (VAA) (fed on an otherwise conventional diet) responds to 105 semiallogeneic cells (a suboptimal dose) in a host-versus-graft (HvG) reaction in mice, whereas mice on a conventional diet do not. Vitamin A acetate can bring a solid and long-lasting state of tolerance induced by the intravenous injection into newborn CBA mice of lymphoid cells from (CBA X C57BL/10ScSn) F1 hybrids to an end, the effect of which is to increase the proportion of the moiety of the T-cell population that produces IL-2. Vitamin A acetate-supplemented diet develops a positive skin reaction to purified protein derivative of mycobacteria in High-dose Mycobacterium bovis-infected mice, and their spleen cells show an increased IL-2 production in vitro.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06319079 | Not yet recruiting | Lactating Mothers|Infants |
Instituto de Desarrollo e Investigaciones Pediátricas Prof. Dr. Fernando E. Viteri |
March 2024 | Not Applicable |
| NCT06058442 | Recruiting | Gastrectomy |
University of Leipzig |
January 24 2024 | Phase 3 |
| NCT06057597 | Recruiting | Obesity|Morbid |
Assistance Publique - Hôpitaux de Paris |
November 13 2023 | Not Applicable |
| NCT06081114 | Recruiting | Micronutrient Status |
Johns Hopkins Bloomberg School of Public Health|International Center for Diarrheal Disease Research Bangladesh (icddrb)|Bill and Melinda Gates Foundation |
October 22 2023 | Not Applicable |
| NCT06155448 | Active not recruiting | Malaria |
Malaria Consortium|Nigerian Institute of Medical Research|London School of Hygiene and Tropical Medicine|Northwestern University |
August 8 2023 | Not Applicable |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































