research use only
TAK-659 Hydrochloride Syk inhibitor
Cat.No.S8442
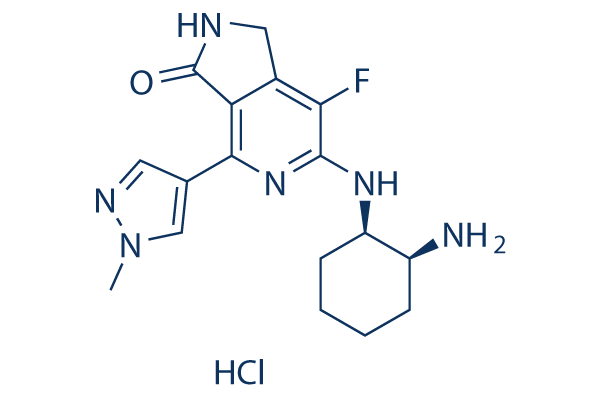
Chemical Structure
Molecular Weight: 380.85
Quality Control
| Related Targets | EGFR VEGFR JAK PDGFR FGFR Src HIF FLT FLT3 HER2 |
|---|---|
| Other Syk Inhibitors | R406 R406 (free base) PRT062607 (P505-15) HCl Entospletinib (GS-9973) Piceatannol BAY 61-3606 dihydrochloride PRT-060318 2HCl Lanraplenib (GS-SYK) RO9021 R112 |
Solubility
|
In vitro |
Water : 2 mg/mL
DMSO
: Insoluble
Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 380.85 | Formula | C17H21FN6 .HCl |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1952251-28-3 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | CN1C=C(C=N1)C2=NC(=C(C3=C2C(=O)NC3)F)NC4CCCCC4N.Cl | ||
Mechanism of Action
| Targets/IC50/Ki |
Syk
(Cell-free assay) 3.2 nM
FLT3
(Cell-free assay) 4.6 nM
ZAP-70
(TR-FRET assays) 75 nM
JAK3
(Cell-free assay) 114 nM
VEGFR2
(Cell-free assay) 135 nM
|
|---|---|
| In vitro |
In a cell proliferation assay, TAK-659 shows inhibition toward a SYK-dependent cell line (OCI-LY10). the sensitivity to TAK-659 is associated with mutations impacting SYK activity in B cell lymphomas, whereas TAK-659 is not cytotoxic for adherent primary or solid tumor cell lines. In cell viability assays, TAK-659 is shown to be sensitive toward FLT3-ITD dependent cell lines, MV4-11 and MOLM-13 while the WT FLT3 RS4-11 (ALL cell line) and RA1 (Burkitt's Lymphoma cell line) are not sensitive toward TAK-659. In cultured human tumor cells, TAK-659 potently inhibits the growth of hematopoietic-derived cell lines, with a concentration producing half-maximal response (EC50) ranging from 11 to 775 nM in sensitive cell systems (eg, diffuse large B-cell lymphoma, and AML). In a broad kinase panel, TAK-659 demonstrates a more than 50-fold selectivity for SYK and FLT-3 over 290 other protein kinases. Treatment with TAK-659 inhibits Syk activation and BCR signaling in co-cultured primary CLL cells and Burkitt's lymphoma cells. In primary CLL cells in suspension culture, TAK-659 treatment results in a dose-dependent reduction in the phosphorylation of SykTyr525, Btk, NFκB, ERK1/2 and STAT3 after BCR stimulation. Inhibition of Syk by TAK-659 induces apoptosis of CLL cells and abrogates BCR and co-culture-derived survival signals. TAK-659 inhibits chemotaxis toward BMSC, CXCL12 and CXCL13 in primary CLL cells, and abrogates microenvironment-induced chemoresistance. TAK-659 does not inhibit TCR signaling and molecular features of T cell activation in primary T cells from patients with CLL.
|
| In vivo |
TAK-659 blocks anti-IgD (immune-globulin D antibody) stimulated CD86 expression in mouse peripheral B cells in vivo. In the FLT3-dependent MV4-11 xenograft model, TAK-659 shows tumor regression at 60 mg/kg daily after 20 days of dosing. Preliminary plasma and urine PK data show that TAK-659 was absorbed quickly (median Tmax 2-3 hrs), with moderate variability in steady-state exposures (40-50% CV for DN-AUCtau), mean peak/trough ratio of 3.2–4.2, and mean accumulation of 2.1- to 2.6-fold after 15 d QD dosing. Renal clearance (CLr) of unchanged drug accounts for 30–34% of apparent oral clearance, suggesting a CLr contribution of ≥30–34% to TAK-659 systemic clearance. Oral TAK-659 has an acceptable PK and safety profile in pts with solid tumors or lymphoma, supporting continuous oral QD dosing.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT03772288 | Withdrawn | Lymphoma Non-Hodgkin |
Calithera Biosciences Inc|Nektar Therapeutics |
April 3 2019 | Phase 1 |
| NCT03338881 | Withdrawn | Advanced Solid Neoplasms|Lymphoma Neoplasms |
Calithera Biosciences Inc |
May 10 2018 | Phase 1 |
| NCT03359733 | Withdrawn | Lymphoma Malignant|Advanced Solid Neoplasms |
Calithera Biosciences Inc |
February 28 2018 | Phase 1 |
| NCT03357627 | Completed | Lymphoma Non-Hodgkin|Lymphoma Large B-cell Diffuse|Lymphoma Follicular |
Calithera Biosciences Inc |
February 16 2018 | Phase 1 |
| NCT03123393 | Terminated | Diffuse Large B-cell Lymphoma |
Calithera Biosciences Inc |
October 10 2017 | Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































