research use only
Sitravatinib (MGCD516) c-Kit inhibitor
Cat.No.S8573
Chemical Structure
Molecular Weight: 629.68
Quality Control
| Related Targets | EGFR VEGFR PDGFR FGFR c-Met Src MEK CSF-1R FLT3 HER2 |
|---|---|
| Other c-Kit Inhibitors | Masitinib Amuvatinib (MP-470) OSI-930 PDGFR inhibitor 1 ISCK03 Elenestinib phosphate M4205(IDRX-42) Masitinib mesylate AZD3229 Bezuclastinib |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| KB-8-5-11 | qHTS assay | P-glycoprotein substrates identified in KB-8-5-11 adenocarcinoma cell line, qHTS therapeutic library screen, Potency = 4.6109 μM. | 31515284 | |||
| KB-3-1 | qHTS assay | P-glycoprotein substrates identified in KB-3-1 adenocarcinoma cell line, qHTS therapeutic library screen | 31515284 | |||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 100 mg/mL
(158.81 mM)
Ethanol : 100 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 629.68 | Formula | C33H29F2N5O4S |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1123837-84-2 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | MG-516 | Smiles | COCCNCC1=CN=C(C=C1)C2=CC3=NC=CC(=C3S2)OC4=C(C=C(C=C4)NC(=O)C5(CC5)C(=O)NC6=CC=C(C=C6)F)F | ||
Mechanism of Action
| Targets/IC50/Ki |
DDR2
(Cell-free assay) 0.5 nM
EPHA3
(Cell-free assay) 1 nM
Axl
(Cell-free assay) 1.5 nM
Mer
(Cell-free assay) 2 nM
VEGFR3 (FLT4)
(Cell-free assay) 2 nM
VEGFR2 (KDR)
(Cell-free assay) 5 nM
TRKA (NTRK1)
(Cell-free assay) 5 nM
VEGFR1 (FLT1)
(Cell-free assay) 6 nM
Kit
(Cell-free assay) 6 nM
FLT3
(Cell-free assay) 8 nM
TRKB (NTRK2)
(Cell-free assay) 9 nM
EPHB2
(Cell-free assay) 10 nM
EPHB4
(Cell-free assay) 12 nM
Met
(Cell-free assay) 20 nM
DDR1
(Cell-free assay) 29 nM
PDGFRα
(Cell-free assay) 30 nM
RON
(Cell-free assay) 43 nM
RET
(Cell-free assay) 44 nM
EphA2
(Cell-free assay) 44 nM
ROS
(Cell-free assay) 59 nM
EPHA4
(Cell-free assay) 76 nM
Src
(Cell-free assay) 156 nM
|
|---|---|
| In vitro |
Sitravatinib (MGCD516) is an oral, potent small molecule inhibitor of a closely related spectrum of RTKs including RET, the split RTKs (VEGFR, PDGFR and KIT), TRK family, DDR2, MET and AXL. Treatment with this compound resulted in significant blockade of phosphorylation of potential driver RTKs and induced potent anti-proliferative effects in vitro. |
| In vivo |
Sitravatinib (MGCD516) has demonstrated antitumor activity in nonclinical cancer models harboring genetic alterations of its targets, including rearrangement of RET, NTRK, or CHR4q12 amplification. Treatment of tumor xenografts in vivo with this compound resulted in significant suppression of tumor growth. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Growth inhibition assay | Cell viability |
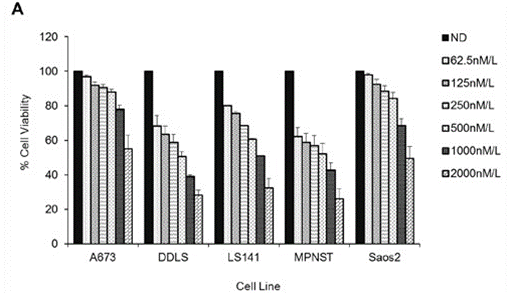
|
26675259 |
| Western blot | p-IGF1-R / IGF1-R / p-PDGFRβ / PDGFRβ/ p-AKT / AKT |
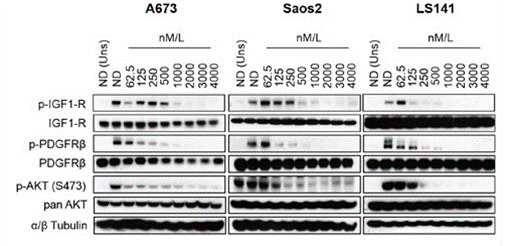
|
26675259 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT04935112 | Completed | Healthy Adults |
Mirati Therapeutics Inc. |
July 7 2021 | Phase 1 |
| NCT04921358 | Terminated | Non-Small Cell Lung Cancer (NSCLC) |
BeiGene |
July 27 2021 | Phase 3 |
| NCT04772612 | Completed | Hepatic Impairment |
Mirati Therapeutics Inc. |
March 12 2021 | Phase 1 |
| NCT04800614 | Completed | Healthy Adults |
Mirati Therapeutics Inc. |
March 15 2021 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































