research use only
CFI-400945 PLK inhibitor
Cat.No.S7552
Chemical Structure
Molecular Weight: 534.65
Quality Control
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| HCT116 | Growth inhibition assay | 5 days | Growth inhibition of human HCT116 cells after 5 days by SRB assay, GI50 = 0.004 μM. | 25723005 | ||
| HCC1954 | Growth inhibition assay | 5 days | Growth inhibition of human HCC1954 cells after 5 days by SRB assay, GI50 = 0.005 μM. | 25723005 | ||
| A549 | Growth inhibition assay | 5 days | Growth inhibition of human A549 cells after 5 days by SRB assay, GI50 = 0.005 μM. | 25723005 | ||
| MDA-MB-468 | Growth inhibition assay | 5 days | Growth inhibition of human MDA-MB-468 cells after 5 days by SRB assay, GI50 = 0.006 μM. | 25723005 | ||
| MCF7 | Growth inhibition assay | 5 days | Growth inhibition of human MCF7 cells after 5 days by SRB assay, GI50 = 0.008 μM. | 25723005 | ||
| COLO205 | Growth inhibition assay | 5 days | Growth inhibition of human COLO205 cells after 5 days by SRB assay, GI50 = 0.017 μM. | 25723005 | ||
| OVCAR3 | Growth inhibition assay | 5 days | Growth inhibition of human OVCAR3 cells after 5 days by SRB assay, GI50 = 0.018 μM. | 25723005 | ||
| BT20 | Growth inhibition assay | 5 days | Growth inhibition of human BT20 cells after 5 days by SRB assay, GI50 = 0.058 μM. | 25723005 | ||
| CAL51 | Growth inhibition assay | 5 days | Growth inhibition of human CAL51 cells after 5 days by SRB assay, GI50 = 0.26 μM. | 25723005 | ||
| SW620 | Growth inhibition assay | 5 days | Growth inhibition of human SW620 cells after 5 days by SRB assay, GI50 = 0.38 μM. | 25723005 | ||
| SKBR3 | Growth inhibition assay | 5 days | Growth inhibition of human SKBR3 cells after 5 days by SRB assay, GI50 = 5.3 μM. | 25723005 | ||
| MDA-MB-231 | Growth inhibition assay | 5 days | Growth inhibition of human MDA-MB-231 cells after 5 days by SRB assay, GI50 = 8.6 μM. | 25723005 | ||
| HMEC | Growth inhibition assay | 5 days | Growth inhibition of human HMEC cells after 5 days by SRB assay, GI50 = 9 μM. | 25723005 | ||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 100 mg/mL
(187.03 mM)
Ethanol : 100 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 534.65 | Formula | C33H34N4O3 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1338806-73-7 | -- | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | CC1CN(CC(O1)C)CC2=CC=C(C=C2)C=CC3=NNC4=C3C=CC(=C4)C5CC56C7=C(C=CC(=C7)OC)NC6=O | ||
Mechanism of Action
| Targets/IC50/Ki |
PLK4
(Cell-free assay) 2.8 nM
TrkA
(Cell-free assay) 6 nM
TrkB
(Cell-free assay) 9 nM
Tie-2
(Cell-free assay) 22 nM
Aurora B
(Cell-free assay) 98 nM
Aurora A
(Cell-free assay) 140 nM
|
|---|---|
| In vitro |
CFI-400945 is a potent, orally active antitumor agent with the IC50 and Ki values of 2.8 and 0.26 nM, respectively. No significant inhibition against PLKs 1-3 is observed for this compound at a concentration of 50 μM. It can attenuate the growth of breast cell line, as well as other tumor cell lines significantly. This compound selectively inhibits PLK4 in cells, but also has certain activity against AURKB, TRKA, TRKB and Tie2/TEK (only 10 kinases showed more than 50% inhibition among 290 kinases). The cytokinesis failure and subsequent polyploidization by this chemical treatment indicate that the cell death in cancer cell lines is at least partly achieved through inhibition of AURKB. No significant inhibition is observed for PLKs 1-3 (IC50s > 50 μM) probably due to the most divergent structure of PLK4 compared to other polo-like kinases 1-3. Cancer cells treated with this agent exhibit effects consistent with PLK4 kinase inhibition, including dysregulated centriole duplication, mitotic defects, and cell death.
|
| In vivo |
CFI-400945 is well tolerated in breast cancer xenograft models, in particular those deficient in the tumor suppressor PTEN. Upon intermittent oral dosing, in a mouse model of colon cancer, this compound is an effective inhibitor of HCT116 tumor growth and was well tolerated. It is absorbed rapidly after oral administration, reaching maximum plasma concentrations (Cmax) of 0.25-11.68 μg/mL for the doses tested (3.75-104 mg/kg). This chemical can inhibit the growth of a range of tumor types and may be effective in a clinical setting even in advanced tumors. Following oral administration of efficacious doses of this compound in mice, plasma levels of it are sustained and remained above both the EC50 value for half-maximal inhibition of cellular PLK4 autophosphorylation and growth inhibition GI50 values for 24 hr. Moreover, it demonstrates dose-dependent antitumor activity. Analysis of xenograft tumors from mice treated with an efficacious dose of this chemical shows a pharmacodynamic effect that is suggestive of complete rather than partial inhibition of PLK4 kinase activity.
|
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Growth inhibition assay | Cell viability |
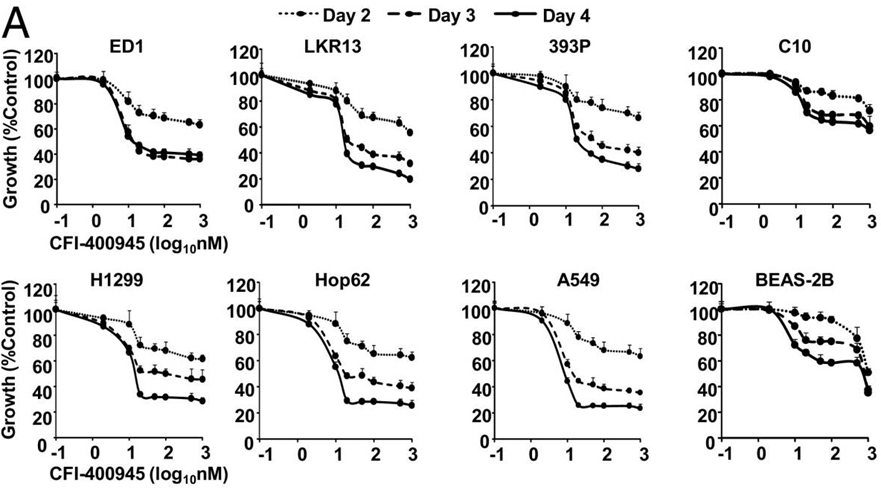
|
29434041 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT04730258 | Recruiting | Acute Myeloid Leukemia|Myelodysplastic Syndromes|Chronic Myelomonocytic Leukemia|AML|MDS|CMML |
Treadwell Therapeutics Inc |
April 16 2021 | Phase 1|Phase 2 |
| NCT04176848 | Unknown status | Breast Cancer |
Canadian Cancer Trials Group|AstraZeneca|University Health Network Toronto |
December 19 2019 | Phase 2 |
| NCT03624543 | Active not recruiting | Breast Cancer |
Canadian Cancer Trials Group|Stand Up To Cancer Canada-Canadian Cancer Society Breast Cancer Dream Team |
February 14 2019 | Phase 2 |
| NCT03187288 | Active not recruiting | Acute Myeloid Leukemia|Myelodysplastic Syndromes|Relapsed Cancer|Refractory Cancer |
University Health Network Toronto |
May 25 2018 | Phase 1 |
| NCT01954316 | Completed | Advanced Cancer |
University Health Network Toronto|The Princess Margaret Cancer Foundation|California Institute for Regenerative Medicine (CIRM) |
March 2014 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































