research use only
Ceftriaxone disodium salt hemi (heptahydrate) Antibiotics chemical
Cat.No.S4158
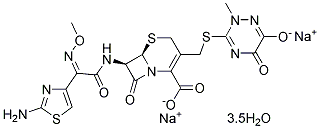
Chemical Structure
Molecular Weight: 661.6
Quality Control
| Related Targets | Integrase Bacterial Anti-infection Fungal Antiviral COVID-19 Parasite Reverse Transcriptase HIV HCV Protease |
|---|---|
| Other Antibiotics Inhibitors | G418 Sulfate (Geneticin) Puromycin Nanchangmycin Sitafloxacin Hydrate Trovafloxacin Mesylate Spiramycin Nadifloxacin G-418 Gamithromycin Thiamphenicol |
Solubility
|
In vitro |
Water : 100 mg/mL
DMSO
: 1 mg/mL
(1.51 mM)
Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 661.6 | Formula | C18H16N8O7S3.7/2H2O.2Na |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 104376-79-6 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | CN1C(=NC(=O)C(=O)N1)SCC2=C(N3C(C(C3=O)NC(=O)C(=NOC)C4=CSC(=N4)N)SC2)C(=O)[O-].[Na+] | ||
Mechanism of Action
| In vitro |
Ceftriaxone disodium salt hemi(heptahydrate) is a third-generation cephalosporin antibiotic has a broad spectrum of activity in vitro which includes Gram-positive and Gram-negative aerobic and some anaerobic bacteria. Both penicillin-sensitive and –resistant strains of Staphyococcus aureus are sensitive to Ceftriaxone (MIC90, 2 to 8 mg/L), but the drug is poorly active against methicillin- or oxacillin-resistant strains. Creftriaxone is similar in activity to several penicillins and other cephalosporins against Streptococcus pyogenes, and is also active in low concentrations against other Streptococcus species. Staphylococcus epidermidis is only moderately sensitive and Streptococcus faecalis is usually resistant to Ceftriaxone. Ceftriaxone is generally more active than cefoperazone, and is similar in activity to cefotaxime and latamoxef against Gram-negative bacilli. 90% of tested strains of most Enterobacteroacae are inhibited by a Ceftriaxone concentration of < 1 mg/L. The reported activity of Ceftriaxone against Pseudomonas aeruginosa varies widely, but generally at least 32 mg/L is required to inhibit most strains, and is thus usually less active than the aminoglycosides, aztreonam, imipenem, cefoperazone, ceftazidime, carumonam and piperacillin. |
References |
|---|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05960006 | Recruiting | Antibiotic Toxicity|Liver Cirrhosis|Renal Insufficiency|Ceftriaxone Overdose|Ascites Hepatic|Infection Bacterial |
Academisch Medisch Centrum - Universiteit van Amsterdam (AMC-UvA)|Erasmus Medical Center |
July 10 2023 | -- |
| NCT04268342 | Active not recruiting | Drug Resistance Microbial|Antimicrobial Stewardship|Gonorrhea |
The University of Queensland|Kirby Institute|Monash University|South Australian Health and Medical Research Institute|University of Melbourne|University of California Los Angeles|Griffith University|Queensland Health|St Vincent''s Hospital Sydney|SpeeDx Pty Ltd|NSW Health Pathology|University of Sydney |
April 26 2022 | Not Applicable |
| NCT04902560 | Completed | Gonorrhea |
University Hospital Montpellier |
May 1 2021 | -- |
| NCT05061875 | Unknown status | Ceftriaxone Poisoning |
Fundación Pública Andaluza para la gestión de la Investigación en Sevilla |
March 1 2021 | -- |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































