research use only
Avapritinib (BLU-285) PDGFRα inhibitor
Cat.No.S8553
Chemical Structure
Molecular Weight: 498.56
Quality Control
| Related Targets | EGFR VEGFR FGFR c-Met Src MEK CSF-1R FLT3 HER2 c-Kit |
|---|---|
| Other PDGFR Inhibitors | CP-673451 Orantinib (SU6668) Tyrphostin AG 1296 Trapidil AZD2932 PP121 Sennoside B Tyrphostin AG1433 AG 1295 N-(p-Coumaroyl) Serotonin |
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| BA/F3 cells | Growth inhibiton assay | 72 h | Inhibition of Kit D816V mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.008 μM. | 30204441 | ||
| BA/F3 cells | Growth inhibiton assay | 72 h | Inhibition of PDGFRalpha V561D/D842V mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.01 μM. | 30204441 | ||
| BA/F3 cells | Growth inhibiton assay | 72 h | Inhibition of Kit exon 11 deletion (557 to 558 residues) and D816H mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.017 μM. | 30204441 | ||
| BA/F3 cells | Growth inhibiton assay | 72 h | Inhibition of Kit exon 11 deletion (557 to 558 residues) and D820A mutant and D820A mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.019 μM. | 30204441 | ||
| BA/F3 cells | Growth inhibiton assay | 72 h | Inhibition of TEL-fused PDGFRbeta (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.022 μM. | 30204441 | ||
| BA/F3 cells | Growth inhibiton assay | 72 h | Inhibition of Kit exon 11 deletion (557 to 558 residues) and A829P mutant and Y823D mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.028 μM. | 30204441 | ||
| BA/F3 cells | Growth inhibiton assay | 72 h | Inhibition of Kit V560D/D816H mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.029 μM. | 30204441 | ||
| BA/F3 cells | Growth inhibiton assay | 72 h | Inhibition of TEL-fused PDGFRalpha (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.034 μM. | 30204441 | ||
| BA/F3 cells | Growth inhibiton assay | 72 h | Inhibition of Kit exon 11 deletion (557 to 558 residues) and N822K mutant and Y823D mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.034 μM. | 30204441 | ||
| BA/F3 cells | Growth inhibiton assay | 72 h | Inhibition of Kit V560D mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.042 μM. | 30204441 | ||
| BA/F3 cells | Growth inhibiton assay | 72 h | Inhibition of Kit exon 9 AY502 to 503 insertion and D816 mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.069 μM. | 30204441 | ||
| BA/F3 cells | Growth inhibiton assay | 72 h | Inhibition of Kit exon 11 deletion (557 to 558 residues) mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.078 μM. | 30204441 | ||
| BA/F3 cells | Growth inhibiton assay | 72 h | Inhibition of Kit exon 11 deletion (560 to 578 residues) mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.117 μM. | 30204441 | ||
| BA/F3 cells | Growth inhibiton assay | 72 h | Inhibition of Kit exon 9 AY502 to 503 insertion mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.167 μM. | 30204441 | ||
| BA/F3 cells | Growth inhibiton assay | 72 h | Inhibition of Kit exon 11 deletion (557 to 558 residues) and V654A mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.292 μM. | 30204441 | ||
| BA/F3 cells | Growth inhibiton assay | 72 h | Inhibition of Kit V560D/V654A mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.427 μM. | 30204441 | ||
| BA/F3 cells | Growth inhibiton assay | 72 h | Inhibition of Kit exon 9 AY502 to 503 insertion and V654 mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.751 μM. | 30204441 | ||
| BA/F3 cells | Growth inhibiton assay | 72 h | Inhibition of Kit exon 11 deletion (557 to 558 residues) and D816H mutant and T670I mutant (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 0.86 μM. | 30204441 | ||
| BA/F3 cells | Cytotoxicity assay | 72 h | Cytotoxicity in mouse parental BA/F3 cells incubated for 72 hrs by MTS assay, GI50 = 4.075 μM. | 30204441 | ||
| BA/F3 cells | Growth inhibiton assay | 72 h | Inhibition of KDR (unknown origin) transfected in mouse BA/F3 cells assessed as inhibition of cell growth incubated for 72 hrs by MTS assay, GI50 = 4.952 μM. | 30204441 | ||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 100 mg/mL
(200.57 mM)
Ethanol : 1.5 mg/mL Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 498.56 | Formula | C26H27FN10 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1703793-34-3 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | BLU-285 | Smiles | CC(C1=CC=C(C=C1)F)(C2=CN=C(N=C2)N3CCN(CC3)C4=NC=NN5C4=CC(=C5)C6=CN(N=C6)C)N | ||
Mechanism of Action
| Targets/IC50/Ki |
PDGFRα (D842V)
0.5 nM
c-Kit (D816V)
0.5 nM
|
|---|---|
| In vitro |
Avapritinib (BLU-285) is a selective oral inhibitor that targets KIT Exon 17 and PDGFRα D842 activation loop mutants. Cellular assays measuring inhibition of KIT mutant autophosphorylation confirm its activity against the KIT D816 mutants D816V (HMC1.2 cells, IC50 = 3 nM) and D816Y (P815 cells, IC50 = 22 nM) as well as other KIT Exon 17 mutants such as N822K (Kasumi cells, IC50 = 40 nM) found in treatment-refractory GIST. |
| In vivo |
In vivo, Avapritinib (BLU-285) is a well-tolerated, orally bioavailable agent that achieves dose dependent tumor growth inhibition in a D816Y-driven xenograft model. A PK-PD-efficacy relationship with this compound has been established demonstrating that tumor regression results from >90% target suppression and is observed with 30 mg/kg once daily dosing. With potent activity against PDGFRα D842V and KIT Exon 17 mutants, it targets previously unaddressed genomic drivers of disease and provides promise for the treatment of PDGFRα D842V-driven GIST(gastrointestinal stromal tumor) or SM(systemic mastocytosis), where more than 90% of patients carry the KIT D816V mutation. Besides single agent activity, the highly selective BLU-285 offers an opportunity for combination with other agents in GIST to cover the entirety of KIT primary and resistance mutants. |
References |
|
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | PDGFRA / c-KIT / Wee1 / Cdc2 / PARP / γH2AX / CyclinD1 / β-actin |
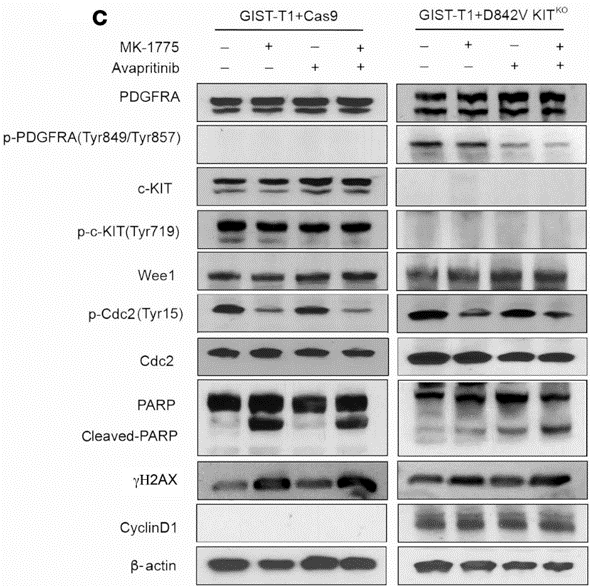
|
33320833 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT04908176 | Completed | Gastrointestinal Stromal Tumors|GIST|Non-resectable Advanced Solid Tumors|Recurrent or Unresectable Central Nervous System (CNS) Tumors |
Blueprint Medicines Corporation |
August 24 2022 | Phase 1 |
| NCT04695431 | Completed | Advanced Systemic Mastocytosis|Aggressive Systemic Mastocytosis|Systemic Mastocytosis With an Associated Hematological Neoplasm|Mast Cell Leukemia |
Blueprint Medicines Corporation|Analysis Group Inc. |
December 2 2020 | -- |
| NCT03731260 | Active not recruiting | Indolent Systemic Mastocytosis |
Blueprint Medicines Corporation |
April 16 2019 | Phase 2 |
| NCT03580655 | Active not recruiting | Advanced Systemic Mastocytosis|Aggressive Systemic Mastocytosis|Systemic Mastocytosis With an Associated Hematologic Neoplasm|Mast Cell Leukemia |
Blueprint Medicines Corporation |
November 21 2018 | Phase 2 |
| NCT03465722 | Completed | GIST |
Blueprint Medicines Corporation |
March 26 2018 | Phase 3 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































