research use only
Tomivosertib (eFT508) MNK1/2 Inhibitor
Cat.No.S8275
Chemical Structure
Molecular Weight: 340.38
Quality Control
Cell Culture, Treatment & Working Concentration
| Cell Lines | Assay Type | Concentration | Incubation Time | Formulation | Activity Description | PMID |
|---|---|---|---|---|---|---|
| TMD8 | Function assay | 0.01, 0.1, 1, 3 and 10 μM | 2 h | potent dose-dependent inhibition of eIF4E Ser209 phosphorylation | 29526098 | |
| TMD8 | Proliferation assay | 12 h | IC50=2.53 μM | 30824167 | ||
| MV4-11 | Proliferation assay | 12 h | IC50=14.49 μM | 30824167 | ||
| K562 | Proliferation assay | 12 h | IC50=13.54 μM | 30824167 | ||
| Vero E6 | Antiviral assay | 48 h | IC50 for antiviral activity against SARS-CoV-2 in the Vero E6 cell line at 48 h by immunofluorescence-based assay (detecting the viral NP protein in the nucleus of the Vero E6 cells)., IC50 = 0.34674 μM. | 32353859 | ||
| Click to View More Cell Line Experimental Data | ||||||
Solubility
|
In vitro |
DMSO
: 13 mg/mL
(38.19 mM)
Water : Insoluble Ethanol : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such
as vortex, ultrasound or hot water bath can be used to aid dissolving.
Chemical Information, Storage & Stability
| Molecular Weight | 340.38 | Formula | C17H20N6O2 |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 1849590-01-7 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | CC1=C2C(=O)NC3(N2C(=O)C(=C1)NC4=NC=NC(=C4)N)CCCCC3 | ||
Mechanism of Action
| Targets/IC50/Ki |
MNK2
(Cell-free assay) 1 nM
MNK1
(Cell-free assay) 2.4 nM
|
|---|---|
| In vitro |
Tomivosertib (eFT-508) has a half-maximal inhibitory concentration (IC50) of 1-2 nM against both MNK isoforms in enzyme assays and inhibits the kinase through a reversible, ATP-competitive mechanism of action. Treatment of tumor cell lines with this compound leads to a dose-dependent reduction in eIF4E phosphorylation at serine 209 (IC50 = 2-16 nM). In a panel of ~50 hematological cancers, this chemical shows anti-proliferative activity against multiple DLBCL cell lines. Sensitivity to this compound in TMD8, OCI-Ly3 and HBL1 DLBCL cell lines is associated with dose-dependent decreases in production of pro-inflammatory cytokines including TNFα, IL-6, IL-10 and CXCL10. |
| In vivo |
Tomivosertib (eFT-508) is well-tolerated and shows efficacy against MyD88-mutant DLBCL models in vivo. Significant anti-tumor activity of this compound is observed in the TMD8 and HBL-1 ABC-DLBCL models(subcutaneous human lymphoma xenograft models), both of which harbor activating MyD88 mutations.
|
References |
Applications
| Methods | Biomarkers | Images | PMID |
|---|---|---|---|
| Western blot | p-eIF4E |
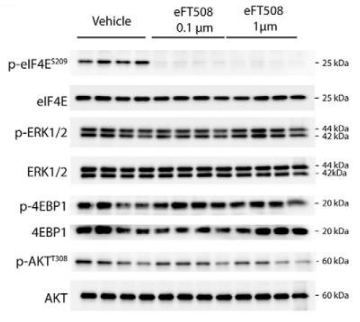
|
30459229 |
| Growth inhibition assay | Cell viability |
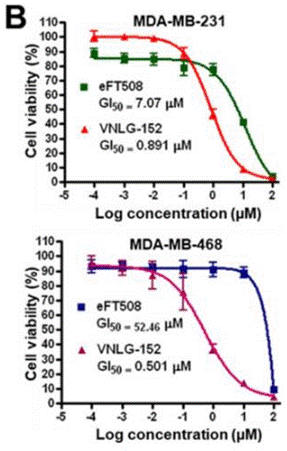
|
30832411 |
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05744739 | Recruiting | Acute Myeloid Leukemia |
Northwestern University|National Cancer Institute (NCI)|EFFECTOR Therapeutics Inc. |
September 29 2023 | Phase 1 |
| NCT04261218 | Completed | Breast Cancer |
Translational Research in Oncology|Effector Therapeutics|Stand Up To Cancer |
August 25 2020 | Phase 1 |
| NCT02937675 | Terminated | Lymphoma |
Effector Therapeutics |
February 8 2017 | Phase 1|Phase 2 |
| NCT02605083 | Terminated | Cancer |
Effector Therapeutics |
December 3 2015 | Phase 1|Phase 2 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.






































